Immune Netw.
2017 Oct;17(5):352-364. 10.4110/in.2017.17.5.352.
Functional Defects in Type 3 Innate Lymphoid Cells and Classical Monocytes in a Patient with Hyper-IgE Syndrome
- Affiliations
-
- 1Department of Biomedical Sciences, Seoul National University College of Medicine, Seoul 03080, Korea. hykim11@snu.ac.kr
- 2Laboratory of Mucosal Immunology in Department of Biomedical Sciences, Seoul National University College of Medicine, Seoul 03080, Korea.
- 3Department of Internal Medicine, Seoul National University College of Medicine, Seoul 03080, Korea. helenmed@snu.ac.kr
- 4Institute of Allergy and Clinical Immunology, Seoul National University Medical Research Center, Seoul 03080, Korea.
- KMID: 2400640
- DOI: http://doi.org/10.4110/in.2017.17.5.352
Abstract
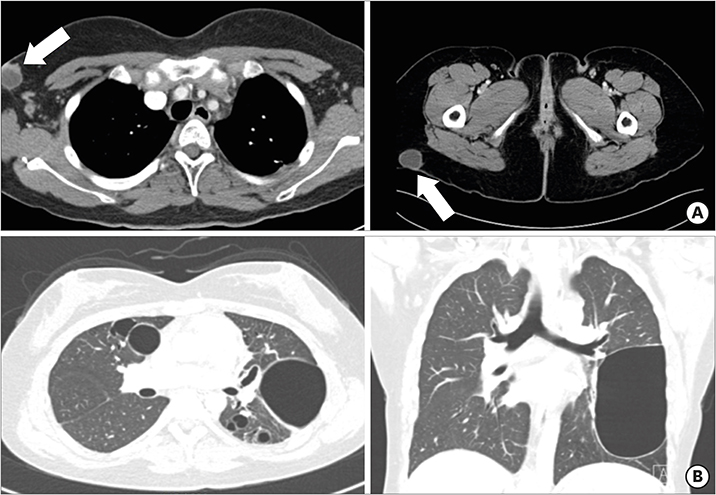
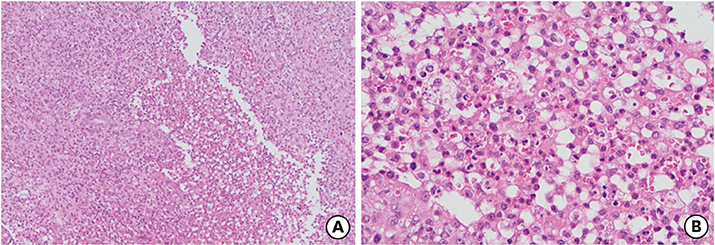
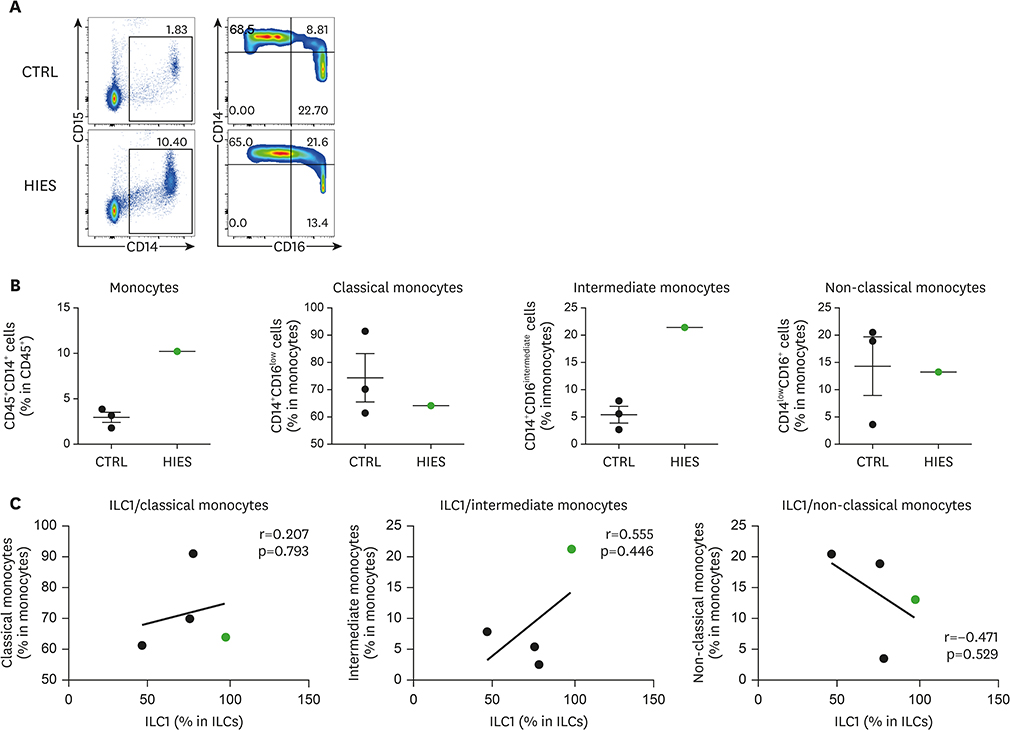
- Hyper-IgE syndrome (HIES) is a very rare primary immune deficiency characterized by elevated serum IgE levels, recurrent bacterial infections, chronic dermatitis, and connective tissue abnormalities. Autosomal dominant (AD) HIES involves a mutation in signal transducer and activator of transcription 3 (STAT3) that leads to an impaired T(H)17 response. STAT3 signaling is also involved in the function of RORγt⺠type 3 innate lymphoid cells (ILC3s) and RORγtâºT(H)17 cells. The aim of this study was to investigate the role of innate immune cells such as innate lymphoid cells (ILCs), granulocytes, and monocytes in a patient with HIES. Peripheral blood mononuclear cells (PBMCs) from a patient with HIES and three age-matched healthy controls were obtained for the analysis of the innate and adaptive immune cells. The frequencies of ILCs in PBMCs were lower in the patient with HIES than in the controls. Moreover, granulocyte-macrophage colony-stimulating factor (GM-CSF) and IL-17A produced by ILC3s in PBMCs were lower in the patient with HIES than the controls. Compared with the controls, classical monocytes (CD14âºCD16(low)), which have a high antimicrobial capability, were also lower in the patient with HIES, while non-classical monocytes (CD14(low)CD16âº) as well as intermediate monocytes (CD14âºCD16(intermediate)) were higher. Taken together, these results indicate that the impaired immune defense against pathogenic microbes in the patient with HIES might be partially explained by functional defects in ILC3s and inflammatory monocytes.
Keyword
MeSH Terms
-
Bacterial Infections
Connective Tissue
Cytokines
Dermatitis
Granulocyte-Macrophage Colony-Stimulating Factor
Granulocytes
Humans
Immunity, Innate
Immunoglobulin E
Interleukin-17
Job Syndrome*
Lymphocytes*
Monocytes*
STAT3 Transcription Factor
Cytokines
Granulocyte-Macrophage Colony-Stimulating Factor
Immunoglobulin E
Interleukin-17
STAT3 Transcription Factor
Figure
Cited by 1 articles
-
Decreased Expression of Sphingosine-1-Phosphate Receptor 1 in the Blood Leukocyte of Rheumatoid Arthritis Patients
Hyun Suk Choi, Kyong Hoon Kim, Seohoon Jin, Jinhyun Kim, Inseol Yoo, Seung Pil Pack, Un-Hwan Ha, Tae Won Park, Soo An Choi, Soon Hong Yuk, Seong Wook Kang, Yong Woo Jung
Immune Netw. 2018;18(5):. doi: 10.4110/in.2018.18.e39.
Reference
-
1. Heimall J, Freeman A, Holland SM. Pathogenesis of hyper IgE syndrome. Clin Rev Allergy Immunol. 2010; 38:32–38.
Article2. Holland SM, DeLeo FR, Elloumi HZ, Hsu AP, Uzel G, Brodsky N, Freeman AF, Demidowich A, Davis J, Turner ML, et al. STAT3 mutations in the hyper-IgE syndrome. N Engl J Med. 2007; 357:1608–1619.3. Minegishi Y, Saito M, Morio T, Watanabe K, Agematsu K, Tsuchiya S, Takada H, Hara T, Kawamura N, Ariga T, et al. Human tyrosine kinase 2 deficiency reveals its requisite roles in multiple cytokine signals involved in innate and acquired immunity. Immunity. 2006; 25:745–755.
Article4. Zhang Q, Davis JC, Lamborn IT, Freeman AF, Jing H, Favreau AJ, Matthews HF, Davis J, Turner ML, Uzel G, et al. Combined immunodeficiency associated with DOCK8 mutations. N Engl J Med. 2009; 361:2046–2055.
Article5. Saito M, Nagasawa M, Takada H, Hara T, Tsuchiya S, Agematsu K, Yamada M, Kawamura N, Ariga T, Tsuge I, et al. Defective IL-10 signaling in hyper-IgE syndrome results in impaired generation of tolerogenic dendritic cells and induced regulatory T cells. J Exp Med. 2011; 208:235–249.
Article6. Lin JX, Migone TS, Tsang M, Friedmann M, Weatherbee JA, Zhou L, Yamauchi A, Bloom ET, Mietz J, John S, et al. The role of shared receptor motifs and common Stat proteins in the generation of cytokine pleiotropy and redundancy by IL-2, IL-4, IL-7, IL-13, and IL-15. Immunity. 1995; 2:331–339.
Article7. Wei L, Laurence A, Elias KM, O'Shea JJ. IL-21 is produced by Th17 cells and drives IL-17 production in a STAT3-dependent manner. J Biol Chem. 2007; 282:34605–34610.
Article8. Hankey PA. Regulation of hematopoietic cell development and function by Stat3. Front Biosci (Landmark Ed). 2009; 14:5273–5290.
Article9. de Jong PR, Schadenberg AW, van den Broek T, Beekman JM, van Wijk F, Coffer PJ, Prakken BJ, Jansen NJ. STAT3 regulates monocyte TNF-alpha production in systemic inflammation caused by cardiac surgery with cardiopulmonary bypass. PLoS One. 2012; 7(5):–. e35070.
Article10. Milner JD, Brenchley JM, Laurence A, Freeman AF, Hill BJ, Elias KM, Kanno Y, Spalding C, Elloumi HZ, Paulson ML, et al. Impaired T(H)17 cell differentiation in subjects with autosomal dominant hyper-IgE syndrome. Nature. 2008; 452:773–776.
Article11. Ma CS, Chew GY, Simpson N, Priyadarshi A, Wong M, Grimbacher B, Fulcher DA, Tangye SG, Cook MC. Deficiency of Th17 cells in hyper IgE syndrome due to mutations in STAT3. J Exp Med. 2008; 205:1551–1557.
Article12. Laan M, Cui ZH, Hoshino H, Lötvall J, Sjöstrand M, Gruenert DC, Skoogh BE, Lindén A. Neutrophil recruitment by human IL-17 via C-X-C chemokine release in the airways. J Immunol. 1999; 162:2347–2352.13. Minegishi Y, Saito M, Nagasawa M, Takada H, Hara T, Tsuchiya S, Agematsu K, Yamada M, Kawamura N, Ariga T, et al. Molecular explanation for the contradiction between systemic Th17 defect and localized bacterial infection in hyper-IgE syndrome. J Exp Med. 2009; 206:1291–1301.
Article14. Hill HR, Ochs HD, Quie PG, Clark RA, Pabst HF, Klebanoff SJ, Wedgwood RJ. Defect in neutrophil granulocyte chemotaxis in Job’s syndrome of recurrent “cold” staphylococcal abscesses. Lancet. 1974; 2:617–619.
Article15. Buckley RH, Becker WG. Abnormalities in the regulation of human IgE synthesis. Immunol Rev. 1978; 41:288–314.
Article16. Giacomelli M, Tamassia N, Moratto D, Bertolini P, Ricci G, Bertulli C, Plebani A, Cassatella M, Bazzoni F, Badolato R. SH2-domain mutations in STAT3 in hyper-IgE syndrome patients result in impairment of IL-10 function. Eur J Immunol. 2011; 41:3075–3084.
Article17. Yeganeh M, Henneke P, Rezaei N, Ehl S, Thiel D, Matamoros N, Pietrogrande C, Espanol T, Litzman J, Franco JL, et al. Toll-like receptor stimulation induces higher TNF-alpha secretion in peripheral blood mononuclear cells from patients with hyper IgE syndrome. Int Arch Allergy Immunol. 2008; 146:190–194.
Article18. Panopoulos AD, Zhang L, Snow JW, Jones DM, Smith AM, El Kasmi KC, Liu F, Goldsmith MA, Link DC, Murray PJ, et al. STAT3 governs distinct pathways in emergency granulopoiesis and mature neutrophils. Blood. 2006; 108:3682–3690.
Article19. Moniuszko M, Bodzenta-Lukaszyk A, Kowal K, Lenczewska D, Dabrowska M. Enhanced frequencies of CD14++CD16+, but not CD14+CD16+, peripheral blood monocytes in severe asthmatic patients. Clin Immunol. 2009; 130:338–346.
Article20. Wong KL, Yeap WH, Tai JJ, Ong SM, Dang TM, Wong SC. The three human monocyte subsets: implications for health and disease. Immunol Res. 2012; 53:41–57.
Article21. Artis D, Spits H. The biology of innate lymphoid cells. Nature. 2015; 517:293–301.
Article22. Chang YJ, Kim HY, Albacker LA, Baumgarth N, McKenzie AN, Smith DE, Dekruyff RH, Umetsu DT. Innate lymphoid cells mediate influenza-induced airway hyper-reactivity independently of adaptive immunity. Nat Immunol. 2011; 12:631–638.
Article23. Kim HY, Lee HJ, Chang YJ, Pichavant M, Shore SA, Fitzgerald KA, Iwakura Y, Israel E, Bolger K, Faul J, et al. Interleukin-17-producing innate lymphoid cells and the NLRP3 inflammasome facilitate obesity-associated airway hyperreactivity. Nat Med. 2014; 20:54–61.
Article24. Wang WB, Levy DE, Lee CK. STAT3 negatively regulates type I IFN-mediated antiviral response. J Immunol. 2011; 187:2578–2585.
Article25. Gutierrez-Hincapié S, Muskus-López CE, Montoya CJ, Trujillo-Vargas CM. Quantitative defects in invariant NKT cells and TLR responses in patients with hyper-IgE syndrome. Allergol Immunopathol (Madr). 2015; 43:553–561.26. Xiong H, Keith JW, Samilo DW, Carter RA, Leiner IM, Pamer EG. Innate Lymphocyte/Ly6C(hi) monocyte crosstalk promotes klebsiella pneumoniae clearance. Cell. 2016; 165:679–689.
Article27. Minegishi Y, Saito M, Tsuchiya S, Tsuge I, Takada H, Hara T, Kawamura N, Ariga T, Pasic S, Stojkovic O, et al. Dominant-negative mutations in the DNA-binding domain of STAT3 cause hyper-IgE syndrome. Nature. 2007; 448:1058–1062.
Article28. Taga T, Hibi M, Hirata Y, Yamasaki K, Yasukawa K, Matsuda T, Hirano T, Kishimoto T. Interleukin-6 triggers the association of its receptor with a possible signal transducer, gp130. Cell. 1989; 58:573–581.
Article29. O'Shea JJ, Plenge R. JAK and STAT signaling molecules in immunoregulation and immune-mediated disease. Immunity. 2012; 36:542–550.30. Del Prete G, Tiri A, Maggi E, De Carli M, Macchia D, Parronchi P, Rossi ME, Pietrogrande MC, Ricci M, Romagnani S. Defective in vitro production of gamma-interferon and tumor necrosis factor-alpha by circulating T cells from patients with the hyper-immunoglobulin E syndrome. J Clin Invest. 1989; 84:1830–1835.
Article31. Guo X, Qiu J, Tu T, Yang X, Deng L, Anders RA, Zhou L, Fu YX. Induction of innate lymphoid cell-derived interleukin-22 by the transcription factor STAT3 mediates protection against intestinal infection. Immunity. 2014; 40:25–39.
Article32. Ziegler-Heitbrock L, Hofer TP. Toward a refined definition of monocyte subsets. Front Immunol. 2013; 4:23.
Article33. Cooper DL, Martin SG, Robinson JI, Mackie SL, Charles CJ, Nam J, Isaacs JD, Emery P, Morgan AW. YEAR Consortium. FcγRIIIa expression on monocytes in rheumatoid arthritis: role in immune-complex stimulated TNF production and non-response to methotrexate therapy. PLoS One. 2012; 7:e28918.34. Schauer D, Starlinger P, Reiter C, Jahn N, Zajc P, Buchberger E, Bachleitner-Hofmann T, Bergmann M, Stift A, Gruenberger T, et al. Intermediate monocytes but not TIE2-expressing monocytes are a sensitive diagnostic indicator for colorectal cancer. PLoS One. 2012; 7:e44450.
Article35. Skrzeczyńska-Moncznik J, Bzowska M, Loseke S, Grage-Griebenow E, Zembala M, Pryjma J. Peripheral blood CD14high CD16+ monocytes are main producers of IL-10. Scand J Immunol. 2008; 67:152–159.36. Tsukamoto M, Seta N, Yoshimoto K, Suzuki K, Yamaoka K, Takeuchi T. CD14(bright)CD16+ intermediate monocytes are induced by interleukin-10 and positively correlate with disease activity in rheumatoid arthritis. Arthritis Res Ther. 2017; 19:28.
Article37. Grimbacher B, Holland SM, Puck JM. Hyper-IgE syndromes. Immunol Rev. 2005; 203:244–250.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Role of Type 2 Innate Lymphoid Cells in Allergic Diseases
- A Case of Hyper-IgE Syndrome Presenting as a Vesiculopustular Eruption in an Infant
- Guillain-Barré syndrome associated with hyper-IgE-emia
- Innate Lymphoid Cells and Infection
- Regulation of IgE-Mediated Food Allergy by IL-9 Producing Mucosal Mast Cells and Type 2 Innate Lymphoid Cells