Hanyang Med Rev.
2015 May;35(2):113-120. 10.7599/hmr.2015.35.2.113.
Regenerative Cell Therapy for the Sensorineural Hearing Loss
- Affiliations
-
- 1Department of Otorhinolaryngology-Head and Neck Surgery, The Catholic University of Korea, College of Medicine, Seoul, Korea. khpent@catholic.ac.kr
- KMID: 1793741
- DOI: http://doi.org/10.7599/hmr.2015.35.2.113
Abstract
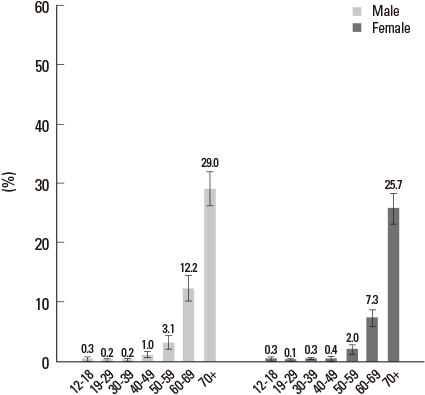
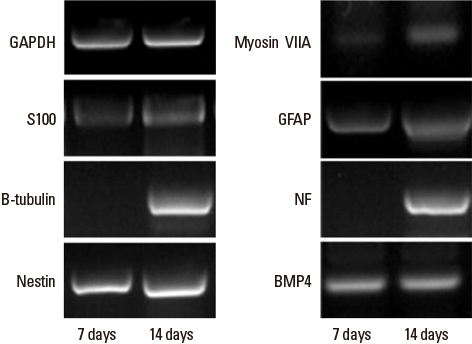
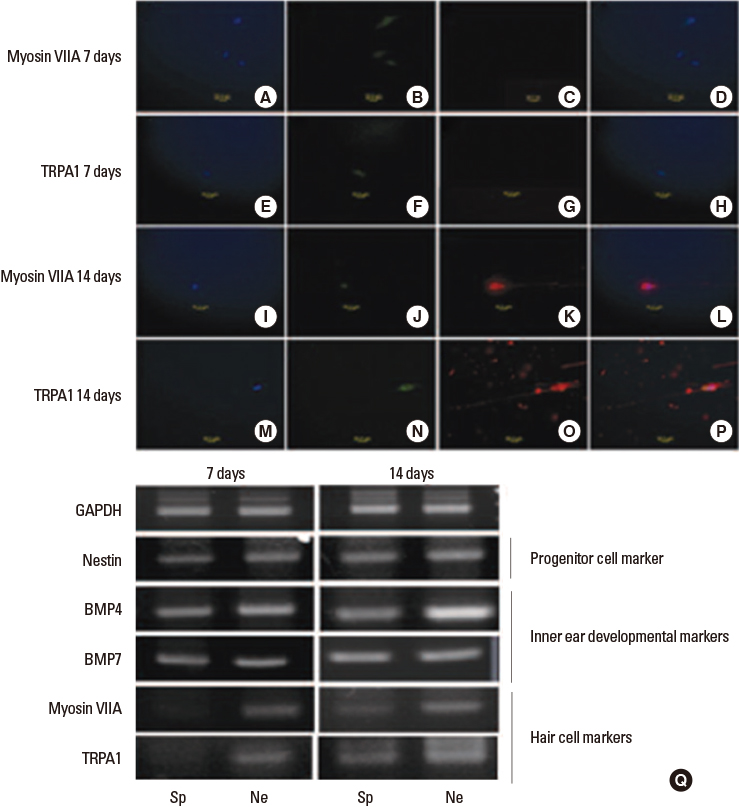
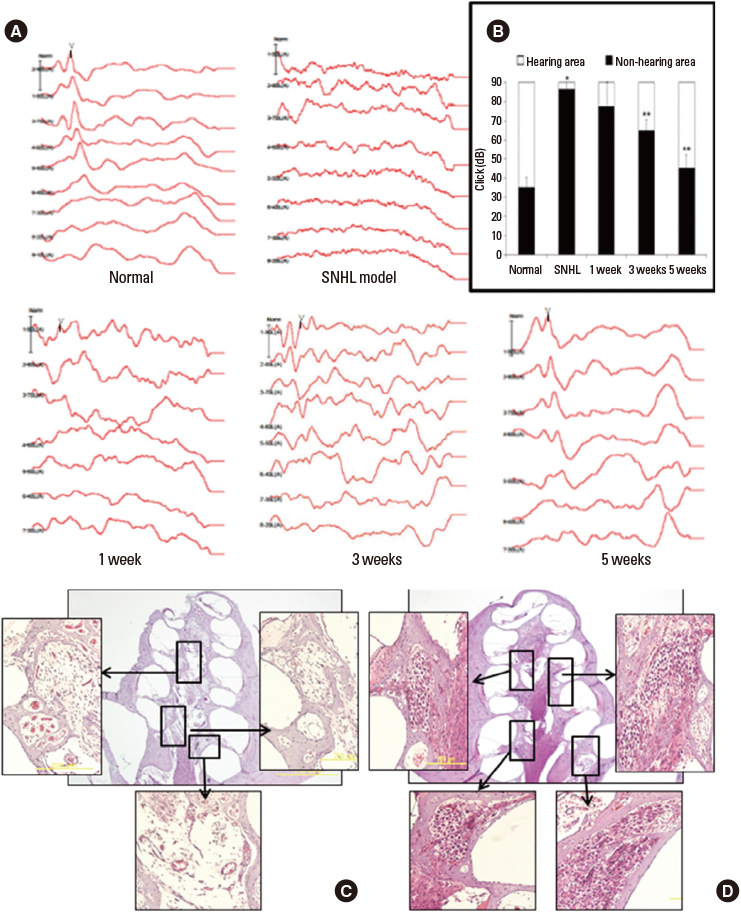
- Sensorineural hearing loss is the most common disability in the world and nearly one third of all individuals over the age of 65 are affected. For hearing handicapped people, many devices (hearing aid, cochlear implant, middle ear implant etc.) have been developed to reduce or overcome the disability. But these devices do not give perfect benefit to the patients functionally and there are aesthetic problems. That is why researchers have interest in regenerative measures to restore or prevent hearing loss. Recently there were fruitful results from gene and stem cell therapy research for hearing loss. Gene therapy with Atoh 1 gene and transplantation of stem cells into the cochlea regenerate damaged hair cells and morphologically restore spiral ganglion neurons allowing functional hearing in the deaf animal model. Based on these results, many countries including Korea have done clinical trials in deaf patients. The past ten years have shown an incredible advancement in medical biotechnology in the otologic field and this progress may someday substitute the medical devices for the hard of hearing patients.
MeSH Terms
Figure
Cited by 1 articles
-
Etiology and Rehabilitation of Sensorineural Hearing Loss
Seung Hwan Lee
Hanyang Med Rev. 2015;35(2):55-56. doi: 10.7599/hmr.2015.35.2.55.
Reference
-
1. Li H, Roblin G, Liu H, Heller S. Generation of hair cells by stepwise differentiation of embryonic stem cells. Proc Natl Acad Sci. 2003; 100:13495–13500.
Article2. Corrales CE, Pan L, Li H, Liberman MC, Heller S, Edge AS. Engraftment and differentiation of embryonic stem cell-derived neural progenitor cells in the cochlear nerve trunk: growth of processes into the organ of corti. J Neurobiol. 2006; 66:1489–1500.
Article3. Kojima K, Tamura S, Nishida AT, Ito J. Generation of inner ear hair cell immunophenotypes from neurospheres obtained from fetal rat central nervous system in vitro. Acta Otolaryngol Suppl. 2004; 26–30.
Article4. Hakuba N, Hata R, Morizane I, Feng G, Shimizu Y, Fujita K, et al. Neural stem cells suppress the hearing threshold shift caused by cochlear ischemia. Neuroreport. 2005; 16:1545–1549.5. Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006; 126:663–676.
Article6. Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007; 131:861–872.
Article7. Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, Tian S, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007; 318:1917–1920.
Article8. Nishimura K, Nakagawa T, Ono K, Ogita H, Sakamoto T, Yamamoto N, et al. Transplantation of mouse induced pluripotent stem cells into the cochlea. Neuroreport. 2009; 20:1250–1254.
Article9. Nishimura K, Nakagawa T, Sakamoto T, Ito J. Fates of murine pluripotent stem cell-derived neural progenitors following transplantation into mouse cochleae. Cell Transplant. 2012; 21:763–771.
Article10. Kopen GC, Prockop DJ, Phinney DG. Marrow stromal cells migrate throughout forebrain and cerebellum, and they differentiate into astrocytes after injection into neonatal mouse brains. Proc Natl Acad Sci. 1999; 96:10711–10716.
Article11. Woodbury D, Schwarz EJ, Prockop DJ, Black IB. Adult rat and human bone marrow stromal cells differentiate into neurons. J Neurosci Res. 2000; 61:364–370.
Article12. Jiang Y, Jahagirdar BN, Reinhardt RL, Schwartz RE, Keene CD, Ortiz-Gonzalez XR, et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature. 2002; 418:41–49.
Article13. Jeon SJ, Oshima K, Heller S, Edge AS. Bone marrow mesenchymal stem cells are progenitors in vitro for inner ear hair cells. Mol Cell Neurosci. 2007; 34:59–68.
Article14. Lee JH, Kang WK, Seo JH, Choi MY, Lee YH, Kim HM, et al. Neural Differentiation of Bone Marrow-Derived Mesenchymal Stem Cells: applicability for Inner Ear Therapy. Korean J Audiol. 2012; 16:47–53.
Article15. Naito Y, Nakamura T, Nakagawa T, Iguchi F, Endo T, Fujino K, et al. Transplantation of bone marrow stromal cells into the cochlea of chinchillas. Neuroreport. 2004; 15:1–4.
Article16. Cho YB, Cho HH, Jang S, Jeong HS, Park JS. Transplantation of neural differentiated human mesenchymal stem cells into the cochlea of an auditory-neuropathy guinea pig model. J Korean Med Sci. 2011; 26:492–498.
Article17. Portmann-Lanz CB, Schoeberlein A, Huber A, Sager R, Malek A, Holzgreve W, et al. Placental mesenchymal stem cells as potential autologous graft for pre- and perinatal neuroregeneration. Am J Obstet Gynecol. 2006; 194:664–673.
Article18. Tsai MS, Lee JL, Chang YJ, Hwang SM. Isolation of human multipotent mesenchymal stem cells from second-trimester amniotic fluid using a novel two-stage culture protocol. Hum Reprod. 2004; 19:1450–1456.
Article19. Mitchell KE, Weiss ML, Mitchell BM, Martin P, Davis D, Morales L, et al. Matrix cells from Wharton's jelly form neurons and glia. Stem Cells. 2003; 21:50–60.
Article20. Wang HS, Hung SC, Peng ST, Huang CC, Wei HM, Guo YJ, et al. Mesenchymal stem cells in the Wharton's jelly of the human umbilical cord. Stem Cells. 2004; 22:1330–1337.
Article21. Lee OK, Kuo TK, Chen WM, Lee KD, Hsieh SL, Chen TH. Isolation of multipotent mesenchymal stem cells from umbilical cord blood. Blood. 2004; 103:1669–1675.
Article22. Park KS, Lee YS, Kang KS. In vitro neuronal and osteogenic differentiation of mesenchymal stem cells from human umbilical cord blood. J Vet Sci. 2006; 7:343–348.
Article23. Kang XQ, Zang WJ, Bao LJ, Li DL, Xu XL, Yu XJ. Differentiating characterization of human umbilical cord blood-derived mesenchymal stem cells in vitro. Cell Biol Int. 2006; 30:569–575.
Article24. Choi MY, Gil KC, Back SA, Park SN, Yeo SW, Park KH. Auditory cell induction from mesenchymal stem cells of human umbilical cord blood. J Int Adv Otol. 2011; 7:137–147.25. Choi MY, Yeo SW, Park KH. Hearing restoration in a deaf animal model with intravenous transplantation of mesenchymal stem cells derived from human umbilical cord blood. Biochem Biophys Res Commun. 2012; 427:629–636.
Article26. Izumikawa M, Minoda R, Kawamoto K, Abrashkin KA, Swiderski DL, Dolan DF, et al. Auditory hair cell replacement and hearing improvement by Atoh1 gene therapy in deaf mammals. Nat Med. 2005; 11:271–276.
Article27. Richardson RT, Atkinson PJ. Atoh1 gene therapy in the cochlea for hair cell regeneration. Expert Opin Biol Ther. 2015; 15:417–430.
Article28. Zheng JL, Helbig C, Gao WQ. Induction of cell proliferation by fibroblast and insulin-like growth factors in pure rat inner ear epithelial cell cultures. J Neurosci. 1997; 17:216–226.
Article29. Yamamoto N, Nakagawa T, Ito J. Application of insulin-like growth factor-1 in the treatment of inner ear disorders. Front Pharmacol. 2014; 5:208.
Article30. Altschuler RA, Cho Y, Ylikoski J, Pirvola U, Magal E, Miller JM. Rescue and regrowth of sensory nerves following deafferentation by neurotrophic factors. Ann N Y Acad Sci. 1999; 884:305–311.
Article31. Oh JH, Park SN, Choi Hg, Choi NY, Han MA, Park KH, et al. Effect of Neurotrophic factors on hearing restoration and spiral ganglion regeneration in deafened animal model. Tissue Eng Regen Med. 2008; 5:849–854.32. Cho JH, Park KH, Park SN, Yeo SW. Morphologic Change and Hearing Recovery After Intratympanic Application of Insulin-Like Growth Factor-1 in Guinea Pig. Int Adv Otol. 2005; 6:46–52.33. Oshima K, Suchert S, Blevins NH, Heller S. Curing hearing loss: patient expectations, health care practitioners, and basic science. J Commun Disord. 2010; 43:311–318.
Article34. Slowik AD, Bermingham-McDonogh O. Hair cell generation by notch inhibition in the adult mammalian cristae. J Assoc Res Otolaryngol. 2013; 14:813–828.
Article35. Li H, Liu H, Heller S. Pluripotent stem cells from the adult mouse inner ear. Nat Med. 2003; 9:1293–1299.
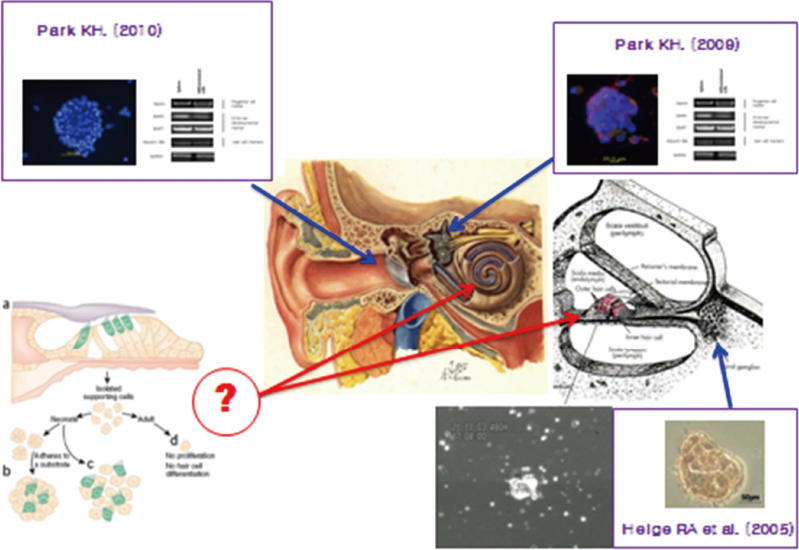
Article36. Park KH, Park SN, Seo JH, Yeo SW, Choi HG, Chang KH. Nestin Expression in Proliferating Cells of Cultured Human Vestibular Organs. Int Adv Otol. 2009; 5:6–10.37. Oshima K, Grimm CM, Corrales CE, Senn P, Monedero RM, Géléoc GS, et al. Differential distribution of stem cells in the auditory and vestibular organs of the inner ear. J Assoc Res Otolaryngol. 2007; 8:18–31.
Article38. Rask-Andersen H, Bostrom M, Gerdin B, Kinnefors A, Nyberg G, Engstrand T, et al. Regeneration of human auditory nerve. In vitro/in video demonstration of neural progenitor cells in adult human and guinea pig spiral ganglion. Hear Res. 2005; 203:180–191.
Article39. Choi HG, Kim CH, Park SN, Choi MY, Nam SG, Park SC, et al. Isolation and Culture of Adult Neural Stem Cells from Guinea Pig Spiral Ganglion. Tissue Eng Regen Med. 2009; 6:938–941.40. Park KH. Endogenous Stem Cells in the Ear. Korean J Otorhinolaryngol-Head Neck Surg. 2013; 56:749–753.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Stem Cell Therapy for Sensorineural Hearing Loss, Still Alive?
- Hearing loss, stem cells, and photobiomodulation
- Rehabilitation of Sensorineural Hearing Loss: Hearing Aid
- The Characteristics and the Changes of Tinnitus according to the Recovery of Hearing Loss in the Patients with Sudden Hearing Loss
- Sensorineural Hearing Loss: Causes and Hearing Rehabilitation