J Korean Med Assoc.
2011 Apr;54(4):409-418. 10.5124/jkma.2011.54.4.409.
Pharmacotherapy for obesity
- Affiliations
-
- 1Department of Family Medicine, Gachon University of Medicine and Science, Incheon, Korea. zaduplum@gilhospital.com
- KMID: 2190426
- DOI: http://doi.org/10.5124/jkma.2011.54.4.409
Abstract
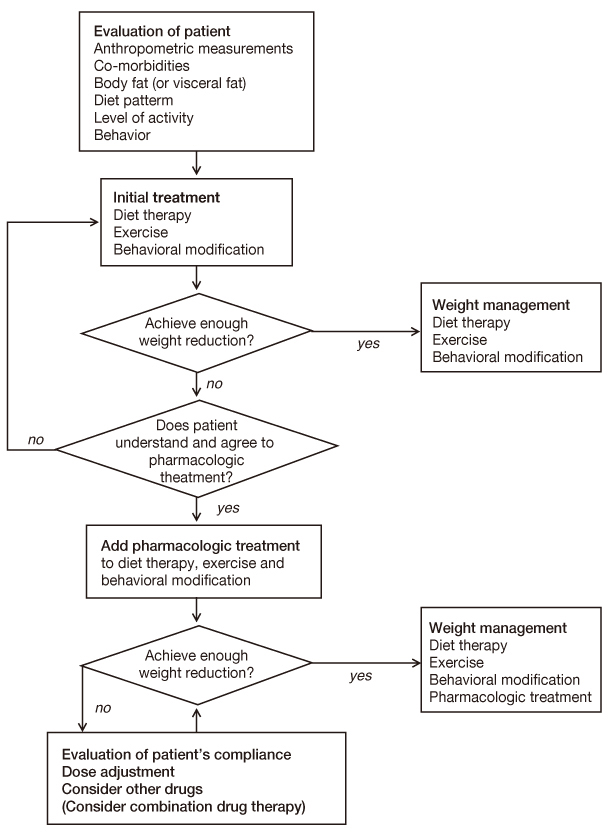
- Due to its serious comorbidities and high prevalence, obesity is one of the heaviest burdens for public health. Although diet, exercise and behavioral modification are the first-line treatment for obesity, their outcomes are not satisfactory. The goal of this article is to review currently available anti-obesity drugs so that physicians may apply the principle of pharmacologic treatment for obesity to obese patients in the real clinical situation. Orlistat, phentermine, diethylpropion, mazindol, and phendimetrazine have been approved as anti-obesity drugs by Korea food and drug administration and administered to patients in Korea. Besides, several non-approved drugs, including fluoxetine, bupropion, topiramate and zonisamide, are being used for weight reduction. Among these drugs, orlistat has been studied most and is the only approved drug for long-term weight management. On the other hand, the rest of the approved drugs lack the evidence of safety issues on the long-term administration. Considering that the non-approved drugs have only a small body of clinical trial results for their efficacy and safety as anti-obesity drugs, it is not appropriate to use them as a first-line therapy in obesity. Because several new medicines and combination therapies are under investigations, more drug therapy options seem to be available in this field in coming years. Although the properly executed pharmacologic treatment is a good option for weight reduction, physicians should recognize that diet, exercise, and behavioral modification are essential to all obese patient and that pharmacologic treatment has several limitations until now.
Keyword
MeSH Terms
-
Anti-Obesity Agents
Bupropion
Comorbidity
Diet
Diethylpropion
Fluoxetine
Fructose
Hand
Humans
Isoxazoles
Korea
Lactones
Mazindol
Morpholines
Obesity
Phentermine
Prevalence
Public Health
United States Food and Drug Administration
Weight Loss
Anti-Obesity Agents
Bupropion
Diethylpropion
Fluoxetine
Fructose
Isoxazoles
Lactones
Mazindol
Morpholines
Phentermine
Figure
Cited by 1 articles
-
A Case of Phendimetrazine Induced-Psychotic Disorder and Dependence
Ji-Ae Yun, Wu-Ri Park, Je-Chun Yu, Kyeong-Sook Choi
J Korean Neuropsychiatr Assoc. 2013;52(5):402-405. doi: 10.4306/jknpa.2013.52.5.402.
Reference
-
1. Lookene A, Skottova N, Olivecrona G. Interactions of lipoprotein lipase with the active-site inhibitor tetrahydrolipstatin (Orlistat). Eur J Biochem. 1994. 222:395–403.
Article2. Zhi J, Melia AT, Eggers H, Joly R, Patel IH. Review of limited systemic absorption of orlistat, a lipase inhibitor, in healthy human volunteers. J Clin Pharmacol. 1995. 35:1103–1108.
Article3. Hartmann D, Hussain Y, Guzelhan C, Odink J. Effect on dietary fat absorption of orlistat, administered at different times relative to meal intake. Br J Clin Pharmacol. 1993. 36:266–270.
Article4. Li Z, Maglione M, Tu W, Mojica W, Arterburn D, Shugarman LR, Hilton L, Suttorp M, Solomon V, Shekelle PG, Morton SC. Meta-analysis: pharmacologic treatment of obesity. Ann Intern Med. 2005. 142:532–546.
Article5. Torgerson JS, Hauptman J, Boldrin MN, Sjöström L. XENical in the prevention of diabetes in obese subjects (XENDOS) study: a randomized study of orlistat as an adjunct to lifestyle changes for the prevention of type 2 diabetes in obese patients. Diabetes Care. 2004. 27:155–161.6. Johansson K, Neovius K, DeSantis SM, Rössner S, Neovius M. Discontinuation due to adverse events in randomized trials of orlistat, sibutramine and rimonabant: a meta-analysis. Obes Rev. 2009. 10:564–575.
Article7. Weber C, Tam YK, Schmidtke-Schrezenmeier G, Jonkmann JH, van Brummelen P. Effect of the lipase inhibitor orlistat on the pharmacokinetics of four different antihypertensive drugs in healthy volunteers. Eur J Clin Pharmacol. 1996. 51:87–90.
Article8. Melia AT, Mulligan TE, Zhi J. Lack of effect of orlistat on the bioavailability of a single dose of nifedipine extended-release tablets (Procardia XL) in healthy volunteers. J Clin Pharmacol. 1996. 36:352–355.
Article9. Hartmann D, Guzelhan C, Zuiderwijk PB, Odink J. Lack of interaction between orlistat and oral contraceptives. Eur J Clin Pharmacol. 1996. 50:421–424.
Article10. Zhi J, Melia AT, Guerciolini R, Koss-Twardy SG, Passe SM, Rakhit A, Sadowski JA. The effect of orlistat on the pharmacokinetics and pharmacodynamics of warfarin in healthy volunteers. J Clin Pharmacol. 1996. 36:659–666.
Article11. Melia AT, Mulligan TE, Zhi J. The effect of orlistat on the pharmacokinetics of phenytoin in healthy volunteers. J Clin Pharmacol. 1996. 36:654–658.
Article12. Zhi J, Melia AT, Koss-Twardy SG, Min B, Guerciolini R, Freundlich NL, Milla G, Patel IH. The influence of orlistat on the pharmacokinetics and pharmacodynamics of glyburide in healthy volunteers. J Clin Pharmacol. 1995. 35:521–525.
Article13. Melia AT, Zhi J, Koss-Twardy SG, Min BH, Smith BL, Freundlich NL, Arora S, Passe SM. The influence of reduced dietary fat absorption induced by orlistat on the pharmacokinetics of digoxin in healthy volunteers. J Clin Pharmacol. 1995. 35:840–843.
Article14. Zhi J, Moore R, Kanitra L, Mulligan TE. Effects of orlistat, a lipase inhibitor, on the pharmacokinetics of three highly lipophilic drugs (amiodarone, fluoxetine, and simvastatin) in healthy volunteers. J Clin Pharmacol. 2003. 43:428–435.
Article15. Ahmad FA, Mahmud S. Acute pancreatitis following orlistat therapy: report of two cases. JOP. 2010. 11:61–63.16. Karamadoukis L, Shivashankar GH, Ludeman L, Williams AJ. An unusual complication of treatment with orlistat. Clin Nephrol. 2009. 71:430–432.
Article17. Kim KK, Cho HJ, Kang HC, Youn BB, Lee KR. Effects on weight reduction and safety of short-term phentermine administration in Korean obese people. Yonsei Med J. 2006. 47:614–625.
Article18. Kang JG, Park CY, Kang JH, Park YW, Park SW. Randomized controlled trial to investigate the effects of a newly developed formulation of phentermine diffuse-controlled release for obesity. Diabetes Obes Metab. 2010. 12:876–882.
Article19. Cercato C, Roizenblatt VA, Leança CC, Segal A, Lopes Filho AP, Mancini MC, Halpern A. A randomized double-blind placebo-controlled study of the long-term efficacy and safety of diethylpropion in the treatment of obese subjects. Int J Obes (Lond). 2009. 33:857–865.
Article20. Hagiwara M, Tsuchida A, Hyakkoku M, Nishizato K, Asai T, Nozawa Y, Tsuchihashi K, Shimamoto K. Delayed onset of pulmonary hypertension associated with an appetite suppressant, mazindol: a case report. Jpn Circ J. 2000. 64:218–221.
Article21. Anderson JW, Greenway FL, Fujioka K, Gadde KM, McKenney J, O'Neil PM. Bupropion SR enhances weight loss: a 48-week double-blind, placebo-controlled trial. Obes Res. 2002. 10:633–641.
Article22. Bray GA, Hollander P, Klein S, Kushner R, Levy B, Fitchet M, Perry BH. A 6-month randomized, placebo-controlled, dose-ranging trial of topiramate for weight loss in obesity. Obes Res. 2003. 11:722–733.
Article23. Wilding J, Van Gaal L, Rissanen A, Vercruysse F, Fitchet M. OBES-002 Study Group. A randomized double-blind placebo-controlled study of the long-term efficacy and safety of topiramate in the treatment of obese subjects. Int J Obes Relat Metab Disord. 2004. 28:1399–1410.
Article24. Gadde KM, Franciscy DM, Wagner HR 2nd, Krishnan KR. Zonisamide for weight loss in obese adults: a randomized controlled trial. JAMA. 2003. 289:1820–1825.25. James WP, Astrup A, Finer N, Hilsted J, Kopelman P, Rössner S, Saris WH, Van Gaal LF. Effect of sibutramine on weight maintenance after weight loss: a randomised trial. STORM Study Group. Sibutramine Trial of Obesity Reduction and Maintenance. Lancet. 2000. 356:2119–2125.
Article26. James WP, Caterson ID, Coutinho W, Finer N, Van Gaal LF, Maggioni AP, Torp-Pedersen C, Sharma AM, Shepherd GM, Rode RA, Renz CL. SCOUT Investigators. Effect of sibutramine on cardiovascular outcomes in overweight and obese subjects. N Engl J Med. 2010. 363:905–917.
Article27. . VIVUS announces positive results from two phase 3 studies: obese patients on qnexa achieve average weight loss up to 14.7% and significant improvements in co-morbidities [Internet]. 2009. cited 2011 Feb 1. Mountain View (CA): VIVUS;Available from: http://ir.vivus.com/releasedetail.cfm?ReleaseID=407933.