Korean J Hepatobiliary Pancreat Surg.
2013 Nov;17(4):186-190. 10.14701/kjhbps.2013.17.4.186.
Collision tumor of the ampulla of Vater - Coexistence of neuroendocrine carcinoma and adenocarcinoma: report of a case
- Affiliations
-
- 1Department of Surgery, Chonnam National University College of Medicine, Gwangju, Korea. surgihur@naver.com
- 2Department of Radiology, Chonnam National University College of Medicine, Gwangju, Korea.
- 3Department of Pathology, Chonnam National University College of Medicine, Gwangju, Korea.
- KMID: 2083346
- DOI: http://doi.org/10.14701/kjhbps.2013.17.4.186
Abstract
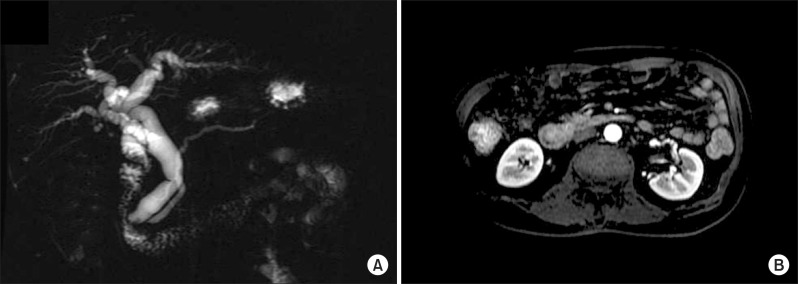
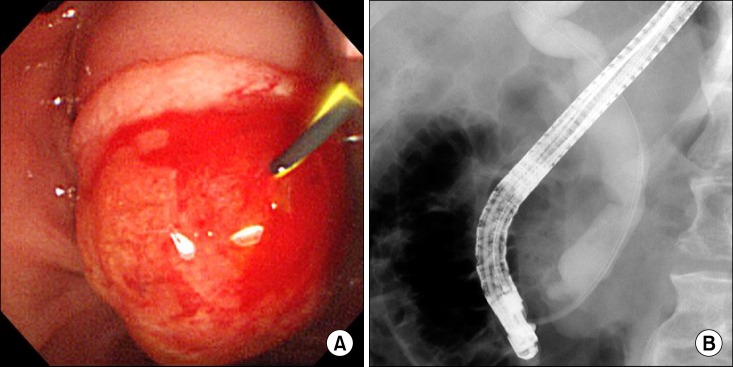
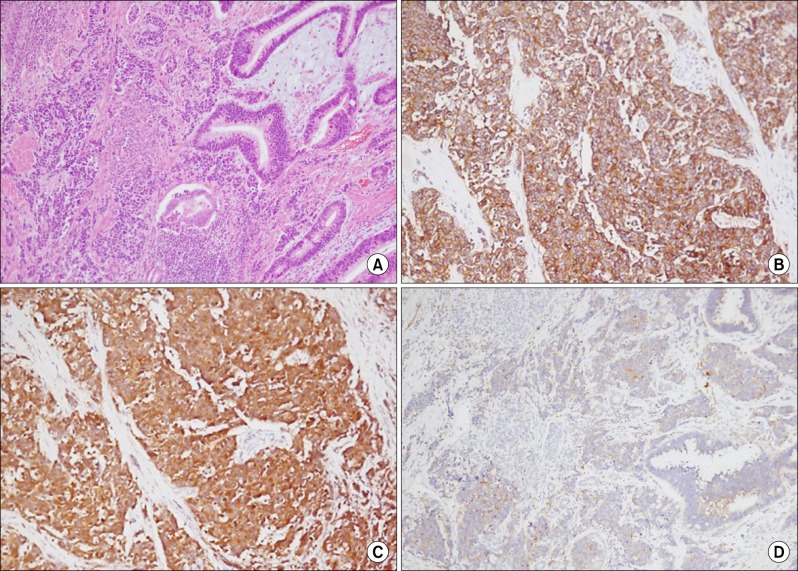
- Herein, we present a case of coexisting neuroendocrine carcinoma and conventional adenocarcinoma (collision tumor) in the ampulla of Vater, which has seldom been reported in the literature. A 51-year-old man presented with a month history of jaundice. MRCP disclosed about 1.9x1.8 cm sized heterogeneously enhancing mass in ampulla of Vater, causing obstructions of distal common bile duct. He underwent pylorus-preserving pancreaticoduodenectomy under the diagnosis on ampulla of Vater cancer. Pathologically, sections on the ampulla of Vater showed conventional ductal adenocarcinoma extended and collided with poorly differentiated neuroendocrine carcinoma. In conclusion, we hereby presented a case of coexisting neuroendocrine carcinoma and conventional adenocarcinoma in the ampulla of Vater.
MeSH Terms
Figure
Reference
-
1. Modlin IM, Lye KD, Kidd M. A 5-decade analysis of 13,715 carcinoid tumors. Cancer. 2003; 97:934–959. PMID: 12569593.
Article2. Sato K, Waseda R, Tatsuzawa Y, et al. Composite large cell neuroendocrine carcinoma and adenocarcinoma of the common bile duct. J Clin Pathol. 2006; 59:105–107. PMID: 16394290.
Article3. Bosman FT. WHO classification of tumours of the digestive system. 4th ed. Lyon: International Agency for Research on Cancer;2010.4. Yao JC, Hassan M, Phan A, et al. One hundred years after "carcinoid": epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J Clin Oncol. 2008; 26:3063–3072. PMID: 18565894.
Article5. Klimstra DS, Modlin IR, Adsay NV, et al. Pathology reporting of neuroendocrine tumors: application of the Delphic consensus process to the development of a minimum pathology data set. Am J Surg Pathol. 2010; 34:300–313. PMID: 20118772.
Article6. Cubilla AL, Fitzgerald PJ. Surgical pathology aspects of cancer of the ampulla-head-of-pancreas region. Monogr Pathol. 1980; 21:67–81. PMID: 6261123.7. Kim HS, Kim HJ, Kim HM, et al. Concurrent occurrence of adenocarcinoma and neuroendocrine type small cell carcinoma in the ampulla of Vater. Korean J Med. 2009; 76:70–73.8. Williams IM, Williams NW, Stock D, et al. Collision tumour of the ampulla of Vater: carcinoid and adenocarcinoma. HPB Surg. 1997; 10:241–244. PMID: 9184878.
Article9. de Leval L, Hardy N, Deprez M, et al. Gastric collision between a papillotubular adenocarcinoma and a gastrinoma in a patient with Zollinger-Ellison syndrome. Virchows Arch. 2002; 441:462–465. PMID: 12447676.
Article10. Komatsu D, Sakurai M, Nakafuji H, et al. Granulocyte colony stimulating factor-producing collision tumor of the gastric cardia. J Gastroenterol. 2003; 38:1013–1015. PMID: 14614613.
Article11. Nishino N, Konno H, Baba S, et al. Synchronous lymphoma and adenocarcinoma occurring as a collision tumor in the stomach: report of a case. Surg Today. 1996; 26:508–512. PMID: 8840432.
Article12. Shah IA, Schlageter MO, Boehm N. Composite carcinoid-adenocarcinoma of ampulla of Vater. Hum Pathol. 1990; 21:1188–1190. PMID: 2227927.
Article13. Wenig BM, Albores-Saavedra J, Buetow PC, et al. Pancreatic mucinous cystic neoplasm with sarcomatous stroma: a report of three cases. Am J Surg Pathol. 1997; 21:70–80. PMID: 8990143.14. Alex WR, Auerbach HE, Pezzi CM. Adenocarcinoid tumor of the ampulla of Vater. Am Surg. 1998; 64:355–359. PMID: 9544149.15. del Vecchio MT, Pergola L, Tripodi SA, et al. Microcystic adenoma associated with a mucinous cystadenocarcinoma: a "collision tumor" of the pancreas. Pancreas. 2002; 24:106–108. PMID: 11741191.
Article16. Moncur JT, Lacy BE, Longnecker DS. Mixed acinar-endocrine carcinoma arising in the ampulla of Vater. Hum Pathol. 2002; 33:449–451. PMID: 12055683.
Article17. Cheng SP, Yang TL, Chang KM, et al. Large cell neuroendocrine carcinoma of the ampulla of Vater with glandular differentiation. J Clin Pathol. 2004; 57:1098–1100. PMID: 15452169.
Article18. Vilardell F, Velasco A, Cuevas D, et al. Composite papillary intestinal-type adenocarcinoma/poorly differentiated neuroendocrine carcinoma of the ampulla of Vater. J Clin Pathol. 2011; 64:174–177. PMID: 21097790.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of a Collision Tumor in the Ampulla of Vater with an Adenocarcinoma and a Large Cell Neuroendocrine Carcinoma
- Concurrent occurrence of adenocarcinoma and neuroendocrine type small cell carcinoma in the ampulla of Vater
- Mixed adenoneuroendocrine carcinoma of the ampulla of Vater: Three case reports and a literature review
- Composite Large Cell Neuroendocrine Carcinoma and Adenocarcinoma of the Ampulla of Vater
- Gastric Collision Tumor (Adenocarcinoma and Neuro-endocrine Carcinoma): A Report of Two Cases