Asia Pac Allergy.
2015 Apr;5(2):98-102. 10.5415/apallergy.2015.5.2.98.
The association of forced expiratory volume in one second and forced expiratory flow at 50% of the vital capacity, peak expiratory flow parameters, and blood eosinophil counts in exercise-induced bronchospasm in children with mild asthma
- Affiliations
-
- 1Department of Pediatric Allergy, Erciyes University School of Medicine, 38039 Kayseri, Turkey. himmetakar@gmail.com
- KMID: 2397056
- DOI: http://doi.org/10.5415/apallergy.2015.5.2.98
Abstract
- BACKGROUND
Exercise-induced bronchoconstriction (EIB), which describes acute airway narrowing that occurs as a result of exercise, is associated with eosinophilic airway inflammation, bronchial hyperresponsiveness. The forced expiratory volume in one second (FEVâ‚) is the most commonly used spirometric test in the diagnosis of EIB in exercise challenge in asthma. Other parameters such as forced expiratory flow at 50% of the vital capacity (FEF(50%)) and peak expiratory flow (PEF) are used less often in the diagnosis of EIB.
OBJECTIVE
The purpose of this study is to evaluate the association of FEVâ‚ and FEF(50%), PEF parameters, blood eosinophil counts in EIB in children with mild asthma.
METHODS
Sixty-seven children (male: 39, female: 28) with mild asthma were included in this study. Pulmonary functions were assessed before and at 1, 5, 10, 15, and 20 minutes after exercise. The values of spirometric FEVâ‚, FEF50(%), PEF, and blood eosinophil counts were evaluated in EIB in children with mild asthma.
RESULTS
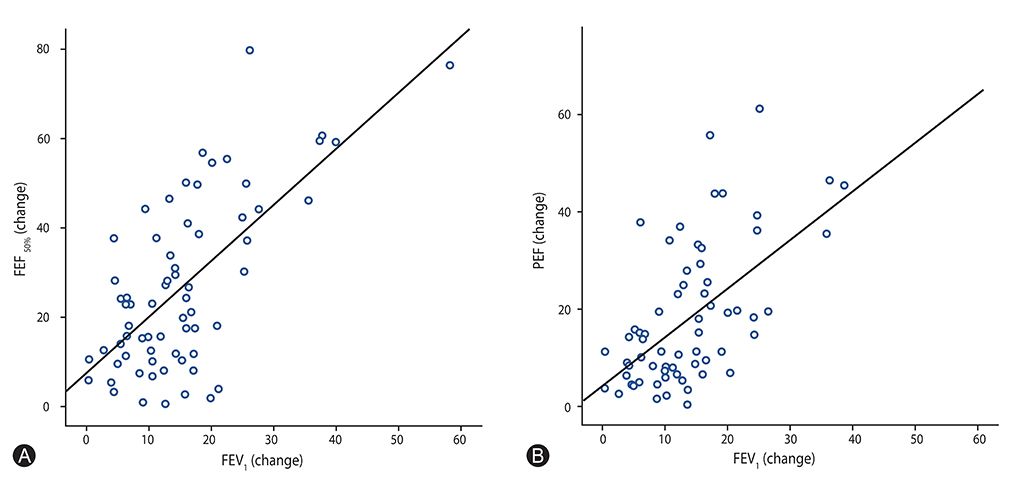
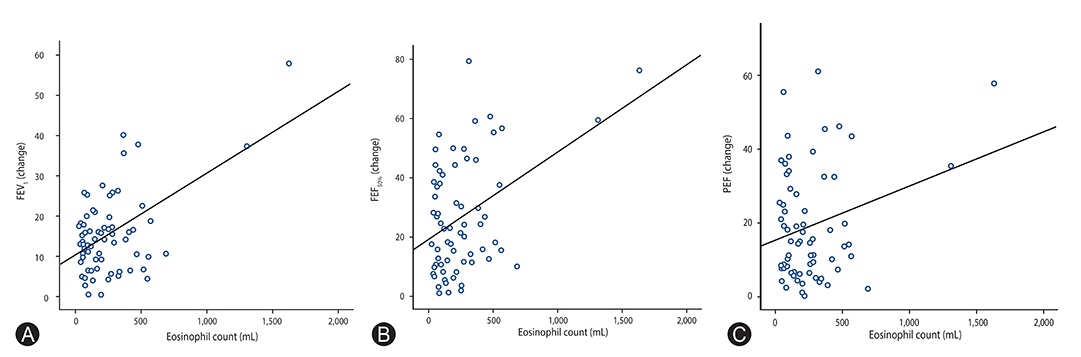
There was a positive correlation between FEVâ‚ with FEF(50%) and PEF values (p < 0.05; FEF(50%), r=0.68; PEF, r=0.65). Also, a positive correlation was found between blood eosinophil counts and the values of spirometric FEVâ‚, FEF(50%), and PEF (p < 0.05; FEVâ‚, r=0.54; FEF(50%), r=0.42; PEF, r=0.26). In addition to these correlations, in the exercise negative group for FEVâ‚, the FEF(50%) and PEF values decreased more than the cutoff values in 3, and 2 patients, respectively.
CONCLUSION
According to the presented study, eosinophil may play a major role in the severity of EIB in mild asthma. FEF(50%) and PEF values can decrease in response to exercise without changes in FEVâ‚ in mild asthmatic patients.
MeSH Terms
Figure
Cited by 1 articles
-
In this issue of Asia Pacific allergy
Constance H. Katelaris
Asia Pac Allergy. 2015;5(2):57-58. doi: 10.5415/apallergy.2015.5.2.57.
Reference
-
1. Parsons JP, Hallstrand TS, Mastronarde JG, Kaminsky DA, Rundell KW, Hull JH, Storms WW, Weiler JM, Cheek FM, Wilson KC, Anderson SD. American Thoracic Society Subcommittee on Exercise-induced Bronchoconstriction. An official American Thoracic Society clinical practice guideline: exercise-induced bronchoconstriction. Am J Respir Crit Care Med. 2013; 187:1016–1027.
Article2. Hallstrand TS, Curtis JR, Koepsell TD, Martin DP, Schoene RB, Sullivan SD, Yorioka GN, Aitken ML. Effectiveness of screening examinations to detect unrecognized exercise-induced bronchoconstriction. J Pediatr. 2002; 141:343–348.
Article3. Becker JM, Rogers J, Rossini G, Mirchandani H, D'Alonzo GE Jr. Asthma deaths during sports: report of a 7-year experience. J Allergy Clin Immunol. 2004; 113:264–267.4. Jones RS, Buston MH, Wharton MJ. The effect of exercise on ventilatory function in the child with asthma. Br J Dis Chest. 1962; 56:78–86.
Article5. Mellis CM, Kattan M, Keens TG, Levison H. Comparative study of histamine and exercise challenges in asthmatic children. Am Rev Respir Dis. 1978; 117:911–915.6. Kawabori I, Pierson WE, Conquest LL, Bierman CW. Incidence of exercise-induced asthma in children. J Allergy Clin Immunol. 1976; 58:447–455.
Article7. Cabral AL, Conceicao GM, Fonseca-Guedes CH, Martins MA. Exercise-induced bronchospasm in children: effects of asthma severity. Am J Respir Crit Care Med. 1999; 159:1819–1823.8. Duong M, Subbarao P, Adelroth E, Obminski G, Strinich T, Inman M, Pedersen S, O'Byrne PM. Sputum eosinophils and the response of exercise-induced bronchoconstriction to corticosteroid in asthma. Chest. 2008; 133:404–411.
Article9. Dickinson JW, Whyte GP, McConnell AK, Nevill AM, Harries MG. Mid-expiratory flow versus FEV1 measurements in the diagnosis of exercise induced asthma in elite athletes. Thorax. 2006; 61:111–114.
Article10. Custovic A, Arifhodzic N, Robinson A, Woodcock A. Exercise testing revisited. The response to exercise in normal and atopic children. Chest. 1994; 105:1127–1132.11. Taylor DR, Bateman ED, Boulet LP, Boushey HA, Busse WW, Casale TB, Chanez P, Enright PL, Gibson PG, de Jongste JC, Kerstjens HA, Lazarus SC, Levy ML, O'Byrne PM, Partridge MR, Pavord ID, Sears MR, Sterk PJ, Stoloff SW, Szefler SJ, Sullivan SD, Thomas MD, Wenzel SE, Reddel HK. A new perspective on concepts of asthma severity and control. Eur Respir J. 2008; 32:545–554.
Article12. Cho HJ, Jung YH, Yang SI, Lee E, Kim HY, Seo JH, Kwon JW, Kim BJ, Kim HB, Lee SY, Song DJ, Kim WK, Jang GC, Shim JY, Hong SJ. Reference values and determinants of fractional concentration of exhaled nitric oxide in healthy children. Allergy Asthma Immunol Res. 2014; 6:169–174.
Article13. Tse SM, Gold DR, Sordillo JE, Hoffman EB, Gillman MW, Rifas-Shiman SL, Fuhlbrigge AL, Tantisira KG, Weiss ST, Litonjua AA. Diagnostic accuracy of the bronchodilator response in children. J Allergy Clin Immunol. 2013; 132:554–559.e5.
Article14. Bar-Yishay E, Amirav I, Goldberg S. Comparison of maximal midexpiratory flow rate and forced expiratory flow at 50% of vital capacity in children. Chest. 2003; 123:731–735.15. Godfrey S, Springer C, Noviski N, Maayan C, Avital A. Exercise but not methacholine differentiates asthma from chronic lung disease in children. Thorax. 1991; 46:488–492.
Article16. Haby MM, Peat JK, Mellis CM, Anderson SD, Woolcock AJ. An exercise challenge for epidemiological studies of childhood asthma: validity and repeatability. Eur Respir J. 1995; 8:729–736.17. Fonseca-Guedes CH, Cabral AL, Martins MA. Exercise-induced bronchospasm in children: comparison of FEV1 and FEF25-75% responses. Pediatr Pulmonol. 2003; 36:49–54.
Article18. Gautrin D, D'Aquino LC, Gagnon G, Malo JL, Cartier A. Comparison between peak expiratory flow rates (PEFR) and FEV1 in the monitoring of asthmatic subjects at an outpatient clinic. Chest. 1994; 106:1419–1426.
Article19. Yoshikawa T, Shoji S, Fujii T, Kanazawa H, Kudoh S, Hirata K, Yoshikawa J. Severity of exercise-induced bronchoconstriction is related to airway eosinophilic inflammation in patients with asthma. Eur Respir J. 1998; 12:879–884.
Article20. Koh YI, Choi S. Blood eosinophil counts for the prediction of the severity of exercise-induced bronchospasm in asthma. Respir Med. 2002; 96:120–125.
Article21. Tahan F, Karaaslan C, Aslan A, Kiper N, Kalayci O. The role of chemokines in exercise-induced bronchoconstriction in asthma. Ann Allergy Asthma Immunol. 2006; 96:819–825.
Article22. Keskin O, Keskin M, Kucukosmanoglu E, Ozkars MY, Gogebakan B, Kul S, Bayram H, Coskun Y. Exhaled RANTES and interleukin 4 levels after exercise challenge in children with asthma. Ann Allergy Asthma Immunol. 2012; 109:303–308.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Ventilatory Dynamics in Hypertensive Heart Disease
- The Relationship of Chronic Obstructive Pulmonary Disease (COPD) to Asthma
- A study of predicted values of peak expiratory flow rates in primary school children
- Partial Flow Volume Curve in Patients with Obstructive Ventilatory Disorder
- Normal Predicted Vlues of Pulmonary Function Test in Children