Clin Endosc.
2019 Mar;52(2):196-200. 10.5946/ce.2018.097.
Endoscopic Ultrasound in the Diagnosis of Pancreatoduodenal Groove Pathology: Report of Three Cases and Brief Review of the Literature
- Affiliations
-
- 1Department of Gastroenterology, Hospital Italiano de Buenos Aires, Buenos Aires, Argentina. Ines.oria@hospitalitaliano.org.ar
- KMID: 2447679
- DOI: http://doi.org/10.5946/ce.2018.097
Abstract
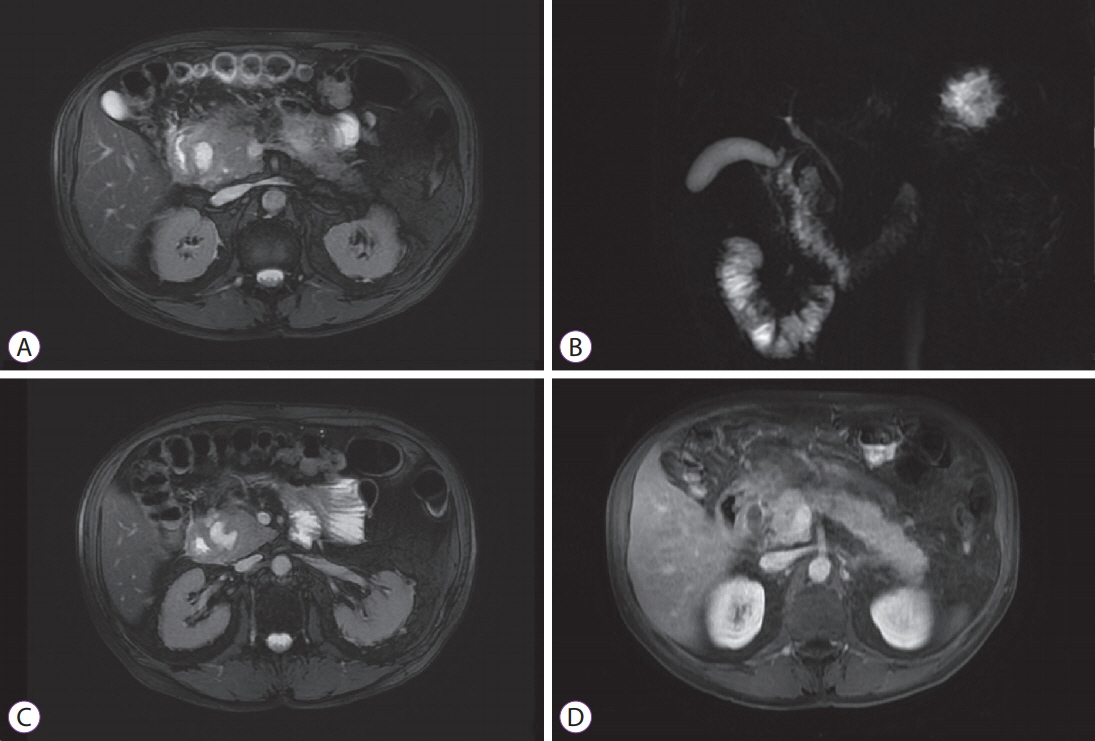
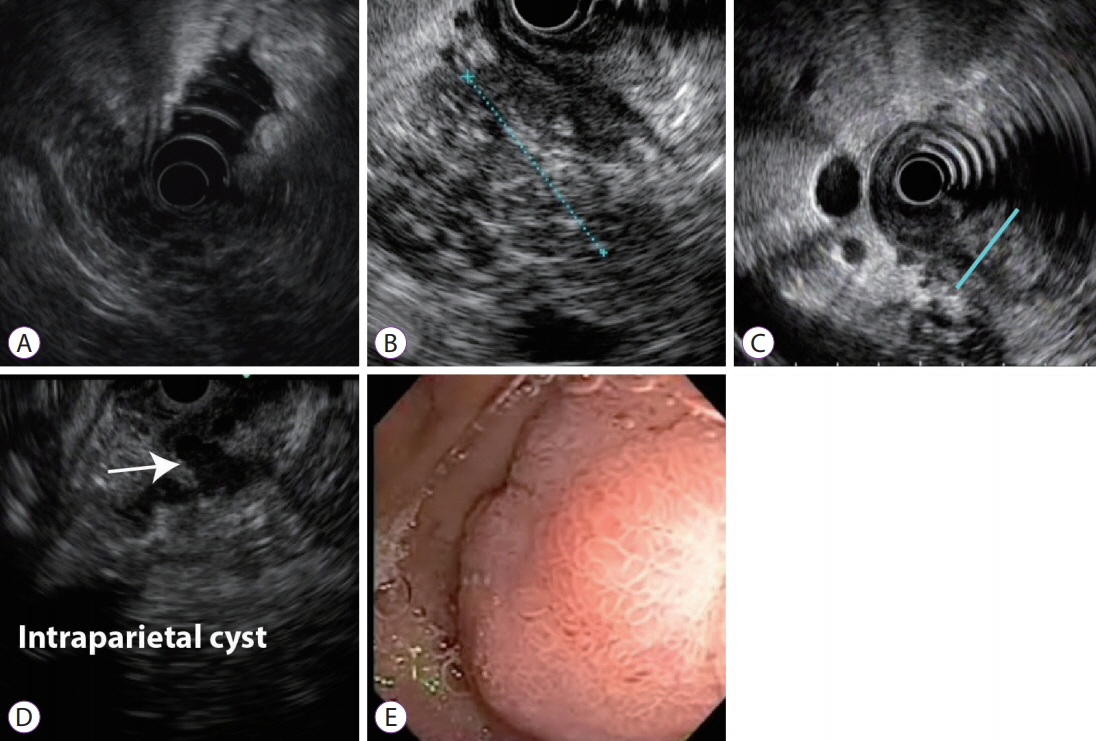
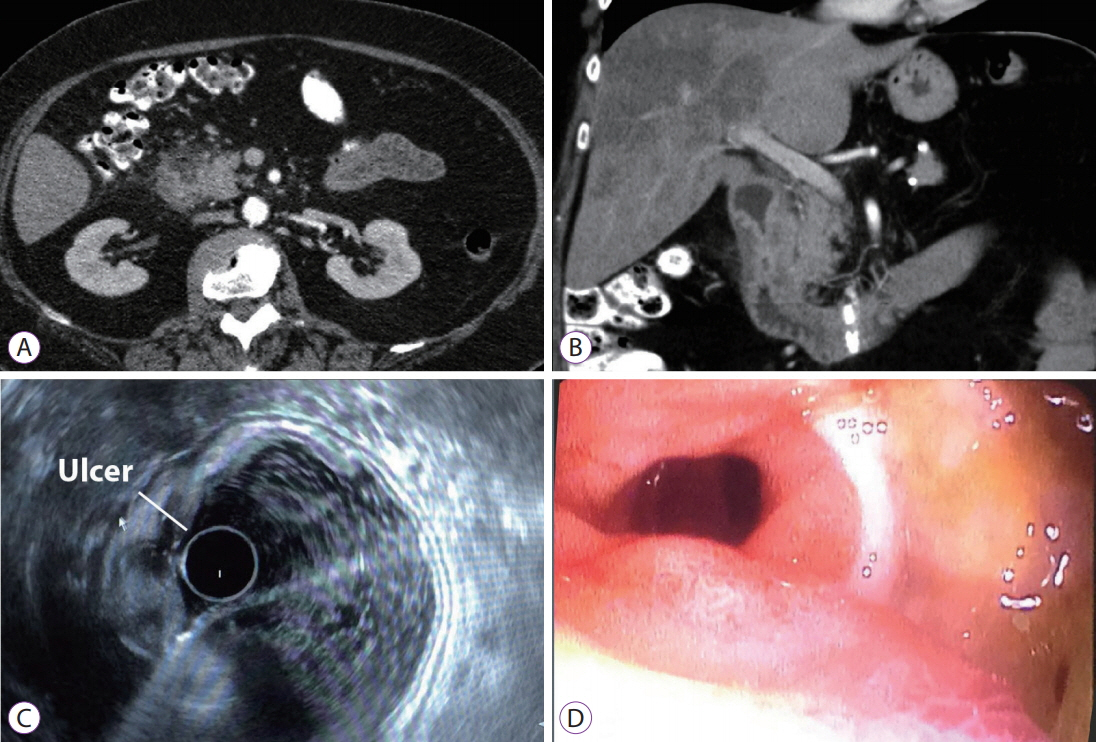
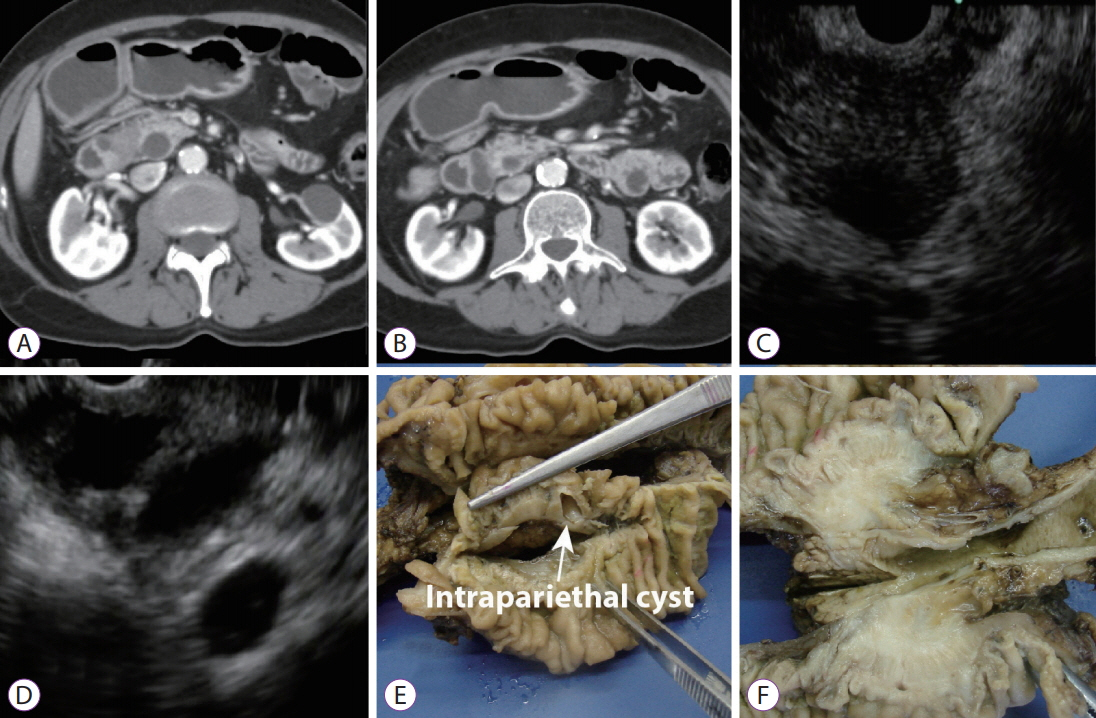
- The pancreatoduodenal groove is a small area where pathologic processes involving the distal bile duct, duodenum, pancreatic head, ampulla of Vater, and retroperitoneum converge. Despite great advances in imaging techniques, a definitive preoperative diagnosis is challenging because of the complex anatomy of this area. Therefore, surgical intervention is frequently required because of the inability to completely exclude malignancy. We report 3 cases of patients with different groove pathologies but similar clinical and imaging presentation, and show the essential role of endoscopic ultrasound (EUS) in making a specific preoperative diagnosis, excluding malignancy in the first case, changing diagnosis in the second case, and confirming malignancy in the third case. EUS was a fundamental tool in this cohort of patients, not only because of its ability to provide superior visualization of a difficult anatomical region, but because of the ability to guide precise, real-time procedures, such as fine-needle aspiration.
Keyword
MeSH Terms
Figure
Reference
-
1. Yu J, Fulcher AS, Turner MA, Halvorsen RA. Normal anatomy and disease processes of the pancreatoduodenal groove: imaging features. AJR Am J Roentgenol. 2004; 183:839–846.
Article2. Hernandez-Jover D, Pernas JC, Gonzalez-Ceballos S, Lupu I, Monill JM, Pérez C. Pancreatoduodenal junction: review of anatomy and pathologic conditions. J Gastrointest Surg. 2011; 15:1269–1281.
Article3. DeSouza K, Nodit L. Groove pancreatitis: a brief review of a diagnostic challenge. Arch Pathol Lab Med. 2015; 139:417–421.
Article4. Raman SP, Salaria SN, Hruban RH, Fishman EK. Groove pancreatitis: spectrum of imaging findings and radiology-pathology correlation. AJR Am J Roentgenol. 2013; 201:W29–W39.
Article5. Goransky J, Alvarez FA, Picco P, Spina JC, Santibañes M, Mazza O. Groove pancreatitis vs groove pancreatic adenocarcinoma. Report of two cases and review of the literature. Acta Gastroenterol Latinoam. 2013; 43:248–253.6. Fusaroli P, Kypraios D, Caletti G, Eloubeidi MA. Pancreatico-biliary endoscopic ultrasound: a systematic review of the levels of evidence, performance and outcomes. World J Gastroenterol. 2012; 18:4243–4256.
Article7. Ray S, Ghatak S, Misra D, et al. Groove pancreatitis: report of three cases with brief review of literature. Indian J Surg. 2017; 79:344–348.
Article8. Vilmann P, Jacobsen GK, Henriksen FW, Hancke S. Endoscopic ultrasonography with guided fine needle aspiration biopsy in pancreatic disease. Gastrointest Endosc. 1992; 38:172–173.
Article9. Shetty D, Bhatnagar G, Sidhu HS, Fox BM, Dodds NI. The increasing role of endoscopic ultrasound (EUS) in the management of pancreatic and biliary disease. Clin Radiol. 2013; 68:323–335.
Article10. ASGE Standards of Practice Committee, Gan SI, Rajan E, et al. Role of EUS. Gastrointest Endosc. 2007; 66:425–434.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Characteristic clinical and pathologic features for preoperative diagnosed groove pancreatitis
- Perineal Groove: A Case Report and Re view of Literature
- A Case of Groove Pancreatitis with a Characteristic Pathologic Feature
- Endoscopic Ultrasound-Guided Fine Needle Aspiration Cytology Diagnosis of Solid Pseudopapillary Neoplasm: Three Case Reports with Review of Literature
- Application of Endoscopic Ultrasound-based Artificial Intelligence in Diagnosis of Pancreatic Malignancies