Korean J Orthod.
2019 Jul;49(4):205-213. 10.4041/kjod.2019.49.4.205.
Comparison of tooth movement and biological response in corticotomy and micro-osteoperforation in rabbits
- Affiliations
-
- 1Graduate School, The Catholic University of Korea, Seoul, Korea.
- 2Department of Orthodontics, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea. kook2002@catholic.ac.kr
- 3Department of Preventive Dentistry, College of Dentistry, King Faisal University, Al-hofuf, Saudi Arabia.
- 4Postgraduate Orthodontic Program, Arizona School of Dentistry & Oral Health, A.T. Still University, Mesa, AZ, USA.
- 5Graduate School of Dentistry, Kyung Hee University, Seoul, Korea.
- 6Department of Dentistry, Uijeongbu St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Uijeongbu, Korea.
- 7Department of Orthodontics, Faculty of Dentistry, Ain Shams University, Cairo, Egypt.
- KMID: 2453057
- DOI: http://doi.org/10.4041/kjod.2019.49.4.205
Abstract
OBJECTIVE
The aim of this study was to evaluate the amount of tooth movement and histologic changes with different corticotomy designs and micro-osteoperforation in rabbits.
METHODS
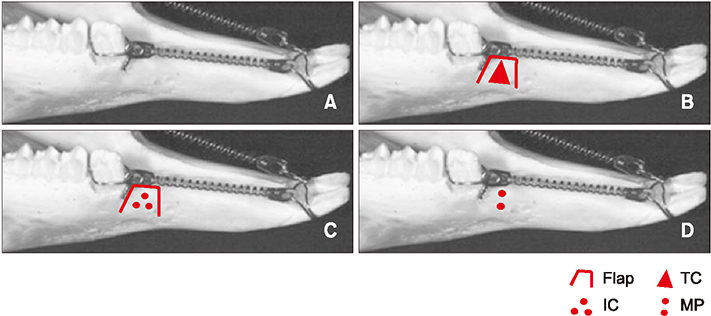
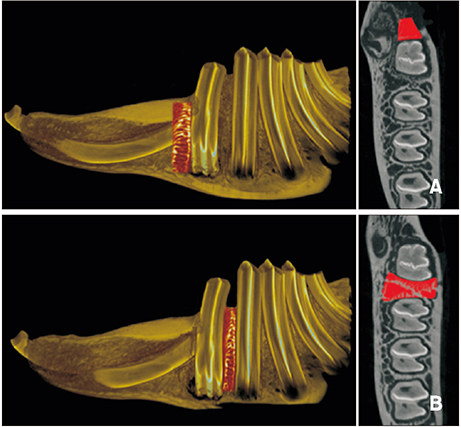
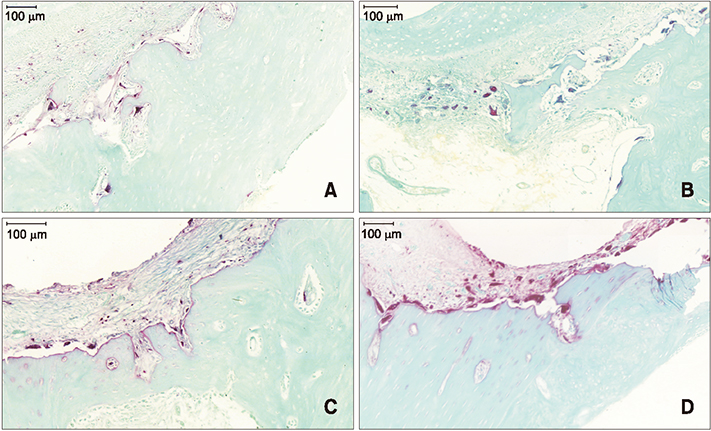
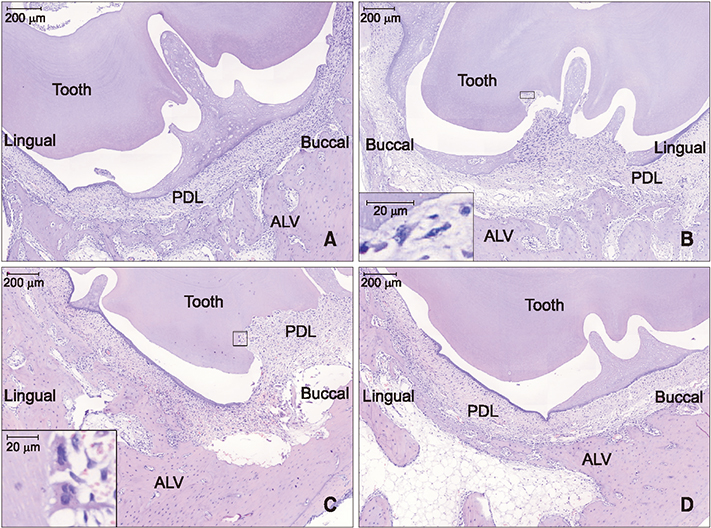
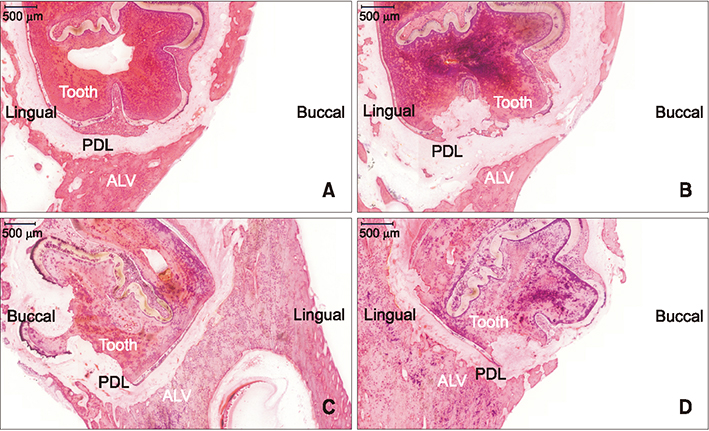
The sample consisted of 24 rabbits divided into three experimental groups (triangular corticotomy [TC] and indentation corticotomy [IC] with flap, and flapless micro-osteoperforations [MP]) and a control. A traction force of 100 cN was applied by connecting the first premolars to the incisors. The amount of tooth movement was measured. Kruskal-Wallis test was used to assess differences in tooth movement between the groups. Micro-computed tomography, hematoxylin and eosin staining, and tartrate-resistant acidic phosphatase (TRAP) analysis were performed. Analysis of variance was applied to assess differences in TRAP-positive osteoclast count between the groups.
RESULTS
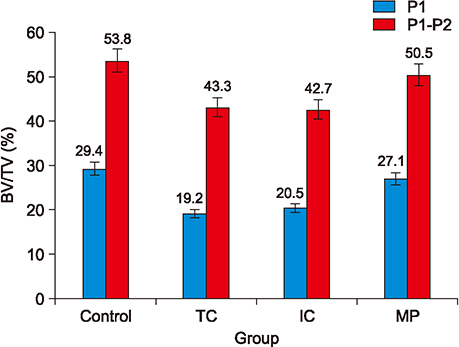
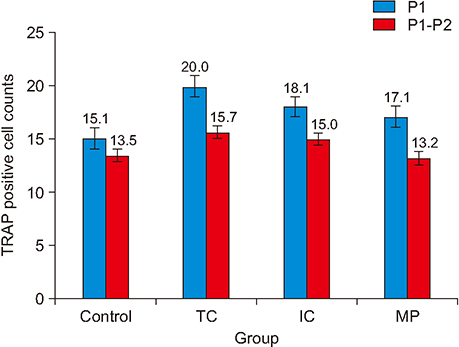
The amount of tooth movement increased by 46.5% and 32.0% in the IC and MP groups, respectively, while the bone fraction analysis showed 69.7% and 8.5% less mineralization compared to the control. There were no significant intergroup differences in the number of TRAP-positive osteoclasts.
CONCLUSIONS
The micro-osteoperforation group showed no significant differences in the amount of tooth movement compared to the corticotomy groups, nor in the TRAP-positive osteoclast count compared to both corticotomy groups and control.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Comparison of the effects of horizontal and vertical micro-osteoperforations on the biological response and tooth movement in rabbits
Seok-gon Kim, Yoon-Ah Kook, Hee Jin Lim, Patrick Park, Won Lee, Jae Hyun Park, Mohamed Bayome, Yoonji Kim
Korean J Orthod. 2021;51(5):304-312. doi: 10.4041/kjod.2021.51.5.304.
Reference
-
1. Igarashi K, Mitani H, Adachi H, Shinoda H. Anchorage and retentive effects of a bisphosphonate (AHBuBP) on tooth movements in rats. Am J Orthod Dentofacial Orthop. 1994; 106:279–289.
Article2. Işeri H, Kişnişci R, Bzizi N, Tüz H. Rapid canine retraction and orthodontic treatment with dentoalveolar distraction osteogenesis. Am J Orthod Dentofacial Orthop. 2005; 127:533–541. quiz 625.
Article3. Liou EJ, Huang CS. Rapid canine retraction through distraction of the periodontal ligament. Am J Orthod Dentofacial Orthop. 1998; 114:372–382.
Article4. Gantes B, Rathbun E, Anholm M. Effects on the periodontium following corticotomy-facilitated orthodontics. Case reports. J Periodontol. 1990; 61:234–238.
Article5. Chung KR, Oh MY, Ko SJ. Corticotomy-assisted orthodontics. J Clin Orthod. 2001; 35:331–339.6. Suya H. Corticotomy in orthodontics. In : Hosl E, Baldauf A, editors. Mechanical and biological basics in orthodontic therapy. Heidelberg: Huthig Buch Verlag;1991. p. 207–226.7. Kook YA, Lee W, Kim SH, Chung KR. Corticotomy-assisted space closure in adult patients with missing lower molars. J Clin Orthod. 2013; 47:85–95. quiz 139.8. McBride MD, Campbell PM, Opperman LA, Dechow PC, Buschang PH. How does the amount of surgical insult affect bone around moving teeth? Am J Orthod Dentofacial Orthop. 2014; 145:S92–S99.
Article9. Mostafa YA, Mohamed Salah, Mehanni S, ElBokle NN, Heider AM. Comparison of corticotomy-facilitated vs standard tooth-movement techniques in dogs with miniscrews as anchor units. Am J Orthod Dentofacial Orthop. 2009; 136:570–577.
Article10. Teixeira CC, Khoo E, Tran J, Chartres I, Liu Y, Thant LM, et al. Cytokine expression and accelerated tooth movement. J Dent Res. 2010; 89:1135–1141.
Article11. Alikhani M, Raptis M, Zoldan B, Sangsuwon C, Lee YB, Alyami B, et al. Effect of micro-osteoperforations on the rate of tooth movement. Am J Orthod Dentofacial Orthop. 2013; 144:639–648.
Article12. Cassetta M, Altieri F, Pandolfi S, Giansanti M. The combined use of computer-guided, minimally invasive, flapless corticotomy and clear aligners as a novel approach to moderate crowding: a case report. Korean J Orthod. 2017; 47:130–141.
Article13. Charavet C, Lecloux G, Bruwier A, Rompen E, Maes N, Limme M, et al. Localized piezoelectric alveolar decortication for orthodontic treatment in adults: a randomized controlled trial. J Dent Res. 2016; 95:1003–1009.
Article14. Ruso S, Campbell PM, Rossmann J, Opperman LA, Taylor RW, Buschang PH. Bone response to buccal tooth movements-with and without flapless alveolar decortication. Eur J Orthod. 2014; 36:613–623.
Article15. Swapp A, Campbell PM, Spears R, Buschang PH. Flapless cortical bone damage has no effect on medullary bone mesial to teeth being moved. Am J Orthod Dentofacial Orthop. 2015; 147:547–558.
Article16. Lindskog-Stokland B, Hansen K, Ekestubbe A, Wennström JL. Orthodontic tooth movement into edentulous ridge areas--a case series. Eur J Orthod. 2013; 35:277–285.
Article17. Nagaraj K, Upadhyay M, Yadav S. Titanium screw anchorage for protraction of mandibular second molars into first molar extraction sites. Am J Orthod Dentofacial Orthop. 2008; 134:583–591.
Article18. Santos PBDD, Herrera Sanches FS, Ferreira MC, de Almeida ALPF, Janson G, Garib D. Movement of mandibular molar into edentulous alveolar ridge: a cone-beam computed tomography study. Am J Orthod Dentofacial Orthop. 2017; 151:907–913.
Article19. Kim T, Handa A, Iida J, Yoshida S. RANKL expression in rat periodontal ligament subjected to a continuous orthodontic force. Arch Oral Biol. 2007; 52:244–250.
Article20. Yu JY, Lee W, Park JH, Bayome M, Kim Y, Kook YA. Histologic effects of intentional-socket-assisted orthodontic movement in rabbits. Korean J Orthod. 2012; 42:207–217.
Article21. Chen YW, Wang HC, Gao LH, Liu C, Jiang YX, Qu H, et al. Osteoclastogenesis in local alveolar bone in early decortication-facilitated orthodontic tooth movement. PLoS One. 2016; 11:e0153937.
Article22. Alkebsi A, Al-Maaitah E, Al-Shorman H, Abu Alhaija E. Three-dimensional assessment of the effect of micro-osteoperforations on the rate of tooth movement during canine retraction in adults with Class II malocclusion: a randomized controlled clinical trial. Am J Orthod Dentofacial Orthop. 2018; 153:771–785.
Article23. Cheung T, Park J, Lee D, Kim C, Olson J, Javadi S, et al. Ability of mini-implant-facilitated micro-osteoperforations to accelerate tooth movement in rats. Am J Orthod Dentofacial Orthop. 2016; 150:958–967.
Article24. Buschang PH. Surgically facilitated orthodontics: what does the evidence say?. In : Kapila SD, Goonewardene M, Kim-Berman H, Koster KY, editors. Interdisciplinary therapy: using contemporary approaches for complex cases. Ann Arbor: The University of Michigan;2016. p. 135–165.25. Aboul-Ela SM, El-Beialy AR, El-Sayed KM, Selim EM, El-Mangoury NH, Mostafa YA. Miniscrew implant-supported maxillary canine retraction with and without corticotomy-facilitated orthodontics. Am J Orthod Dentofacial Orthop. 2011; 139:252–259.
Article26. Roberts WE. Bone tissue interface. Int J Oral Implantol. 1988; 5:71–74.
Article27. Mapara M, Thomas BS, Bhat KM. Rabbit as an animal model for experimental research. Dent Res J (Isfahan). 2012; 9:111–118.
Article28. Castañeda S, Largo R, Calvo E, Rodríguez-Salvanés F, Marcos ME, Díaz-Curiel M, et al. Bone mineral measurements of subchondral and trabecular bone in healthy and osteoporotic rabbits. Skeletal Radiol. 2006; 35:34–41.
Article29. Chan E, Dalci O, Petocz P, Papadopoulou AK, Darendeliler MA. Physical properties of root cementum: part 26. Effects of micro-osteoperforations on orthodontic root resorption: a microcomputed tomography study. Am J Orthod Dentofacial Orthop. 2018; 153:204–213.
Article30. Owen KM, Campbell PM, Feng JQ, Dechow PC, Buschang PH. Elevation of a full-thickness mucoperiosteal flap alone accelerates orthodontic tooth movement. Am J Orthod Dentofacial Orthop. 2017; 152:49–57.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Corticotomy and the Intrusive Tooth Movement
- Comparison of the effects of horizontal and vertical micro-osteoperforations on the biological response and tooth movement in rabbits
- Corticotomy and the molar uprighting
- Corticotomy for orthodontic tooth movement
- The effectiveness of corticotomy and piezocision on canine retraction: A systematic review