Korean J Transplant.
2019 Sep;33(3):47-54. 10.4285/jkstn.2019.33.3.47.
Clinical significance of donor-specific anti-HLA-DR51/52/53 antibodies for antibody-mediated rejection in kidney transplant recipients
- Affiliations
-
- 1Department of Laboratory Medicine, Severance Hospital, Yonsei University College of Medicine, Seoul, Korea. kimhs54@yuhs.ac
- 2Department of Laboratory Medicine, Korea University Guro Hospital, Seoul, Korea.
- 3Department of Surgery, Severance Hospital, Yonsei University College of Medicine, Seoul, Korea.
- KMID: 2468179
- DOI: http://doi.org/10.4285/jkstn.2019.33.3.47
Abstract
- BACKGROUND
The presence of donor-specific antibodies (DSAs) to human leukocyte antigen (HLA) increases the risk of antibody-mediated rejection (ABMR) after kidney transplantation (KT). However, the clinical relevance of anti-HLA-DR51/52/53 antibodies remains unclear because of their weak antigen expression. This study evaluated the association between anti-HLA-DR51/52/53 DSAs and ABMR.
METHODS
We retrospectively reviewed the single-antigen-bead panel reactive antibody (single PRA) results of 130 patients tested between August 1, 2009 and March 6, 2015, based on clinical necessity after allograft KT. Single PRA analysis was performed using Luminex assay kits (Lifecodes LSA class I and II). We reviewed the clinical course and biopsy results of patients with anti-HLA-DR51/52/53 DSAs.
RESULTS
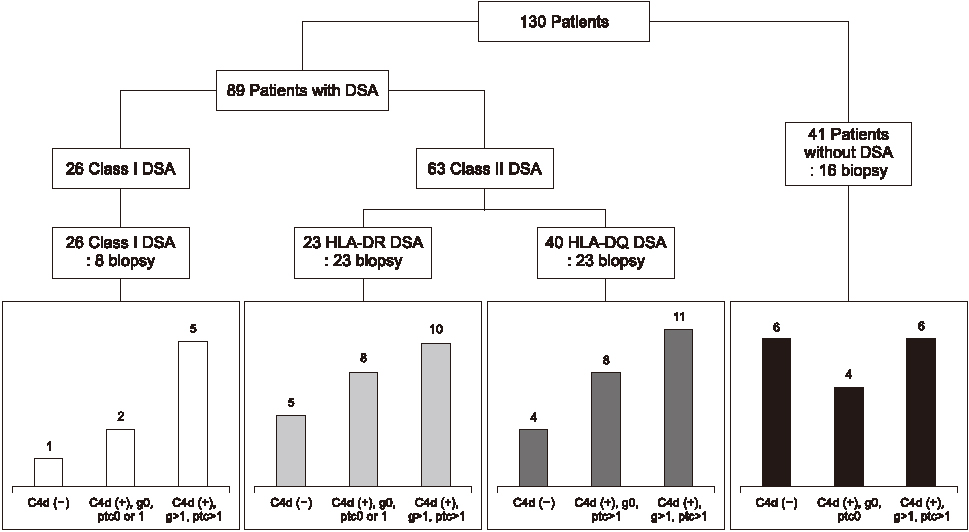
Post-KT DSAs were identified in 89 of the 130 patients (68.5%), with 26 of 32 class I DSAs and 63 of 66 class II DSAs being immunodominant DSAs. Thirteen patients had anti-HLA-DR51/52/53 DSAs. Three patients with anti-HLA-DR51/52/53 immunodominant DSAs alone were diagnosed with biopsy-proven ABMR. One patient who developed anti-HLA-DR DSA 13 days after KT showed a rapid increase in anti-HLA-DR51 DSA and had biopsy-proven ABMR.
CONCLUSIONS
Although the expression of the HLA-DR51/52/53 antigen was weak, anti-HLA-DR51/52/53 DSAs might be correlated with biopsy-proven ABMR. Therefore, anti-HLA-DR51/52/53 DSAs must be evaluated as a cause of ABMR after transplantation.
MeSH Terms
Figure
Reference
-
1. Nankivell BJ, Alexander SI. Rejection of the kidney allograft. N Engl J Med. 2010; 363:1451–1462.
Article2. Cardarelli F, Pascual M, Tolkoff-Rubin N, Delmonico FL, Wong W, Schoenfeld DA, et al. Prevalence and significance of anti-HLA and donor-specific antibodies long-term after renal transplantation. Transpl Int. 2005; 18:532–540.
Article3. Haas M. An updated Banff schema for diagnosis of antibody-mediated rejection in renal allografts. Curr Opin Organ Transplant. 2014; 19:315–322.
Article4. Zemmour J, Parham P. Distinctive polymorphism at the HLA-C locus: implications for the expression of HLA-C. J Exp Med. 1992; 176:937–950.
Article5. Berdoz J, Gorski J, Termijtelen AM, Dayer JM, Irlé C, Schendel D, et al. Constitutive and induced expression of the individual HLA-DR beta and alpha chain loci in different cell types. J Immunol. 1987; 139:1336–1341.6. Stunz LL, Karr RW, Anderson RA. HLA-DRB1 and -DRB4 genes are differentially regulated at the transcriptional level. J Immunol. 1989; 143:3081–3086.7. Kosmoliaptsis V, Gjorgjimajkoska O, Sharples LD, Chaudhry AN, Chatzizacharias N, Peacock S, et al. Impact of donor mismatches at individual HLA-A, -B, -C, -DR, and -DQ loci on the development of HLA-specific antibodies in patients listed for repeat renal transplantation. Kidney Int. 2014; 86:1039–1048.
Article8. Mauiyyedi S, Crespo M, Collins AB, Schneeberger EE, Pascual MA, Saidman SL, et al. Schneeberger EE, Pascual MA, Saidman SL, et al. Acute humoral rejection in kidney transplantation: II. morphology, immunopathology, and pathologic classification. J Am Soc Nephrol. 2002; 13:779–787.9. Zachary AA, Montgomery RA, Leffell MS. Factors associated with and predictive of persistence of donor-specific antibody after treatment with plasmapheresis and intravenous immunoglobulin. Hum Immunol. 2005; 66:364–370.
Article10. Cornell LD, Smith RN, Colvin RB. Kidney transplantation: mechanisms of rejection and acceptance. Annu Rev Pathol. 2008; 3:189–220.
Article11. Singal DP, Ye M, Joseph S. Lack of HLA-DR2-associated DRB1 gene in a family. Tissue Antigens. 1997; 50:85–88.
Article12. Song EY, Park MH, Kang SJ, Park HJ, Kim BC, Tokunaga K, et al. HLA class II allele and haplotype frequencies in Koreans based on 107 families. Tissue Antigens. 2002; 59:475–486.
Article13. Song EY, Kang SJ, Lee YJ, Park MH. HLA-DR2-associated DRB1 and DRB5 alleles and haplotypes in Koreans. Hum Immunol. 2000; 61:937–941.
Article14. Tait BD, Süsal C, Gebel HM, Nickerson PW, Zachary AA, Claas FH, et al. Consensus guidelines on the testing and clinical management issues associated with HLA and non-HLA antibodies in transplantation. Transplantation. 2013; 95:19–47.
Article15. Aubert O, Bories MC, Suberbielle C, Snanoudj R, Anglicheau D, Rabant M, et al. Risk of antibody-mediated rejection in kidney transplant recipients with anti-HLA-C donor-specific antibodies. Am J Transplant. 2014; 14:1439–1445.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Evaluating the effect of Anti-HLA-DR51/52/53 donor-specific antibodies on antibody-mediated rejection in kidney transplantation recipients
- Effect of preexisting human leukocyte antigen donor-specific antibodies especially human leukocyte antigen-DQ on kidney transplant outcome
- Investigation of non-human leukocyte antigen antibodies and epitope mismatch in kidney transplant recipients with chronic antibody-mediated rejection
- Association of preoperative non-HLA antibodies with kidney allograft rejection
- Serum antibody screening for non-human leukocyte antigen antibodies associated with antibody-mediate rejection reveals significance of anti-collagen type I and type III antibodies