Ann Dermatol.
2019 Dec;31(6):631-639. 10.5021/ad.2019.31.6.631.
Selective Inhibition of β-Catenin/Co-Activator Cyclic AMP Response Element-Binding Protein-Dependent Signaling Prevents the Emergence of Hapten-Induced Atopic Dermatitis-Like Dermatitis
- Affiliations
-
- 1Department of Dermatology, Faculty of Medicine, Oita University, Oita, Japan. hatano@oita-u.ac.jp
- 2Translational Chemical Biology, Faculty of Medicine, Oita University, Oita, Japan.
- 3Department of Clinical Pharmacology and Therapeutics, Faculty of Medicine, Oita University, Oita, Japan.
- 4Department of Infectious Disease Control, Faculty of Medicine, Oita University, Oita, Japan.
- KMID: 2461985
- DOI: http://doi.org/10.5021/ad.2019.31.6.631
Abstract
- BACKGROUND
The canonical Wnt/β-catenin signaling pathway is a fundamental regulatory system involved in various biological events. ICG-001 selectively blocks the interaction of β-catenin with its transcriptional co-activator cyclic AMP response element-binding protein (CBP). Recent studies have provided convincing evidence of the inhibitory effects of ICG-001 on Wnt-driven disease models, such as organ fibrosis, cancer, acute lymphoblastic leukemia, and asthma. However, the effects of ICG-001 in atopic dermatitis (AD) have not been investigated.
OBJECTIVE
To investigate whether β-catenin/CBP-dependent signaling was contributed in the pathogenesis of AD and ICG-001 could be a therapeutic agent for AD.
METHODS
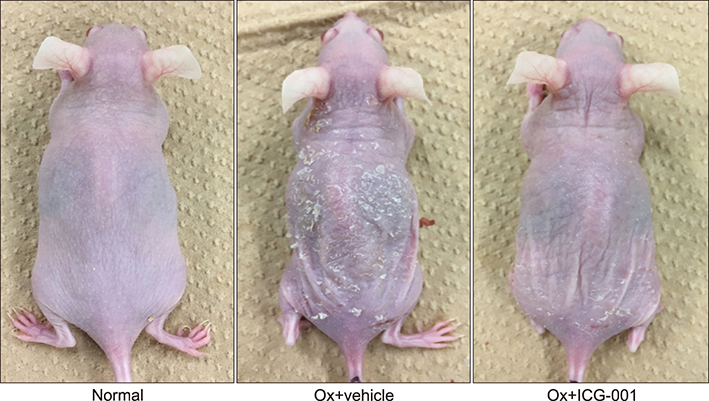
We examined the effects of ICG-001 in an AD-like murine model generated by repeated topical application of the hapten, oxazolone (Ox). ICG-001 or vehicle alone was injected intraperitoneally every day during the development of AD-like dermatitis arising from once-daily Ox treatment.
RESULTS
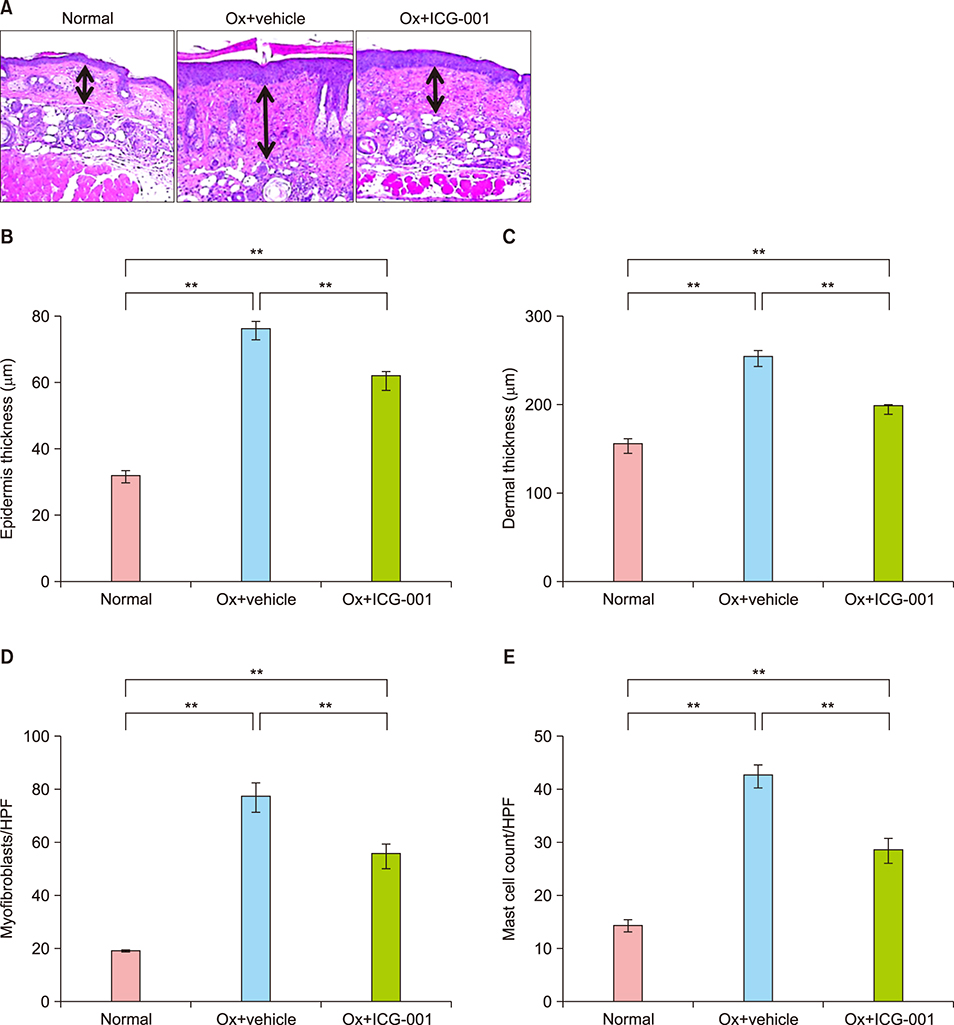
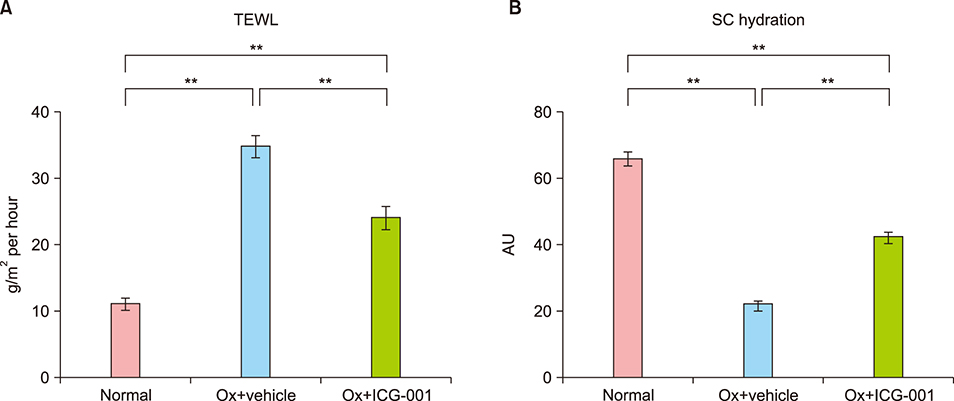
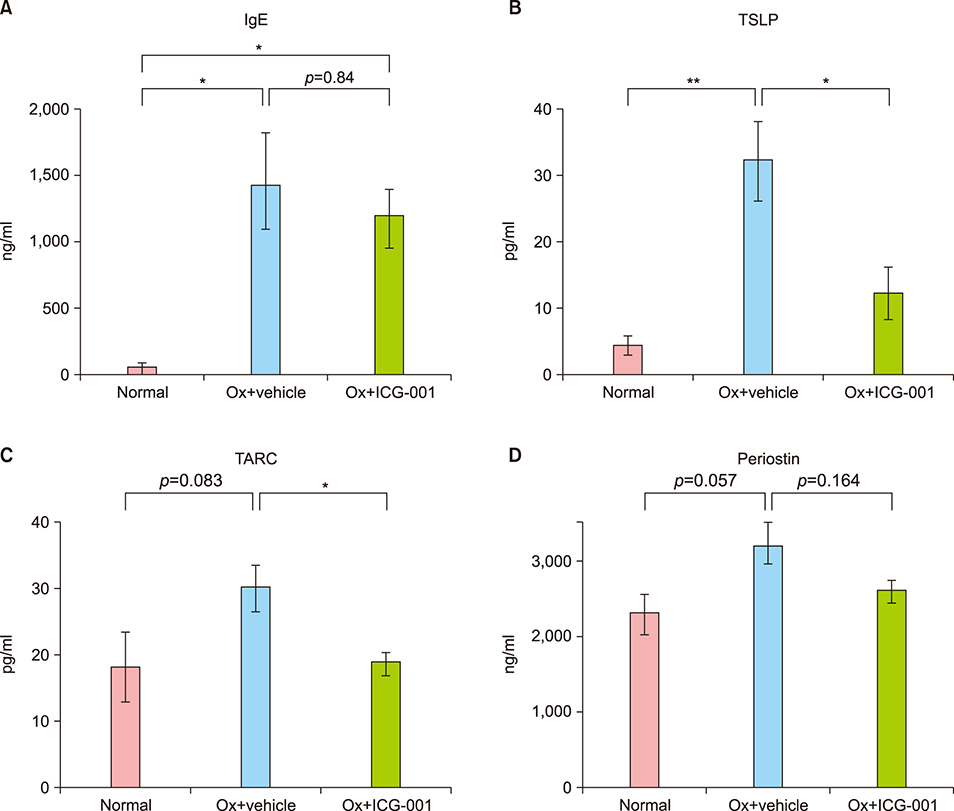
Ox-induced AD-like dermatitis characterized by increases in transepidermal water loss, epidermal thickness, dermal thickness accompanied by increased myofibroblast and mast cell counts, and serum levels of thymic stromal lymphopoietin and thymus and activation-regulated chemokine, and decreases in stratum corneum hydration, were virtually normalized by the treatment with ICG-001. Elevated serum levels of periostin tended to be downregulated, without statistical significance.
CONCLUSION
These results suggest that β-catenin/CBP-dependent signaling might be involved in the pathogenesis of AD and could be a therapeutic target.
Keyword
MeSH Terms
Figure
Reference
-
1. Kabashima K. New concept of the pathogenesis of atopic dermatitis: interplay among the barrier, allergy, and pruritus as a trinity. J Dermatol Sci. 2013; 70:3–11.
Article2. Elias PM, Hatano Y, Williams ML. Basis for the barrier abnormality in atopic dermatitis: outside-inside-outside pathogenic mechanisms. J Allergy Clin Immunol. 2008; 121:1337–1343.
Article3. Leung DY. Atopic dermatitis: the skin as a window into the pathogenesis of chronic allergic diseases. J Allergy Clin Immunol. 1995; 96:302–318. quiz 319.
Article4. Yamaguchi Y. Periostin in skin tissue and skin-related diseases. Allergol Int. 2014; 63:161–170.5. Blauvelt A, de Bruin-Weller M, Gooderham M, Cather JC, Weisman J, Pariser D, et al. Long-term management of moderate-to-severe atopic dermatitis with dupilumab and concomitant topical corticosteroids (LIBERTY AD CHRONOS): a 1-year, randomised, double-blinded, placebo-controlled, phase 3 trial. Lancet. 2017; 389:2287–2303.
Article6. Takahashi-Yanaga F, Kahn M. Targeting Wnt signaling: can we safely eradicate cancer stem cells? Clin Cancer Res. 2010; 16:3153–3162.
Article7. Clevers H, Nusse R. Wnt/β-catenin signaling and disease. Cell. 2012; 149:1192–1205.
Article8. Emami KH, Nguyen C, Ma H, Kim DH, Jeong KW, Eguchi M, et al. A small molecule inhibitor of beta-catenin/CREB-binding protein transcription [corrected]. Proc Natl Acad Sci U S A. 2004; 101:12682–12687.9. Beyer C, Reichert H, Akan H, Mallano T, Schramm A, Dees C, et al. Blockade of canonical Wnt signalling ameliorates experimental dermal fibrosis. Ann Rheum Dis. 2013; 72:1255–1258.
Article10. Gang EJ, Hsieh YT, Pham J, Zhao Y, Nguyen C, Huantes S, et al. Small-molecule inhibition of CBP/catenin interactions eliminates drug-resistant clones in acute lymphoblastic leukemia. Oncogene. 2014; 33:2169–2178.
Article11. Koopmans T, Crutzen S, Menzen MH, Halayko AJ, Hackett TL, Knight DA, et al. Selective targeting of CREB-binding protein/β-catenin inhibits growth of and extracellular matrix remodelling by airway smooth muscle. Br J Pharmacol. 2016; 173:3327–3341.
Article12. Hatano Y, Man MQ, Uchida Y, Crumrine D, Mauro TM, Feingold KR, et al. Murine atopic dermatitis responds to peroxisome proliferator-activated receptors alpha and beta/delta (but not gamma) and liver X receptor activators. J Allergy Clin Immunol. 2010; 125:160–169.e1-5.13. Man MQ, Hatano Y, Lee SH, Man M, Chang S, Feingold KR, et al. Characterization of a hapten-induced, murine model with multiple features of atopic dermatitis: structural, immunologic, and biochemical changes following single versus multiple oxazolone challenges. J Invest Dermatol. 2008; 128:79–86.
Article14. Gaddis M, Gerrard D, Frietze S, Farnham PJ. Altering cancer transcriptomes using epigenomic inhibitors. Epigenetics Chromatin. 2015; 8:9.
Article15. Hatano Y, Adachi Y, Elias PM, Crumrine D, Sakai T, Kurahashi R, et al. The Th2 cytokine, interleukin-4, abrogates the cohesion of normal stratum corneum in mice: implications for pathogenesis of atopic dermatitis. Exp Dermatol. 2013; 22:30–35.
Article16. Zhang W, Sakai T, Hatano Y, Fujiwara S. Betamethasone butyrate propionate inhibits the induction of thymic stromal lymphopoietin in cultured normal human keratinocytes. Ann Dermatol. 2016; 28:772–775.
Article17. Akcora BÖ, Storm G, Bansal R. Inhibition of canonical WNT signaling pathway by β-catenin/CBP inhibitor ICG-001 ameliorates liver fibrosis in vivo through suppression of stromal CXCL12. Biochim Biophys Acta Mol Basis Dis. 2018; 1864:804–818.
Article18. Kinoshita H, Takai T, Le TA, Kamijo S, Wang XL, Ushio H, et al. Cytokine milieu modulates release of thymic stromal lymphopoietin from human keratinocytes stimulated with double-stranded RNA. J Allergy Clin Immunol. 2009; 123:179–186.
Article19. Gattinoni L, Ji Y, Restifo NP. Wnt/beta-catenin signaling in T-cell immunity and cancer immunotherapy. Clin Cancer Res. 2010; 16:4695–4701.
Article20. Krishnamurthy N, Kurzrock R. Targeting the Wnt/beta-catenin pathway in cancer: update on effectors and inhibitors. Cancer Treat Rev. 2018; 62:50–60.
Article21. Nishikawa K, Osawa Y, Kimura K. Wnt/β-Catenin Signaling as a potential target for the treatment of liver cirrhosis using antifibrotic drugs. Int J Mol Sci. 2018; 19:E3103.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- β-Catenin Regulates the Expression of cAMP Response Element-Binding Protein 1 in Squamous Cell Carcinoma Cells
- cAMP signaling increases histone deacetylase 8 expression via the Epac2–Rap1A–Akt pathway in H1299 lung cancer cells
- Effects of dopamine and haloperidol on morphine-induced CREB and AP-1 DNA binding activities in differentiated SH-SY5Y human neuroblastoma cells
- Measurement of Atopic Dermatitis Disability
- Four Cases of Atopic Dermatitis with Topical Tacrolimus Therapy