J Breast Cancer.
2019 Jun;22(2):297-310. 10.4048/jbc.2019.22.e18.
Comparative Analysis of Outcomes and Clinicopathological Characteristics of Synchronous and Metachronous Contralateral Breast Cancer: A Study of the SEER Database
- Affiliations
-
- 1Department of Medical Oncology, The First Affiliated Hospital of Xi'an Jiaotong University, Xi'an, China. qiwudanfeng@sina.com
- 2Breast Surgical Oncology, University of Texas MD Anderson Cancer Center, Houston, USA.
- KMID: 2450123
- DOI: http://doi.org/10.4048/jbc.2019.22.e18
Abstract
- PURPOSE
Numerous previous studies have reported inconsistent results about the differences between synchronous contralateral breast cancer (sCBC) and metachronous contralateral breast cancer (mCBC). This study aimed to compare the clinical characteristics and outcomes between sCBC and mCBC and determine predictive factors for the survival of sCBC and mCBC patients.
METHODS
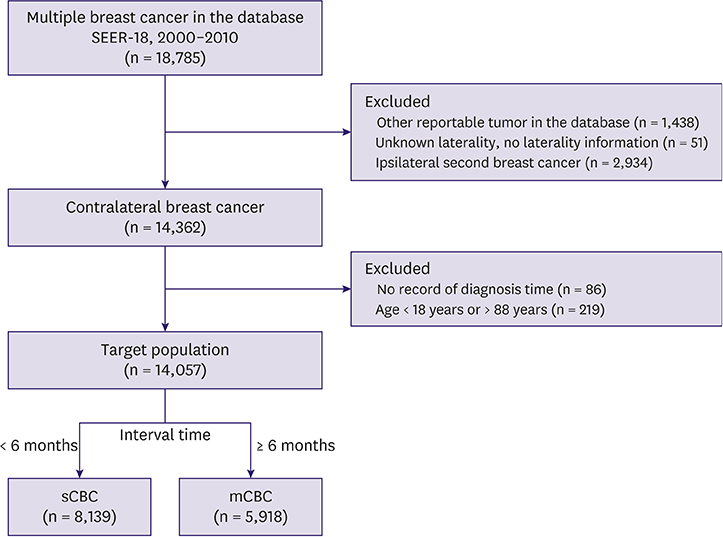
Using the Surveillance, Epidemiology, and End Results Program database, we identified sCBC or mCBC patients from 2000 to 2010. The Kaplan-Meier method and Cox proportional hazards regression analysis were used to analyze overall survival and breast cancer-specific survival (BCSS) rates of sCBCs and mCBCs, respectively.
RESULTS
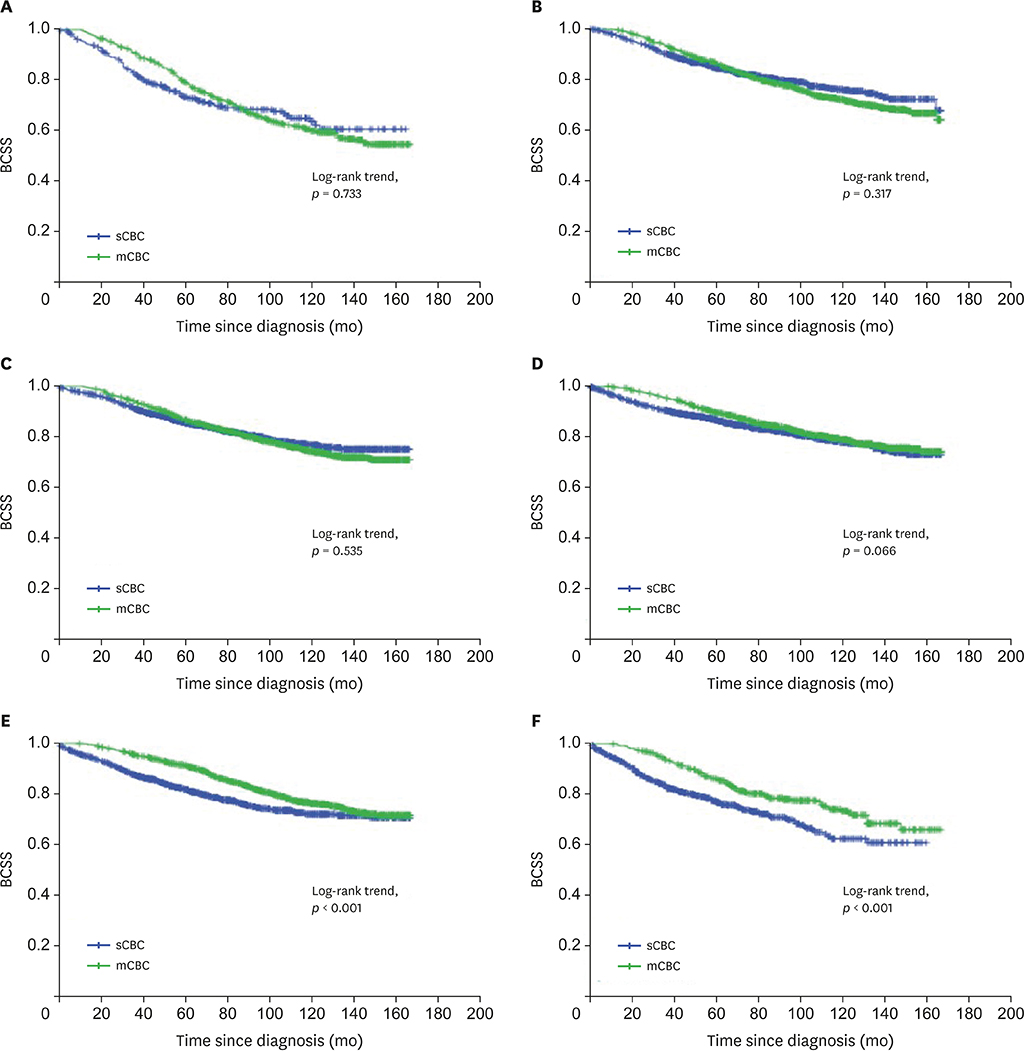
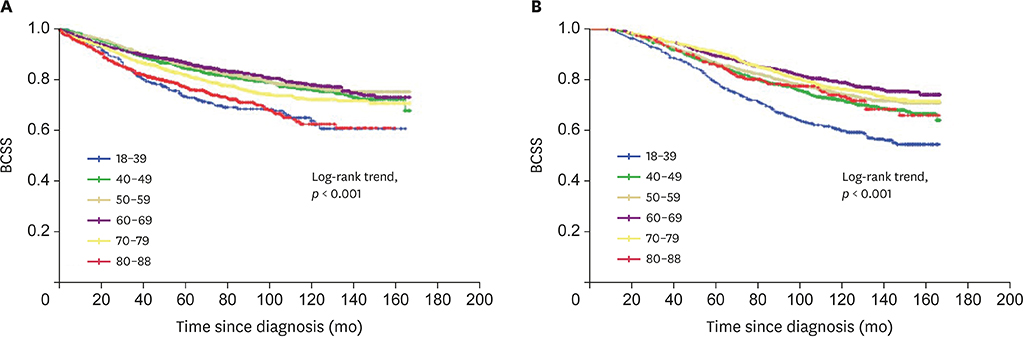
Overall, 14,057 sCBC (n = 8,139, 57.9%) and mCBC (n = 5,918, 42.1%) patients were included. The first tumors of sCBC were more likely to have higher stage and more lymph and distant metastases, whereas those of mCBC were more often infiltrating ductal carcinoma (IDC), had localized stage, were estrogen receptor (ER) and progesterone receptor (PR) negative, and had less axillary nodal involvement. The second tumors of mCBC tended to be IDC and have higher grade, adverse stage, ER and PR-negativity; and more axillary nodal involvement, compared to the second tumors of sCBC. mCBC patients had significantly favorable 5-year BCSS but worse long-term BCSS compared with sCBC patients. Moreover, subgroup analysis revealed no significant difference of BCSS between sCBC and mCBC among patients aged 18-60 years. Multivariate analysis indicated that age, grade, and stage of 2 tumors; surgery for second tumor; and ER status of the second tumor were independent prognostic factors for BCSS of contralateral breast cancer (CBC).
CONCLUSION
The characteristics and outcomes of sCBCs and mCBCs were substantially different. sCBC and mCBC patients may have different prognosis, and the prognosis of CBC depends on the first and second tumors.
Keyword
MeSH Terms
Figure
Reference
-
1. Mezencev R, Švajdler M Jr. Hormone receptor status of contralateral breast cancers: analysis of data from the US SEER population-based registries. Breast Cancer. 2017; 24:400–410.
Article2. Nichols HB, Berrington de González A, Lacey JV Jr, Rosenberg PS, Anderson WF. Declining incidence of contralateral breast cancer in the United States from 1975 to 2006. J Clin Oncol. 2011; 29:1564–1569.
Article3. Basu NN, Barr L, Ross GL, Evans DG. Contralateral risk-reducing mastectomy: review of risk factors and risk-reducing strategies. Int J Surg Oncol. 2015; 2015:901046.
Article4. Reinisch M, Huober J, von Minckwitz G, Blohmer JU, Denkert C, Hanusch C, et al. pCR rates in patients with bilateral breast cancer after neoadjuvant anthracycline-taxane based-chemotherapy - a retrospective pooled analysis of individual patients data of four German neoadjuvant trials. Breast. 2017; 32:73–78.
Article5. Shi YX, Xia Q, Peng RJ, Yuan ZY, Wang SS, An X, et al. Comparison of clinicopathological characteristics and prognoses between bilateral and unilateral breast cancer. J Cancer Res Clin Oncol. 2012; 138:705–714.
Article6. Nichol AM, Yerushalmi R, Tyldesley S, Lesperance M, Bajdik CD, Speers C, et al. A case-match study comparing unilateral with synchronous bilateral breast cancer outcomes. J Clin Oncol. 2011; 29:4763–4768.
Article7. Hartman M, Czene K, Reilly M, Adolfsson J, Bergh J, Adami HO, et al. Incidence and prognosis of synchronous and metachronous bilateral breast cancer. J Clin Oncol. 2007; 25:4210–4216.
Article8. Chen SF, Du CW, Yang P, Zhang HW, Kwan M, Zhang GJ. The molecular and clinicopathologic characteristics of bilateral breast cancer. Sci Rep. 2013; 3:2590.
Article9. Ibrahim NY, Sroor MY, Darwish DO. Impact of bilateral breast cancer on prognosis: synchronous versus metachronous tumors. Asian Pac J Cancer Prev. 2015; 16:1007–1010.
Article10. Londero AP, Bernardi S, Bertozzi S, Angione V, Gentile G, Dri C, et al. Synchronous and metachronous breast malignancies: a cross-sectional retrospective study and review of the literature. BioMed Res Int. 2014; 2014:250727.
Article11. Schmid SM, Pfefferkorn C, Myrick ME, Viehl CT, Obermann E, Schötzau A, et al. Prognosis of early-stage synchronous bilateral invasive breast cancer. Eur J Surg Oncol. 2011; 37:623–628.
Article12. Chowdhury M, Euhus D, Onega T, Biswas S, Choudhary PK. A model for individualized risk prediction of contralateral breast cancer. Breast Cancer Res Treat. 2017; 161:153–160.
Article13. Sandberg ME, Hartman M, Klevebring D, Eloranta S, Ploner A, Hall P, et al. Prognostic implications of estrogen receptor pattern of both tumors in contralateral breast cancer. Breast Cancer Res Treat. 2012; 134:793–800.
Article14. Díaz R, Munárriz B, Santaballa A, Palomar L, Montalar J. Synchronous and metachronous bilateral breast cancer: a long-term single-institution experience. Med Oncol. 2012; 29:16–24.
Article15. Hartman M, Czene K, Reilly M, Bergh J, Lagiou P, Trichopoulos D, et al. Genetic implications of bilateral breast cancer: a population based cohort study. Lancet Oncol. 2005; 6:377–382.
Article16. Pestalozzi BC, Zahrieh D, Mallon E, Gusterson BA, Price KN, Gelber RD, et al. Distinct clinical and prognostic features of infiltrating lobular carcinoma of the breast: combined results of 15 international breast cancer study group clinical trials. J Clin Oncol. 2008; 26:3006–3014.
Article17. Li CI, Uribe DJ, Daling JR. Clinical characteristics of different histologic types of breast cancer. Br J Cancer. 2005; 93:1046–1052.
Article18. Lizarraga IM, Sugg SL, Weigel RJ, Scott-Conner CE. Review of risk factors for the development of contralateral breast cancer. Am J Surg. 2013; 206:704–708.
Article19. Liederbach E, Piro R, Hughes K, Watkin R, Wang CH, Yao K. Clinicopathologic features and time interval analysis of contralateral breast cancers. Surgery. 2015; 158:676–685.
Article20. Zheng YZ, Wang L, Hu X, Shao ZM. Effect of tumor size on breast cancer-specific survival stratified by joint hormone receptor status in a SEER population-based study. Oncotarget. 2015; 6:22985–22995.
Article21. Mellemkjær L, Steding-Jessen M, Frederiksen K, Andersson M, Ejlertsen B, Jensen MB, et al. . Risk of contralateral breast cancer after tamoxifen use among Danish women. Ann Epidemiol. 2014; 24:843–848.
Article22. Aihara T, Tanaka S, Sagara Y, Iwata H, Hozumi Y, Takei H, et al. Incidence of contralateral breast cancer in Japanese patients with unilateral minimum-risk primary breast cancer, and the benefits of endocrine therapy and radiotherapy. Breast Cancer. 2014; 21:284–291.
Article23. Howlader N, Altekruse SF, Li CI, Chen VW, Clarke CA, Ries LA, et al. US incidence of breast cancer subtypes defined by joint hormone receptor and HER2 status. J Natl Cancer Inst. 2014; 106:dju055.
Article24. Kheirelseid EA, Jumustafa H, Miller N, Curran C, Sweeney K, Malone C, et al. Bilateral breast cancer: analysis of incidence, outcome, survival and disease characteristics. Breast Cancer Res Treat. 2011; 126:131–140.
Article25. Jobsen JJ, van der Palen J, Ong F, Riemersma S, Struikmans H. Bilateral breast cancer, synchronous and metachronous; differences and outcome. Breast Cancer Res Treat. 2015; 153:277–283.
Article26. Vichapat V, Garmo H, Holmberg L, Fentiman IS, Tutt A, Gillett C, et al. Prognosis of metachronous contralateral breast cancer: importance of stage, age and interval time between the two diagnoses. Breast Cancer Res Treat. 2011; 130:609–618.
Article27. Beckmann KR, Buckingham J, Craft P, Dahlstrom JE, Zhang Y, Roder D, et al. Clinical characteristics and outcomes of bilateral breast cancer in an Australian cohort. Breast. 2011; 20:158–164.
Article28. Narod SA, Kharazmi E, Fallah M, Sundquist K, Hemminki K. The risk of contralateral breast cancer in daughters of women with and without breast cancer. Clin Genet. 2016; 89:332–335.
Article29. Sandberg ME, Alkner S, Hartman M, Eloranta S, Rydén L, Ploner A, et al. Influence of radiotherapy for the first tumor on aggressiveness of contralateral breast cancer. Int J Cancer. 2013; 132:2388–2394.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical review of bilateral breast cancer
- Comparison of Clinicopathologic Features and Outcomes Between Synchronous and Metachronous Bilateral Breast Cancer
- Ipsilateral Breast Tumor Recurrence with Metachronous Contralateral Axillary Lymph Node Metastasis after Breast-Conserving Surgery with Axillary Lymph Node Dissection
- Bilateral breast carcinoma
- Treatment of Multiple Colorectal Cancers