Tuberc Respir Dis.
2007 Jan;62(1):33-42. 10.4046/trd.2007.62.1.33.
The Effect of Inhibition of Heme Oxygenase-1 on Chemosensitivity of Cisplatin in Lung Cancer Cells
- Affiliations
-
- 1Department of Internal Medicine, College of Medicine Wonkwang University, Iksan, Korea. kshryj@wonkwang.ac.kr
- 2Department of Pathology Kunsan Medical Center of Wonkwang University Hospital, Kunsan, Korea.
- KMID: 1518487
- DOI: http://doi.org/10.4046/trd.2007.62.1.33
Abstract
-
BACKGROUND: Heme oxygenase-1 (HO-1) is known to modulates the cellular functions, including cell proliferation and apoptosis. It is known that a high level of HO-1 expression is found in many tumors, and HO-1 plays an important role in rapid tumor growth on account of its antioxidant and antiapoptotic effects. Cisplatin is a widely used anti-cancer agent for the treatment of lung cancer. However, the development of resistance to cisplatin is a major obstacle to its use in clinical treatment. We previously demonstrated that inhibiting HO-1 expression through the transcriptional activation of Nrf2 induces apoptosis in A549 cells. The aim of this study was to determine of the inhibiting HO-1 enhance the chemosensitivity of A549 cells to cisplatin.
MATERIALS AND METHODS
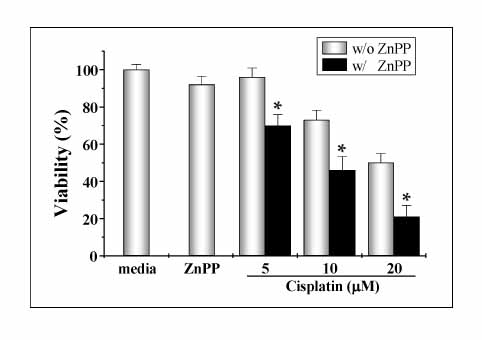
The human lung cancer cell line, A549, was treated cisplatin, and the cell viability was measured by a MTT assay. The change in HO-1, Nrf2, and MAPK expression after the cisplatin treatment was examined by Western blotting. HO-1 inhibition was suppressed by ZnPP, which is a specific pharmacologic inhibitor of HO activity, and small interfering RNA (siRNA). Flow cytometry analysis and Western blot were performed in to determine the level of apoptosis. The level of hydrogen peroxide (H2O2) generation was monitored fluoimetrically using 2',7'-dichlorofluorescein diacetate.
RESULTS
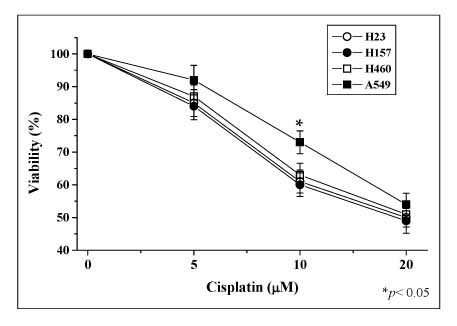
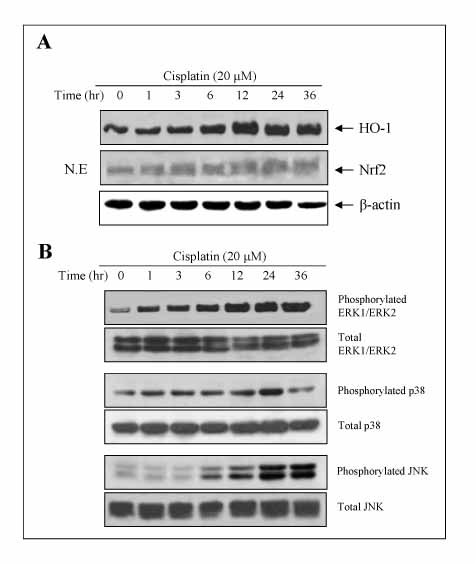
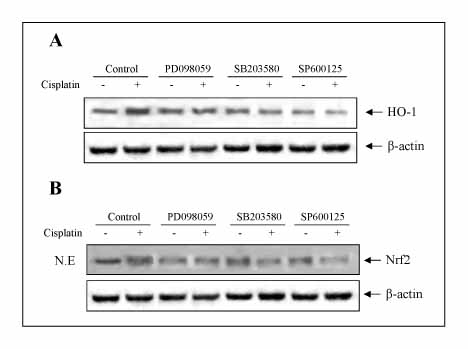
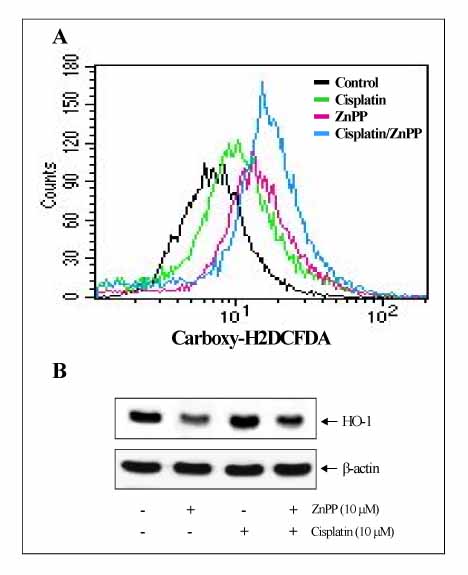
The A549 cells showed more resistance to the cisplatin treatment than the other cell lines examined, whereas cisplatin increased the expression of HO-1 and Nrf2, as well as the phosphorylation of MAPK in a time-dependent fashion. Inhibitors of the MAPK pathway blocked the induction of HO-1 and Nrf2 by the cisplatin treatment in A549 cells. In addition, the cisplatin-treated A549 cells transfected with dither the HO-1 small interfering RNA (siRNA) or ZnPP, specific HO-1 inhibitor, showed in a more significantly decrease in viability than the cisplatin-only-treated group. The combination treatment of ZnPP and cisplatin caused in a marked increase in the ROS generation and a decrease in the HO-1 expression.
CONCLUSION
Cisplatin increases the expression of HO-1, probably through the MAPK-Nrf2 pathway, and the inhibition of HO-1 enhances the chemosensitivity of A549 cells to cisplatin.
Keyword
MeSH Terms
Figure
Reference
-
1. Jaiswal AK. Nrf2 signaling in coordinated activation of antioxidant gene expression. Free Radic Biol Med. 2004. 36:1199–1207.2. Huang HC, Nguyen T, Pickett CB. Phosphorylation of Nrf2 at Ser-40 by protein kinase C regulates antioxidant response element-mediated transcription. J Biol Chem. 2002. 277:42769–42774.3. Alam J, Stewart D, Touchard C, Boinapally S, Choi AM, Cook JL. Nrf2, a Cap'n'Collar transcription factor, regulates induction of the heme oxygenase-1 gene. J Biol Chem. 1999. 274:26071–26078.4. Owuor ED, Kong AN. Antioxidants and oxidants regulated signal transduction pathways. Biochem Pharmacol. 2002. 64:765–770.5. Chen C, Yu R, Owuor ED, Kong AN. Activation of antioxidant-response element (ARE), mitogen-activated protein kinases (MAPKs) and caspases by major green tea polyphenol components during cell survival and death. Arch Pharm Res. 2000. 23:605–612.6. Yu R, Chen C, Mo YY, Hebbar V, Owuor ED, Tan TH, et al. Activation of mitogen-activated protein kinase pathways induces antioxidant response element-mediated gene expression via Nrf2-dependent mechanism. J Biol Chem. 2000. 275:39907–39913.7. Motterlini R, Foresti R, Bassi R, Calabrese V, Clark JE, Green CJ. Endothelial heme oxygenase-1 induction by hypoxia: modulation by inducible nitric-oxide synthase and S-nitrosothiols. J Biol Chem. 2000. 275:13613–13620.8. Doi K, Akaike T, Fujii S, Tanaka S, Ikebe N, Beppu T, et al. Induction of haem oxygenase-1 nitric oxide and ischaemia in experimental solid tumours and implications for tumour growth. Br J Cancer. 1999. 80:1945–1954.9. Elbirt KK, Whitmarsh AJ, Davis RJ, Bonkovsky HL. Mechanism of sodium arsenite-mediated induction of heme oxygenase-1 in hepatoma cells: role of mitogenactivated protein kinases. J Biol Chem. 1998. 273:8922–8931.10. Eyssen-Hernandez R, Ladoux A, Frelin C. Differential regulation of cardiac heme oxygenase-1 and vascular endothelial growth factor mRNA expressions by hemin, heavy metals, heat shock and anoxia. FEBS Lett. 1996. 382:229–233.11. Keyse SM, Tyrrell RM. Heme oxygenase is the major 32-kDa stress protein induced in human skin fibroblasts by UVA radiation, hydrogen peroxide, and sodium arsenite. Proc Natl Acad Sci U S A. 1989. 86:99–103.12. Lautier D, Luscher P, Tyrrell RM. Endogenous glutathione levels modulate both constitutive and UVA radiation/hydrogen peroxide inducible expression of the human heme oxygenase gene. Carcinogenesis. 1992. 13:227–232.13. Wagner M, Cadetg P, Ruf R, Mazzucchelli L, Ferrari P, Redaelli CA. Heme oxygenase-1 attenuates ischemia/ reperfusion-induced apoptosis and improves survival in rat renal allografts. Kidney Int. 2003. 63:1564–1573.14. Choi BM, Pae HO, Chung HT. Nitric oxide priming protects nitric oxide-mediated apoptosis via heme oxygenase-1 induction. Free Radic Biol Med. 2003. 34:1136–1145.15. Amon M, Menger MD, Vollmar B. Heme oxygenase and nitric oxide synthasemediate cooling-associated protection against TNF-alpha-induced microcirculatory dysfunction and apoptotic cell death. FASEB J. 2003. 17:175–185.16. Ke B, Shen XD, Zhai Y, Gao F, Busuttil RW, Volk HD, et al. Heme oxygenase 1 mediates the immunomodulatory and antiapoptotic effects of interleukin 13 gene therapy in vivo and in vitro. Hum Gene Ther. 2002. 13:1845–1857.17. Goodman AI, Choudhury M, da Silva JL, Schwartzman ML, Abraham NG. Overexpression of the heme oxygenase gene in renal cell carcinoma. Proc Soc Exp Biol Med. 1997. 214:54–61.18. Maines MD, Abrahamsson PA. Expression of heme oxygenase-1 (HSP32) in human prostate: normal, hyperplastic, and tumor tissue distribution. Urology. 1996. 47:727–733.19. Sahoo SK, Sawa T, Fang J, Tanaka S, Miyamoto Y, Akaike T, et al. Pegylated zinc protoporphyrin: a water-soluble heme oxygenase inhibitor with tumortargeting capacity. Bioconjug Chem. 2002. 13:1031–1038.20. Fram RJ. Cisplatin and platinum analogues: recent advances. Curr Opin Oncol. 1992. 4:1073–1079.21. Jung JH, Kim HR, Kim EJ, Hwang KE, Kim SY, Park JH, et al. The role of heme oxygenase-1 in lung cancer cells. Tuberc Respir Dis. 2006. 60:304–313.22. Wang G, Reed E, Li QQ. Molecular basis of cellular response to cisplatin chemotherapy in non-small cell lung cancer. Oncol Rep. 2004. 12:955–965.23. So HS, Kim HJ, Lee JH, Lee JH, Park SY, Park C, et al. Flunarizine induces Nrf2-mediated transcriptional activation of heme oxygenase-1 in protection of auditory cells from cisplatin. Cell Death Differ. 2006. 13:1763–1775.24. Kim HJ, So HS, Lee JH, Lee JH, Park C, Park SY, et al. Heme oxygenase-1 attenuates the cisplatin-induced apoptosis of auditory cells via down-regulation of reactive oxygen species generation. Free Radic Biol Med. 2006. 40:1810–1819.25. Fang J, Sawa T, Akaike T, Greish K, Maeda H. Enhancement of chemotherapeutic response of tumor cells by a heme oxygenase inhibitor, pegylated zinc protoporphyrin. Int J Cancer. 2004. 109:1–8.26. Eastman A. The formation, isolation and characterization of DNA adducts produced by anticancer platinum complexes. Pharmacol Ther. 1987. 34:155–166.27. Sorenson CM, Eastman A. Mechanism of cis-diamminedichloroplatinum( II)-induced cytotoxicity: role of G2 arrest and DNA double-strand breaks. Cancer Res. 1988. 48:4484–4488.28. Siddik ZH. Cisplatin: mode of cytotoxic action and molecular basis of resistance. Oncogene. 2003. 22:7265–7279.29. Pae HO, Oh GS, Choi BM, Chae SC, Kim YM, Chung KR, et al. Carbon monoxide produced by heme oxygenase-1 suppresses T cell proliferation via inhibition of IL-2 production. J Immunol. 2004. 172:4744–4751.30. Otterbein LE, Bach FH, Alam J, Soares M, Tao Lu H, Wysk M, et al. Carbon monoxide has anti-inflammatory effects involving the mitogen-activated protein kinase pathway. Nat Med. 2000. 6:422–428.31. Berberat PO, Katori M, Kaczmarek E, Anselmo D, Lassman C, Ke B, et al. Heavy chain ferritin acts as an antiapoptotic gene that protects livers from ischemia reperfusion injury. FASEB J. 2003. 17:1724–1726.32. Tsuji MH, Yanagawa T, Iwasa S, Tabuchi K, Onizawa K, Bannai S, et al. Heme oxygenase-1 expression in oral squamous cell carcinoma as involved in lymph node metastasis. Cancer Lett. 1999. 138:53–59.33. Deininger MH, Meyermann R, Trautmann K, Duffner F, Grote EH, Wickboldt J, et al. Heme oxygenase (HO)-1 expressing macrophages/microglial cells accumulate during oligodendroglioma progression. Brain Res. 2000. 882:1–8.34. Torisu-Itakura H, Furue M, Kuwano M, Ono M. Co-expression of thymidine phosphorylase and heme oxygenase-1 in macrophages in human malignant vertical growth melanomas. Jpn J Cancer Res. 2000. 91:906–910.35. Fang J, Sawa T, Akaike T, Akuta T, Sahoo SK, Khaled G, et al. In vivo antitumor activity of pegylated zinc protoporphyrin: targeted inhibition of heme oxygenase in solid tumor. Cancer Res. 2003. 63:3567–3574.36. Fang J, Akaike T, Maeda H. Antiapoptotic role of heme oxygnease (HO) and the potential of HO as a target in anticancer treatment. Apoptosis. 2004. 9:27–35.37. Kweon MH, Adhami VM, Lee JS, Mukhtar H. Constitutive overexpression of Nrf2-dependent heme oxygenase-1 in A549 cells contributes to resistance to apoptosis induced by EGCG. J Biol Chem. 2006. (In press).38. Nowis D, Legat M, Grzela T, Niderla J, Wilczek E, Wilczynski GM, et al. Heme oxygenase-1 protects tumor cells against photodynamic therapy-mediated cytotoxicity. Oncogene. 2006. 25:3365–3374.39. Tanaka S, Akaike T, Fang J, Beppu T, Ogawa M, Tamura F, et al. Antiapoptotic effect of haem oxygenase-1 induced by nitric oxide in experimental solid tumour. Br J Cancer. 2003. 88:902–909.40. Sasaki T, Yoshida K, Kondo H, Ohmori H, Kuniyasu H. Heme oxygenase-1 accelerates protumoral effects of nitric oxide in cancer cells. Virchows Arch. 2005. 446:525–531.41. Barr RK, Bogoyevitch MA. The c-Jun N-terminal protein kinase family of mitogen-activated protein kinases (JNK MAPKs). Int J Biochem Cell Biol. 2001. 33:1047–1063.42. Zanke BW, Boudreau K, Rubie E, Winnett E, Tibbles LA, Zon L, et al. The stress-activated protein kinase pathway mediates cell death following injury induced by cis-platinum, UV irradiation or heat. Curr Biol. 1996. 6:606–613.43. Sanchez-Perez I, Murguia JR, Perona R. Cisplatin induces a persistent activation of JNK that is relatedto cell death. Oncogene. 1998. 16:533–540.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Relationship between Heme Oxygenase-1 Expression and Response to Cisplatin Containing Chemotherapy in Advanced Non-Small Cell Lung Cancer
- Heme Oxygenase-1 via Transcriptional Activation of Nrf2 Attenuates Apoptosis in Lung Cancer Cells
- Heme Oxygenase-1: Its Therapeutic Roles in Inflammatory Diseases
- Effects of Heme Oxygenase-1 Expression in Mycophenolic Acid Induced Apoptosis of Jurkat Cell Lines
- The Role of Heme Oxygenase-1 in Lung Cancer Cells