J Gynecol Oncol.
2014 Jan;25(1):36-42. 10.3802/jgo.2014.25.1.36.
Correlation between the overexpression of epidermal growth factor receptor and mesenchymal makers in endometrial carcinoma
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Shanghai Sixth People's Hospital, Shanghai Jiaotong University, Shanghai, China. teng_yc@126.com
- KMID: 1811186
- DOI: http://doi.org/10.3802/jgo.2014.25.1.36
Abstract
OBJECTIVE
The objective of this study was to evaluate the effect of overexpression of epidermal growth factor receptor (EGFR) on the expression of epithelial cell markers (E-cadherin and alpha-catenin) and mesenchymal cell markers (N-cadherin and vimentin) in endometrial carcinoma.
METHODS
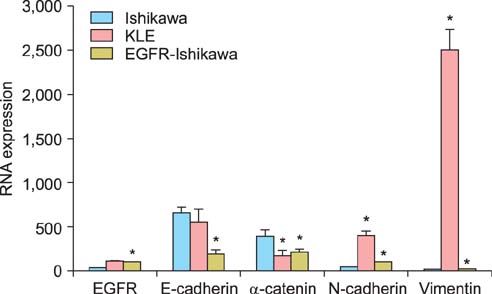
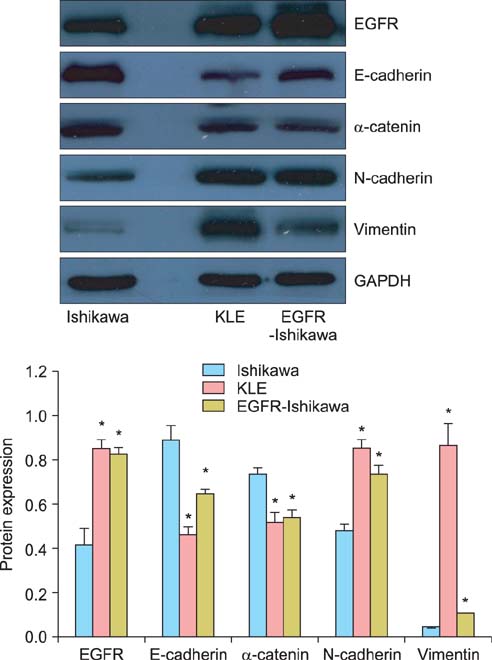
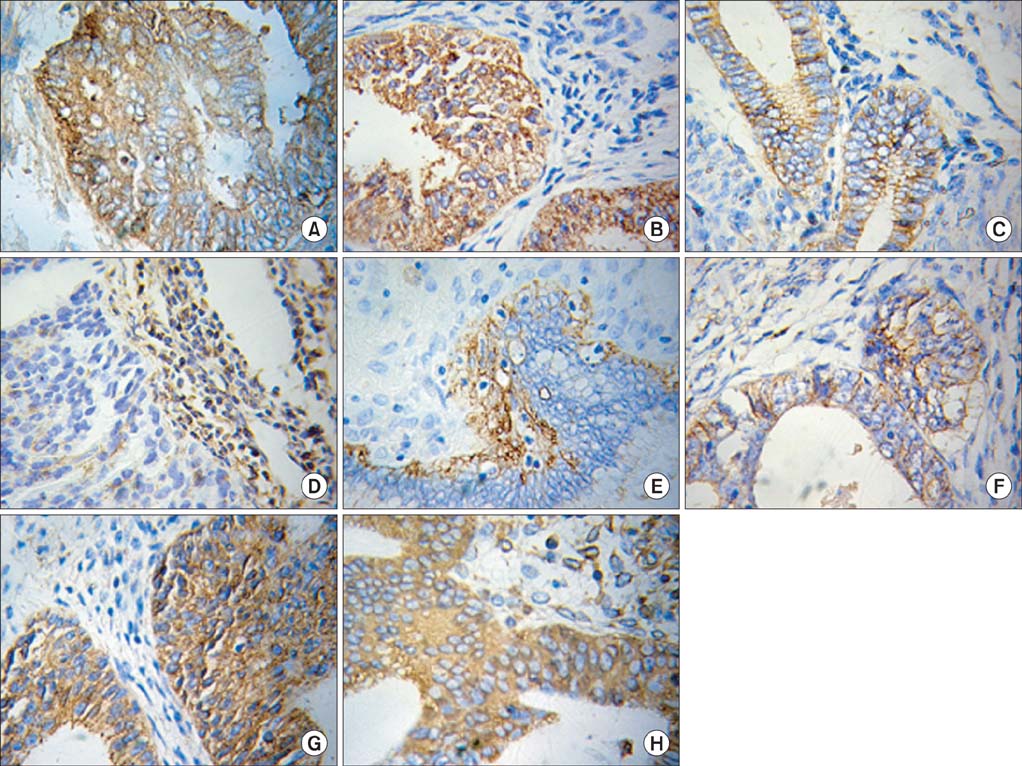
The expression of all 4 markers was evaluated in EGFR overexpressing Ishikawa cells, control Ishikawa cells, and KLE cells using reverse transcription polymerase chain reaction (RT-PCR) and Western blotting. The expression of these 4 markers was also determined in cancerous tissues of patients with endometrial carcinoma using immunohistochemical staining.
RESULTS
Ishikawa cells transfected with EGFR showed decreased expression of E-cadherin and alpha-catenin and increased expression of N-cadherin and vimentin compared with control Ishikawa cells (p<0.01 for all). The expression of N-cadherin and vimentin was higher and the expression of E-cadherin and alpha-catenin was lower in stage II-III than stage I and in grade II-III than grade I endometrial carcinoma tissue (p<0.01 for all).
CONCLUSION
Decreased expression of epithelial markers (E-cadherin and alpha-catenin) and increased expression of mesenchymal markers (N-cadherin and vimentin) were observed in human endometrial carcinoma tissue. These findings correlate with high EGFR expression in cultured endometrial carcinoma cells.
Keyword
MeSH Terms
-
alpha Catenin
Blotting, Western
Cadherins
Endometrial Neoplasms*
Epidermal Growth Factor*
Epithelial Cells
Epithelial-Mesenchymal Transition
Female
Humans
Polymerase Chain Reaction
Receptor, Epidermal Growth Factor*
Reverse Transcription
Vimentin
Cadherins
Epidermal Growth Factor
Receptor, Epidermal Growth Factor
Vimentin
alpha Catenin
Figure
Reference
-
1. Abal M, Llaurado M, Doll A, Monge M, Colas E, Gonzalez M, et al. Molecular determinants of invasion in endometrial cancer. Clin Transl Oncol. 2007; 9:272–277.2. Tsukamoto H, Shibata K, Kajiyama H, Terauchi M, Nawa A, Kikkawa F. Irradiation-induced epithelial-mesenchymal transition (EMT) related to invasive potential in endometrial carcinoma cells. Gynecol Oncol. 2007; 107:500–504.3. Yang S, Thiel KW, De Geest K, Leslie KK. Endometrial cancer: reviving progesterone therapy in the molecular age. Discov Med. 2011; 12:205–212.4. Kang Y, Massague J. Epithelial-mesenchymal transitions: twist in development and metastasis. Cell. 2004; 118:277–279.5. Garber K. Epithelial-to-mesenchymal transition is important to metastasis, but questions remain. J Natl Cancer Inst. 2008; 100:232–239.6. Boyer B, Valles AM, Edme N. Induction and regulation of epithelial-mesenchymal transitions. Biochem Pharmacol. 2000; 60:1091–1099.7. Savagner P. The epithelial-mesenchymal transition (EMT) phenomenon. Ann Oncol. 2010; 21:Suppl 7. vii89–vii92.8. Singh A, Settleman J. EMT, cancer stem cells and drug resistance: an emerging axis of evil in the war on cancer. Oncogene. 2010; 29:4741–4751.9. Ouyang G, Wang Z, Fang X, Liu J, Yang CJ. Molecular signaling of the epithelial to mesenchymal transition in generating and maintaining cancer stem cells. Cell Mol Life Sci. 2010; 67:2605–2618.10. Jing Y, Han Z, Zhang S, Liu Y, Wei L. Epithelial-Mesenchymal Transition in tumor microenvironment. Cell Biosci. 2011; 1:29.11. Saitoh M, Shirakihara T, Miyazono K. Regulation of the stability of cell surface E-cadherin by the proteasome. Biochem Biophys Res Commun. 2009; 381:560–565.12. Cavallaro U, Schaffhauser B, Christofori G. Cadherins and the tumour progression: is it all in a switch? Cancer Lett. 2002; 176:123–128.13. Scholten AN, Aliredjo R, Creutzberg CL, Smit VT. Combined E-cadherin, alpha-catenin, and beta-catenin expression is a favorable prognostic factor in endometrial carcinoma. Int J Gynecol Cancer. 2006; 16:1379–1385.14. Bansal N, Yendluri V, Wenham RM. The molecular biology of endometrial cancers and the implications for pathogenesis, classification, and targeted therapies. Cancer Control. 2009; 16:8–13.15. Nicholson RI, Staka C, Boyns F, Hutcheson IR, Gee JM. Growth factor-driven mechanisms associated with resistance to estrogen deprivation in breast cancer: new opportunities for therapy. Endocr Relat Cancer. 2004; 11:623–641.16. Ai Z, Wang J, Wang Y, Lu L, Tong J, Teng Y. Overexpressed epidermal growth factor receptor (EGFR)-induced progestin insensitivity in human endometrial carcinoma cells by the EGFR/mitogen-activated protein kinase signaling pathway. Cancer. 2010; 116:3603–3613.17. Fromowitz FB, Viola MV, Chao S, Oravez S, Mishriki Y, Finkel G, et al. ras p21 expression in the progression of breast cancer. Hum Pathol. 1987; 18:1268–1275.18. Cano A, Perez-Moreno MA, Rodrigo I, Locascio A, Blanco MJ, del Barrio MG, et al. The transcription factor snail controls epithelial-mesenchymal transitions by repressing E-cadherin expression. Nat Cell Biol. 2000; 2:76–83.19. Oft M, Heider KH, Beug H. TGFbeta signaling is necessary for carcinoma cell invasiveness and metastasis. Curr Biol. 1998; 8:1243–1252.20. Dedes KJ, Wetterskog D, Ashworth A, Kaye SB, Reis-Filho JS. Emerging therapeutic targets in endometrial cancer. Nat Rev Clin Oncol. 2011; 8:261–271.21. Boivin D, Bilodeau D, Beliveau R. Regulation of cytoskeletal functions by Rho small GTP-binding proteins in normal and cancer cells. Can J Physiol Pharmacol. 1996; 74:801–810.22. Teng Y, Zeisberg M, Kalluri R. Transcriptional regulation of epithelial-mesenchymal transition. J Clin Invest. 2007; 117:304–306.23. Venkov CD, Link AJ, Jennings JL, Plieth D, Inoue T, Nagai K, et al. A proximal activator of transcription in epithelial-mesenchymal transition. J Clin Invest. 2007; 117:482–491.24. Frixen UH, Behrens J, Sachs M, Eberle G, Voss B, Warda A, et al. E-cadherin-mediated cell-cell adhesion prevents invasiveness of human carcinoma cells. J Cell Biol. 1991; 113:173–185.25. Sakuragi N, Nishiya M, Ikeda K, Ohkouch T, Furth EE, Hareyama H, et al. Decreased E-cadherin expression in endometrial carcinoma is associated with tumor dedifferentiation and deep myometrial invasion. Gynecol Oncol. 1994; 53:183–189.26. Hipp S, Walch A, Schuster T, Losko S, Laux H, Bolton T, et al. Activation of epidermal growth factor receptor results in snail protein but not mRNA overexpression in endometrial cancer. J Cell Mol Med. 2009; 13:3858–3867.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Prognostic significance of epidermal growth factor receptor expression in human gastric carcinoma
- Correlation of epidermal growth factor receptor expression with prognostic factors in patients with ovarian neoplasms
- Amplification of epidermal growth factor receptor gene in primary cervical cancer
- Amplification of epidermal growth factor receptor gene in primary cervical cancer
- Epidermal growth factor receptors increase in rabbit embryonal implantation