Infect Chemother.
2011 Jun;43(3):251-257. 10.3947/ic.2011.43.3.251.
Efficacy of Vancomycin against Staphylococcus aureus according to Inoculum Size in a Neutropenic Mouse Infection Model
- Affiliations
-
- 1Department of Biomedical Science, The Catholic University of Korea, Graduate School, Seoul, Korea.
- 2Department of Pharmacology, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- 3Department of Internal Medicine, College of Medicine, The Catholic University of Korea, Seoul, Korea. symonlee@catholic.ac.kr
- 4Catholic Research Institutes of Medical Science, The Catholic University of Korea, Seoul, Korea.
- KMID: 2170346
- DOI: http://doi.org/10.3947/ic.2011.43.3.251
Abstract
- BACKGROUND
Staphylococcus aureus is one of the most important gram-positive pathogens in many clinical situations. Use of vancomycin against methicillin resistant S aureus (MRSA) has been anecdotally associated with treatment failure, which could be attributable to an inoculum effect (IE). Using a neutropenic mouse thigh infection model, we tried to evaluate the in vivo IE of vancomycin against S. aureus.
MATERIALS AND METHODS
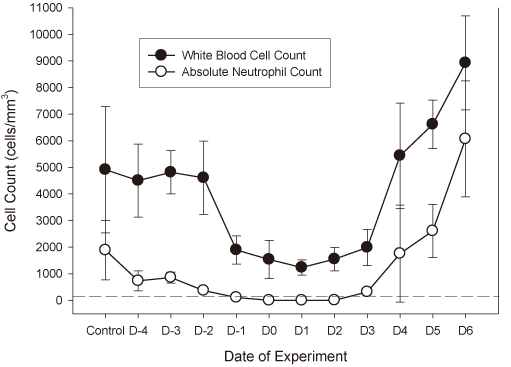
Twenty strains of S aureus were used. Minimum inhibitory concentrations (MICs) were determined by the Clinical and Laboratory Standards Institute guideline. Six-week-old specific-pathogen-free, female CD-1 mice weighing 23-27 grams were used. The neutropenic mice received inoculations of 5.02-5.74 log10 CFU/thigh in one thigh (low inoculum, LI), and 7.22-7.73 log10 CFU/thigh in the other thigh (high inoculum, HI) before therapy. The mice were treated with 6 hourly subcutaneous doses of vancomycin (3.125-100 mg/kg) for 24 h. Single-dose serum pharmacokinetics of vancomycin was determined. Dose-response data were analyzed by an Emax model using non-linear regression. Static doses and area under the curve (AUC)/MIC for bacteriostatic effect at each inoculum were calculated and compared. The ratio of static dose and AUC/MIC between HI and LI (IE index) provided the magnitude of IE for each organism.
RESULTS
Five methicillin-susceptible S aureus (MSSA) strains and 15 MRSA strains were used. Vancomycin MICs of the 20 strains varied by 4-fold (0.5-2 mg/L). The AUC/MIC ratio was the major parameter determining the efficacy of vancomycin against S aureus . Mean (range) static dose on LI and HI was 20.7 (11.8-35.1) and 136.7 (32.1-314), respectively. The mean IE index of static dose between them was 7.39. Mean (range) of AUC/MIC on LI and HI was 27.0 (6.61-66.6) and 152.3 (46.2-344), respectively, which produced a mean IE index of AUC/MIC of 7.47. The IE indices of the MSSA strains were significantly higher than those of the MRSA strains (11.3 vs. 6.1 on static dose [P=0.018], 11.4 vs. 6.2 on AUC/MIC [P=0.034]).
CONCLUSIONS
With a 100-fold inoculum increment of S aureus , at least a 7-fold dose of vancomycin would be required to show the same bacteriostatic effect. Thus, IE as well as MICs is an important parameter in selecting and adjusting a dose and dosage interval along with the resistance profile in the treatment of S. aureus infections. IE to vancomycin observed in the in vivo neutropenic mouse model was more evident for MSSA strains than for MRSA strains.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
The Cefazolin Inoculum Effect and the Presence of type A blaZ Gene according to agr Genotype in Methicillin-Susceptible Staphylococcus aureus Bacteremia
Soon Ok Lee, Shinwon Lee, Sohee Park, Jeong Eun Lee, Sun Hee Lee
Infect Chemother. 2019;51(4):376-385. doi: 10.3947/ic.2019.51.4.376.
Reference
-
1. Wisplinghoff H, Bischoff T, Tallent SM, Seifert H, Wenzel RP, Edmond MB. Nosocomial bloodstream infections in US hospitals: analysis of 24,179 cases from a prospective nationwide surveillance study. Clin Infect Dis. 2004. 39:309–317.
Article2. Weber JT. Community-associated methicillin-resistant Staphylococcus aureus. Clin Infect Dis. 2005. 41:Suppl 4. S269–S272.3. Levine DP. Vancomycin: a history. Clin Infect Dis. 2006. 42:Suppl 1. S5–S12.
Article4. Karchmer AW. Staphylococcus aureus and vancomycin: the sequel. Ann Intern Med. 1991. 115:739–741.5. Fridkin SK, Hageman J, McDougal LK, Mohammed J, Jarvis WR, Perl TM, Tenover FC. Vancomycin-Intermediate Staphylococcus aureus Epidemiology Study Group. Epidemiological and microbiological characterization of infections caused by Staphylococcus aureus with reduced susceptibility to vancomycin, United States, 1997-2001. Clin Infect Dis. 2003. 36:429–439.
Article6. LaPlante KL, Rybak MJ. Impact of high-inoculum Staphylococcus aureus on the activities of nafcillin, vancomycin, linezolid, and daptomycin, alone and in combination with gentamicin, in an in vitro pharmacodynamic model. Antimicrob Agents Chemother. 2004. 48:4665–4672.
Article7. Kim SH, Kim KH, Kim HB, Kim NJ, Kim EC, Oh MD, Choe KW. Outcome of vancomycin treatment in patients with methicillin-susceptible Staphylococcus aureus bacteremia. Antimicrob Agents Chemother. 2008. 52:192–197.
Article8. Stryjewski ME, Szczech LA, Benjamin DK Jr, Inrig JK, Kanafani ZA, Engemann JJ, Chu VH, Joyce MJ, Reller LB, Corey GR, Fowler VG Jr. Use of vancomycin or first-generation cephalosporins for the treatment of hemodialysis-dependent patients with methicillin-susceptible Staphylococcus aureus bacteremia. Clin Infect Dis. 2007. 44:190–196.
Article9. Rybak MJ, Akins RL. Emergence of methicillin-resistant Staphylococcus aureus with intermediate glycopeptide resistance: clinical significance and treatment options. Drugs. 2001. 61:1–7.
Article10. Brook I. Inoculum effect. Rev Infect Dis. 1989. 11:361–368.
Article11. Lamp KC, Rybak MJ, Bailey EM, Kaatz GW. In vitro pharmacodynamic effects of concentration, pH, and growth phase on serum bactericidal activities of daptomycin and vancomycin. Antimicrob Agents Chemother. 1992. 36:2709–2714.
Article12. Mercier RC, Houlihan HH, Rybak MJ. Pharmacodynamic evaluation of a new glycopeptide, LY333328, and in vitro activity against Staphylococcus aureus and Enterococcus faecium. Antimicrob Agents Chemother. 1997. 41:1307–1312.
Article13. Moise PA, North D, Steenbergen JN, Sakoulas G. Susceptibility relationship between vancomycin and daptomycin in Staphylococcus aureus: facts and assumptions. Lancet Infect Dis. 2009. 9:617–624.
Article14. Craig WA. Pharmacokinetic/pharmacodynamic parameters: rationale for antibacterial dosing of mice and men. Clin Infect Dis. 1998. 26:1–10. quiz 11-2.
Article15. Ambrose PG, Bhavnani SM, Rubino CM, Louie A, Gumbo T, Forrest A, Drusano GL. Pharmacokinetics-pharmacodynamics of antimicrobial therapy: it's not just for mice anymore. Clin Infect Dis. 2007. 44:79–86.
Article16. Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing; eighteenth informational supplement. CLSI document M100-S18. 2008. Wayne,PA: CLSI.17. Craig WA, Andes DR. In vivo pharmacodynamics of ceftobiprole against multiple bacterial pathogens in murine thigh and lung infection models. Antimicrob Agents Chemother. 2008. 52:3492–3496.
Article18. Craig WA, Andes DR, Stamstad T. In vivo pharmacodynamics of new lipopeptide MX-2401. Antimicrob Agents Chemother. 2010. 54:5092–5098.
Article19. Stratton CW. In vitro susceptibility testing versus in vivo effectiveness. Med Clin North Am. 2006. 90:1077–1088.20. Woods DD. The relation of p-aminobenzoic acid to the mechanism of the action of sulphanilamide. Br J Exp Pathol. 1940. 21:74–90.21. Stevens DL. The role of vancomycin in the treatment paradigm. Clin Infect Dis. 2006. 42:Suppl 1. S51–S57.
Article22. Nannini EC, Stryjewski ME, Singh KV, Rude TH, Corey GR, Fowler VG Jr, Murray BE. Determination of an inoculum effect with various cephalosporins among clinical isolates of methicillin-susceptible Staphylococcus aureus. Antimicrob Agents Chemother. 2010. 54:2206–2208.
Article23. Gould IM. Is vancomycin redundant for serious staphylococcal infection? Int J Antimicrob Agents. 2010. 36:Suppl 2. S55–S57.
Article24. Rybak MJ, Lomaestro BM, Rotschafer JC, Moellering RC, Craig WA, Billeter M, Dalovisio JR, Levine DP. Vancomycin therapeutic guidelines: a summary of consensus recommendations from the infectious diseases Society of America, the American Society of Health-System Pharmacists, and the Society of Infectious Diseases Pharmacists. Clin Infect Dis. 2009. 49:325–327.
Article25. Nettey H, Haswani D, D'Souza M, Oettinger C. In vitro antimicrobial effect of encapsulated vancomycin on internalized Staphylococcus aureus within endothelial cells. Drug Dev Ind Pharm. 2007. 33:133–139.
Article26. Carryn S, Chanteux H, Seral C, Mingeot-Leclercq MP, Van Bambeke F, Tulkens PM. Intracellular pharmacodynamics of antibiotics. Infect Dis Clin North Am. 2003. 17:615–634.
Article27. Donlan RM, Costerton JW. Biofilms: survival mechanisms of clinically relevant microorganisms. Clin Microbiol Rev. 2002. 15:167–193.
Article28. Conly JM, Johnston BL. VISA, hetero-VISA and VRSA: The end of the vancomycin era? Can J Infect Dis. 2002. 13:282–284.
Article29. Proctor RA, Kahl B, von Eiff C, Vaudaux PE, Lew DP, Peters G. Staphylococcal small colony variants have novel mechanisms for antibiotic resistance. Clin Infect Dis. 1998. 27:Suppl 1. S68–S74.
Article30. Park YH, Lee DG, Chun HS, Park C, Park SH, Choi SM, Lee HK, Park YJ, Kim SY, Hur JA, Choi JH, Yoo JH, Kang JH, Shin WS, Kim CC. Once-daily dosing of arbekacin can suppress the formation of small colony variants of methicillin resistant Staphylococcus aureus in an in vitro pharmacodynamic infection model. Infect Chemother. 2006. 38:154–163.31. Ekdahl C, Hanberger H, Hällgren A, Nilsson M, Svensson E, Nilsson LE. Rapid decrease of free vancomycin in dense staphylococcal cultures. Eur J Clin Microbiol Infect Dis. 2005. 24:596–602.
Article32. Chambers HF. Crossley KB, Archer GL, editors. Parenteral antibiotics for the treatment of bacteremia and other serious staphylococcal infections. The staphylococci in human disease. 1997. New York: Churchill Livingstone, Inc.;583–601.33. Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing; sixteenth informational supplement. CLSI document M100-S15. 2006. Wayne, PA: CLSI.34. Chung G, Cha J, Han S, Jang H, Lee K, Yoo J, Yoo J, Kim H, Eun S, Kim B, Park O, Lee Y. Nationwide surveillance study of vancomycin intermediate Staphylococcus aureus strains in Korean hospitals from 2001 to 2006. J Microbiol Biotechnol. 2010. 20:637–642.35. Howden BP, Ward PB, Charles PG, Korman TM, Fuller A, du Cros P, Grabsch EA, Roberts SA, Robson J, Read K, Bak N, Hurley J, Johnson PD, Morris AJ, Mayall BC, Grayson ML. Treatment outcomes for serious infections caused by methicillin-resistant Staphylococcus aureus with reduced vancomycin susceptibility. Clin Infect Dis. 2004. 38:521–528.
Article