Ann Surg Treat Res.
2018 Oct;95(4):222-229. 10.4174/astr.2018.95.4.222.
Usefulness of dynamic risk stratification in pediatric patients with differentiated thyroid carcinoma
- Affiliations
-
- 1Department of Surgery, Yonsei University College of Medicine, Seoul, Korea. JUNGJONGJ@yuhs.ac
- KMID: 2421200
- DOI: http://doi.org/10.4174/astr.2018.95.4.222
Abstract
- PURPOSE
Recently, the American Thyroid Association (ATA) dynamic risk stratification (DRS) has been verified to be more valuable than the static anatomical staging system for predicting prognosis in patients with differentiated thyroid carcinoma (DTC). The purpose of this retrospective study was to compare the clinical usefulness of DRS, which is based on the response to initial treatment, with that of ATA initial risk stratification in pediatric patients.
METHODS
A total of 144 pediatric patients underwent thyroid operation from August 1982 to December 2013 at Yonsei University Hospital (Seoul, Korea). Among them, 128 patients with complete clinical data were enrolled in this study. Clinicopathologic features and surgical outcomes were retrospectively analyzed by medical chart review. The mean follow-up duration was 11.5 years.
RESULTS
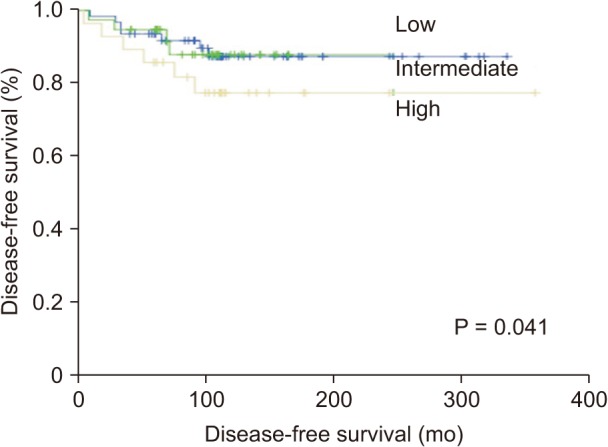
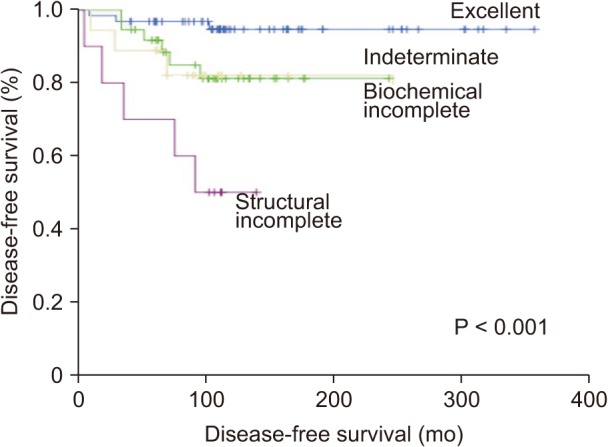
The mean tumor size was 2.1 cm; 80.4% of patients were diagnosed with conventional papillary thyroid carcinoma, and 7.0% of patients were diagnosed with follicular thyroid carcinoma. Low-risk patients had the highest probability of an excellent response to initial treatment (66.6%). High-risk patients had the highest probability of a structural incomplete response (100%) and the lowest probability of an excellent response (11.1%). The ATA risk stratification and the DRS system were independent risk factors for disease-free survival (DFS) (P = 0.041 and P < 0.001, respectively).
CONCLUSION
The DRS system, which is based on the response to initial treatment, can offer more useful prognostic information compared with ATA risk stratification in pediatric patients with DTC.
Keyword
MeSH Terms
Figure
Reference
-
1. Oh CM, Won YJ, Jung KW, Kong HJ, Cho H, Lee JK, et al. Cancer statistics in Korea: incidence, mortality, survival, and prevalence in 2013. Cancer Res Treat. 2016; 48:436–450. PMID: 26987395.
Article2. Enewold L, Zhu K, Ron E, Marrogi AJ, Stojadinovic A, Peoples GE, et al. Rising thyroid cancer incidence in the United States by demographic and tumor characteristics, 1980–2005. Cancer Epidemiol Biomarkers Prev. 2009; 18:784–791. PMID: 19240234.
Article3. Davies L, Welch HG. Current thyroid cancer trends in the United States. JAMA Otolaryngol Head Neck Surg. 2014; 140:317–322. PMID: 24557566.
Article4. Collini P, Massimino M, Leite SF, Mattavelli F, Seregni E, Zucchini N, et al. Papillary thyroid carcinoma of childhood and adolescence: a 30-year experience at the Istituto Nazionale Tumori in Milan. Pediatr Blood Cancer. 2006; 46:300–306. PMID: 16047353.
Article5. Park S, Jeong JS, Ryu HR, Lee CR, Park JH, Kang SW, et al. Differentiated thyroid carcinoma of children and adolescents: 27-year experience in the yonsei university health system. J Korean Med Sci. 2013; 28:693–699. PMID: 23678260.
Article6. Pires BP, Alves PA Jr, Bordallo MA, Bulzico DA, Lopes FP, Farias T, et al. Prognostic factors for early and long-term remission in pediatric differentiated thyroid carcinoma: the role of sex, age, clinical presentation, and the newly proposed American Thyroid Association Risk Stratification System. Thyroid. 2016; 26:1480–1487. PMID: 27540892.
Article7. Demidchik YE, Demidchik EP, Reiners C, Biko J, Mine M, Saenko VA, et al. Comprehensive clinical assessment of 740 cases of surgically treated thyroid cancer in children of Belarus. Ann Surg. 2006; 243:525–532. PMID: 16552205.
Article8. Welch Dinauer CA, Tuttle RM, Robie DK, McClellan DR, Svec RL, Adair C, et al. Clinical features associated with metastasis and recurrence of differentiated thyroid cancer in children, adolescents and young adults. Clin Endocrinol (Oxf). 1998; 49:619–628. PMID: 10197078.
Article9. Schlumberger M, De Vathaire F, Travagli JP, Vassal G, Lemerle J, Parmentier C, et al. Differentiated thyroid carcinoma in childhood: long term follow-up of 72 patients. J Clin Endocrinol Metab. 1987; 65:1088–1094. PMID: 3680475.
Article10. Lazar L, Lebenthal Y, Steinmetz A, Yackobovitch-Gavan M, Phillip M. Differentiated thyroid carcinoma in pediatric patients: comparison of presentation and course between pre-pubertal children and adolescents. J Pediatr. 2009; 154:708–714. PMID: 19167722.
Article11. Nam KH, Lim CY, Lee J, Chang HS, Chung WY, Choi SH, et al. Differentiated thyroid carcinoma in patients less than 20 years of age at diagnosis: clinicopathologic characteristics and prognostic factors. J Korean Surg Soc. 2005; 69:443–449.12. Jarzab B, Handkiewicz Junak D, Wloch J, Kalemba B, Roskosz J, Kukulska A, et al. Multivariate analysis of prognostic factors for differentiated thyroid carcinoma in children. Eur J Nucl Med. 2000; 27:833–841. PMID: 10952495.13. Papendieck P, Gruneiro-Papendieck L, Venara M, Acha O, Cozzani H, Mateos F, et al. Differentiated thyroid cancer in children: prevalence and predictors in a large cohort with thyroid nodules followed prospectively. J Pediatr. 2015; 167:199–201. PMID: 26117640.
Article14. Alzahrani AS, Alkhafaji D, Tuli M, Al-Hindi H, Sadiq BB. Comparison of differentiated thyroid cancer in children and adolescents (≤20 years) with young adults. Clin Endocrinol (Oxf). 2016; 84:571–577. PMID: 26118454.15. Markovina S, Grigsby PW, Schwarz JK, DeWees T, Moley JF, Siegel BA, et al. Treatment approach, surveillance, and outcome of well-differentiated thyroid cancer in childhood and adolescence. Thyroid. 2014; 24:1121–1126. PMID: 24731094.
Article16. Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010; 17:1471–1474. PMID: 20180029.
Article17. Francis GL, Waguespack SG, Bauer AJ, Angelos P, Benvenga S, Cerutti JM, et al. Management guidelines for children with thyroid nodules and differentiated thyroid cancer. Thyroid. 2015; 25:716–759. PMID: 25900731.
Article18. Tuttle RM, Tala H, Shah J, Leboeuf R, Ghossein R, Gonen M, et al. Estimating risk of recurrence in differentiated thyroid cancer after total thyroidectomy and radioactive iodine remnant ablation: using response to therapy variables to modify the initial risk estimates predicted by the new American Thyroid Association staging system. Thyroid. 2010; 20:1341–1349. PMID: 21034228.
Article19. Sung TY, Jeon MJ, Lee YH, Lee YM, Kwon H, Yoon JH, et al. Initial and dynamic risk stratification of pediatric patients with differentiated thyroid cancer. J Clin Endocrinol Metab. 2017; 102:793–800. PMID: 27809646.
Article20. Jeon MJ, Kim WG, Park WR, Han JM, Kim TY, Song DE, et al. Modified dynamic risk stratification for predicting recurrence using the response to initial therapy in patients with differentiated thyroid carcinoma. Eur J Endocrinol. 2013; 170:23–30. PMID: 24088549.
Article21. Krajewska J, Chmielik E, Jarząb B. Dynamic risk stratification in the follow-up of thyroid cancer: what is still to be discovered in 2017? Endocr Relat Cancer. 2017; 24:R387–R402. PMID: 28821573.
Article22. Schemper M. The relative importance of prognostic factors in studies of survival. Stat Med. 1993; 12:2377–2382. PMID: 8134740.
Article23. Lazar L, Lebenthal Y, Segal K, Steinmetz A, Strenov Y, Cohen M, et al. Pediatric thyroid cancer: postoperative classifications and response to initial therapy as prognostic factors. J Clin Endocrinol Metab. 2016; 101:1970–1979. PMID: 26930182.
Article24. Spencer CA. Clinical review: clinical utility of thyroglobulin antibody (TgAb) measurements for patients with differentiated thyroid cancers (DTC). J Clin Endocrinol Metab. 2011; 96:3615–3627. PMID: 21917876.25. Sigurdson AJ, Ronckers CM, Mertens AC, Stovall M, Smith SA, Liu Y, et al. Primary thyroid cancer after a first tumour in childhood (the Childhood Cancer Survivor Study): a nested case-control study. Lancet. 2005; 365:2014–2023. PMID: 15950715.
Article26. Grigsby PW, Gal-or A, Michalski JM, Doherty GM. Childhood and adolescent thyroid carcinoma. Cancer. 2002; 95:724–729. PMID: 12209714.
Article27. Alessandri AJ, Goddard KJ, Blair GK, Fryer CJ, Schultz KR. Age is the major determinant of recurrence in pediatric differentiated thyroid carcinoma. Med Pediatr Oncol. 2000; 35:41–46. PMID: 10881006.
Article28. Lee YA, Jung HW, Kim HY, Choi H, Kim HY, Hah JH, et al. Pediatric patients with multifocal papillary thyroid cancer have higher recurrence rates than adult patients: a retrospective analysis of a large pediatric thyroid cancer cohort over 33 years. J Clin Endocrinol Metab. 2015; 100:1619–1629. PMID: 25632969.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- TSH Suppression after Differentiated Thyroid Cancer Surgery and Osteoporosis
- Risk stratification of thyroid nodules on ultrasonography with the French TI-RADS: description and reflections
- Clinical Analysis of Risk Factors Related to Recurrent Well Differentiated Thyroid Carcinoma
- N Stage: Controversies and Recent Issues
- Surgery for Advanced Nodal Metastasis in Thyroid Cancer