Cancer Res Treat.
2004 Feb;36(1):85-90.
Influence of Estrogen and Polyamines on Mifepristone-induced Apoptosis in Prostate Cancer Cells
- Affiliations
-
- 1Department of Biology, Pusan National University, Busan, Korea.
- 2Bioscience Division, Los Alamos National Laboratory, Los Alamos, NM, USA.
Abstract
- PURPOSE
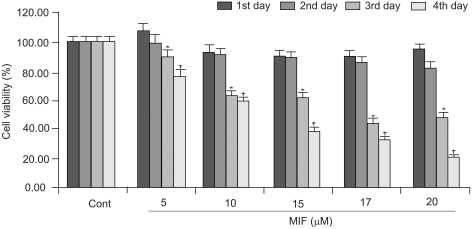
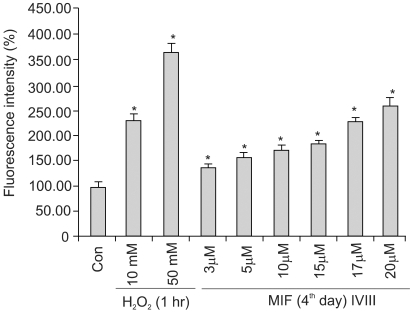
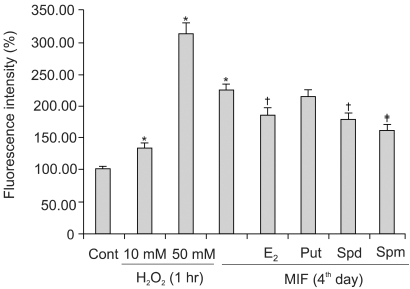
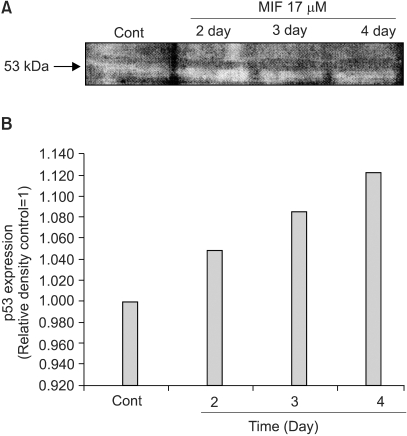
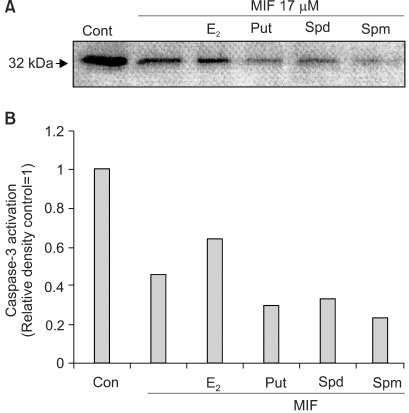
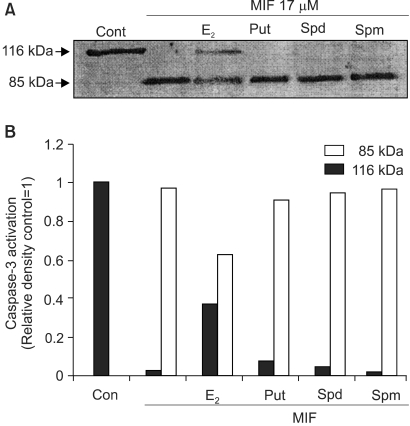
Although androgens are the main steroids controlling the growth of prostate glands, estrogens are also important in the regulation of its growth. Prostate cancer cells, like other cancer cells, maintain high levels of polyamines. In LNCaP cells, apoptosis is induced by mifepristone. During the process of cell death, the regulation of ROS production, caspase-3 activation and poly (ADP-ribose) polymerase cleavage were investigated in the presence of estrogen and polyamines to identify their possible roles. MATERIALS AND METHODS: The cell growth was assessed using the MTT assay, and the intracellular ROS production by the DCFH-DA assay. The p53 protein expression, activation of caspase-3 and PARP cleavage were checked by Western blotting, with specific antibodies to each. RESULTS: The growth and viability of the cells were significantly inhibited, in a dose- and time-dependent manners, by mifepristone (MIF) treatment. The production of ROS were dependent on the MIF dosage. The activation of caspase-3 and cleavage of PARP also increased with the duration of MIF treatment. The expression of p53 protein also increased with increases in the MIF incubation time. E2 severely inhibited the ROS production, caspase-3 activation and PARP cleavage. However, polyamines only inhibited the ROS production, without influencing the caspase-3 activation or PARP cleavage. CONCLUSION: In LNCaP cells, MIF induces apoptosis through ROS production. The expression of p53 protein, caspase-3 activation and PARP cleavage accompanied the process of apoptosis. The apoptotic processes were inhibited by E2, but polyamines only inhibited the ROS production, implying the multifunctional role of E2, in addition to its role as a free radical scavenger.
Keyword
MeSH Terms
Figure
Reference
-
1. El Etreby MF, Liang Y, Lewis RW. Induction of apoptosis by mifepristone and tamoxifen in human LNCaP prostate cancer cells in culture. Prostate. 2000; 43:31–42. PMID: 10725863.
Article2. Eid MA, Lewis RW, Kumar MV. Mifepristone pretreatment overcomes resistance of prostate cancer cells to tumor necrosis factor α-related apoptosis-induced ligand. Mol Cancer Ther. 2002; 1:831–840. PMID: 12492116.3. Kang CM, Suh Y, Jang IS, Park SC. Thymidine-dependent attenuation of the mitochondrial apoptotic pathway in adenosine-induced apoptosis of HL-60 cells. J Cancer Res Clin Oncol. 2001; 127(9):570–576. PMID: 11570579.
Article4. Slee EA, Harte MT, Kluck RM, Wolf BB, Casiano CA, Newmeyer DD, Wang HG, Reed JC, Nicholson DW, Alnemri ES, Green DR, Martin SJ. Ordering the cytochrome c-initiated caspase cascade: hierarchical activation of caspases-2, -3, -6, -7, -8, and -10 in a caspase-9-dependent manner. J Cell Biol. 1999; 144:281–292. PMID: 9922454.
Article5. Nakamura T, Sakamoto K. Reactive oxygen species up-regulates cyclooxygenase-2, p53, and Bax mRNA expression in bovine luteal cells. Biochem Biophys Res Commun. 2000; 284:203–210. PMID: 11374891.
Article6. Wang S, Leonard SS, Ye J, Ding M, Shi X. The role of hydroxyl radical as a messenger in Cr(VI)-induced p53 activation. Am J Physiol Cell Physiol. 2000; 279:C868–C875. PMID: 10942736.7. Winter RN, Kramer A, Borkowski A, Kyprianou N. Loss of caspase-1 and caspase-3 protein expression in human prostate cancer. Cancer Res. 2001; 61:1227–1232. PMID: 11221855.8. Marcelli M, Cunningham GR, Walkup M, He Z, Sturgis L, Kagan C, Mannucci R, Nicoletti I, Teng B, Denner L. Signaling pathway activated during apoptosis of the prostate cancer cell line LNCaP: overexpression of caspase-7 as a new gene therapy strategy for prostate cancer. Cancer Res. 1999; 59:382–390. PMID: 9927051.9. Schor N, Tyurina YY, Tyurin VA, Kagan VE. Differential membrane antioxidant effects of immediate and long-term estradiol treatment of MCF-7 breast cancer cells. Biochem Biophys Res Commun. 1999; 5:410–415. PMID: 10403783.
Article10. Pegg AE. Polyamine metabolism and its importance in neoplastic growth and a target for chemotherapy. Cancer Res. 1988; 48:759–774. PMID: 3123052.11. Kim JY, Kim KY, Lee KH, Hong KH, Kim BG. Effects of polyamines on TNF- or tamoxifen-induced apoptosis in human breast cancer cells. Cancer Res Treat. 2001; 33:385–391.12. Ha HC, Yager JD, Woster PA, Casero RA Jr. Structural specificity of polyamines and polyamine analogues in the protection of DNA from strand breaks induced by reactive oxygen species. Biochem Biophys Res Commun. 1998; 244:298–303. PMID: 9514920.
Article13. Zagaja GP, Shrivastav M, Fleig MJ, Marton LJ, Rinker-Schaeffer CW, Dolan ME. Effects of polyamine analogues on prostatic adenocarcinoma cells in vitro and in vivo. Cancer Chemother Pharmacol. 1998; 41:505–512. PMID: 9554596.
Article14. Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976; 72:248–254. PMID: 942051.
Article15. Lin MF, Kawachi MH, Stallcup MR, Grunberg SM, Lin FF. Growth inhibition of androgen-insensitive human prostate carcinoma cells by a 19-norsteroid derivative agent, mifepristone. Prostate. 1995; 26(4):194–204. PMID: 7716084.
Article16. Polyake K, Xia Y, Zweier JL, Kinzler KW, Vogelstein B. A model for p53-induced apoptosis. Nature. 1997; 389:300–305. PMID: 9305847.
Article17. Li PF, Dietz R, von Harsdorf R. p53 regulates mitochondrial membrane potential through reactive oxygen species and induces cytochrome c-independent apoptosis blocked by Bcl-2. EMBO J. 1999; 18:6027–6036. PMID: 10545114.
Article18. Grande M, Carlstrom K, Stege R, Pousette A, Faxen M. Estrogens increase the endothelial nitric oxide synthase (ecNOS) mRNA level in LNCaP human prostate carcinoma cells. Prostate. 2000; 45:232–237. PMID: 11074525.
Article19. Risbridger GP, Bianco JJ, Ellem SJ, Mcpherson SJ. Oestrogens and prostate cancer. Endocr Relat Cancer. 2003; 10:187–191. PMID: 12790781.
Article20. Ayres S, Abplanalp W, Liu JH, Subbiah MT. Mechanisms involved in the protective effect of estradiol-17beta on lipid peroxidation and DNA damage. Am J Physiol. 1998; 274(6 Pt 1):E1002–E1008. PMID: 9611149.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of Polyamines on TNFalpha- or Tamoxifen-induced Apoptosis in Human Breast Cancer Cells
- Alfa - difluoromethylornithine Reduced Protein Phosphorylation in MCF-7 Human Breast Cancer Cells
- Comparative Studies on the Polyarnine Involvement in MCF - 7 and MDA - MB - 231 Breast Cancer Cell Proliferation
- Association of dexamethasone-induced apoptosis of G|1-arrest of human leukemic CEM cells with polyamine deficit
- Polyamines Regulate Growth Factor-Induced Protein Phosphorylation in MCF-7 Human Breast Cancer Cells