J Korean Med Assoc.
2004 Feb;47(2):127-132. 10.5124/jkma.2004.47.2.127.
Optical Imaging in the Field of Molecular Imaging
- Affiliations
-
- 1Department of Neurology, Seoul National University College of Medicine and Hospital, Korea. rohjk@snu.ac.kr
- 2Center for Molecular Imaging Research, Harvard Medical School, Korea. kdongeog@partners.org
- KMID: 1936466
- DOI: http://doi.org/10.5124/jkma.2004.47.2.127
Abstract
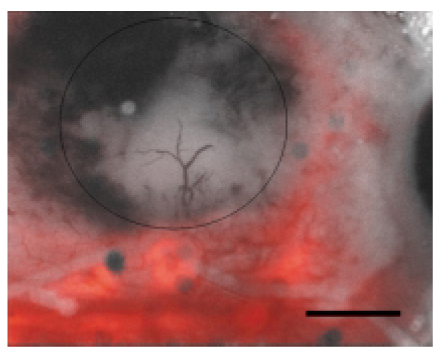
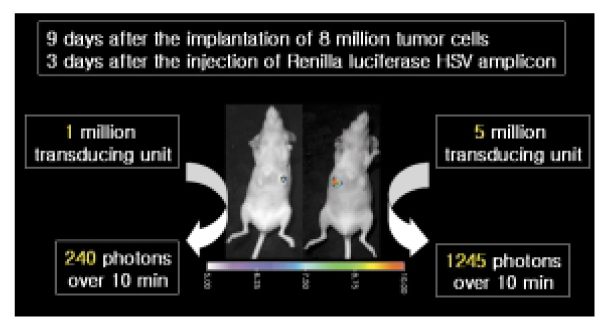
- Molecular imaging is leading an important role in the era of molecular medicine. Optical imaging, a rising star in the filed of molecular imaging, largely consists of fluorescent imaging and bioluminescent imaging. In the fluorescence imaging, an illuminating light excites fluorescent reporters in the living subject, and a charged coupled device (CCD) camera collects an emission light of shifted wavelength. In the bioluminescent imaging, reporter genes code for the luciferase that is responsible for fireflies' glow. After the injection of the substrate iuciferin, animals carrying the luciferase gene are imaged with a supersensitive CCD camera to pick up the small number of photons transmitted through tissues. It has been shown that well aimed and creatively built reporters let researchers explore and answer a lot of biologically important questions in living subjects. Despite its relatively short history, optical imaging is rapidly being implemented in various clinical areas as well as research fields.
Keyword
MeSH Terms
Figure
Reference
-
1. Greer LF 3rd, Szalay AA. Imaging of light emission from the expression of luciferases in living cells and organisms: a review. Luminescence. 2002. 17:43–74.
Article2. Blasberg RG. Plenary session II: Identifying biological targets and pathways for in vivo molecular imaging. First annual meeting of the society for molecular imaging. 2002. USA: Boston.3. Massoud TF, Gambhir SS. Molecular imaging in living subjects: seeing fundamental biological processes in a new light. Genes Dev. 2003. 17:545–580.
Article4. Contag CH, Spilman SD, Contag PR, Oshiro M, Eames B, Benaron DA, et al. Visualizing gene expression in living mammals using a bioluminescent reporter. Photochem Photobiol. 1997. 66:523–531.
Article5. Weissleder R, Mahmood U. Molecular imaging. Radiology. 2001. 219:316–333.
Article6. Lippincott-Schwartz J, Snapp E, Kenworthy A. Studying protein dynamics in living cells. Nat Rev Mol Cell Biol. 2001. 2:444–456.
Article7. Tung CH, Zeng Q, Shah K, Kim DE, Schellingerhout D, Weissleder R. In vivo imaging of beta-galactosidase activity using far red fluorescent switch. Cancer Res. 2004. In press.8. Luker GD, Luker KE, Sharma V, Pica CM, Dahlheimer JL, Piwnica-Worms D, et al. In vitro and in vivo characterization of a dual-function green fluorescent protein-HSV1-thymidine kinase reporter gene driven by the human elongation factor 1 alpha promoter. Mol Imaging. 2002. 1:65–73.
Article9. Josephson L, Kircher MF, Mahmood U, Tang Y, Weissleder R. Near-infrared fluorescent nanoparticles as combined MR/optical imaging probes. Bioconjug Chem. 2002. 13:554–560.
Article10. Weissleder R. A clearer vision for in vivo imaging. Nat Biotechnol. 2001. 19:316–317.
Article11. Moon WK, Lin Y, O'Loughlin T, Tang Y, Kim DE, Weissleder R, Tung CH. Enhanced tumor detection using a folate receptor-targeted near-infrared fluorochrome conjugate. Bioconjug Chem. 2003. 14:539–545.
Article12. Ntziachristos V, Tung CH, Bremer C, Weissleder R. Fluorescence molecular tomography resolves protease activity in vivo. Nat Med. 2002. 8:757–760.
Article13. Ntziachristos V, Chance B. Probing physiology and molecular function using optical imaging:applications to breast cancer. Breast Cancer Res. 2001. 3:41–46.14. Tung CH, Mahmood U, Bredow S, Weissleder R. In vivo imaging of proteolytic enzyme activity using a novel molecular reporter. Cancer Res. 2000. 60:4953–4958.15. Bremer C, Tung CH, Weissleder R. In vivo molecular target assessment of matrix metalloproteinase inhibition. Nat Med. 2001. 7:743–748.
Article16. Kircher MF, Mahmood U, King RS, Weissleder R, Josephson L. A multimodal nanoparticle for preoperative magnetic resonance imaging and intraoperative optical brain tumor delineation. Cancer Res. 2003. 63:8122–8125.17. Detter C, Russ D, Iffland A, Wipper S, Schurr MO, Reichart B, et al. Near-infrared fluorescence coronary angiography: a new noninvasive technology for intraoperative graft patency control. Heart Surg Forum. 2002. 5:364–369.18. Shah K, Tang Y, Breakefield X, Weissleder R. Real-time imaging of TRAIL-induced apoptosis of glioma tumors in vivo. Oncogene. 2003. 22:6865–6872.
Article19. Kim DE, Ishii K, Shah K, Weissleder R, Schellingerhout D. Imaging of stem cell recruitment to ischemic infarcts in a murine model. Stroke. 2004. in press.
Article20. Shah K, Bureau E, Tang Y, Kim DE, Weissleder R, Breakefield X. Induction of apoptosis in glioma cells by novel TRAIL-secreting neural precursor cells (NPC) and in vivo tracking of NPC migration and tumor apoptosis. 33rd annual meeting of society for neuroscience. 2003. New Orleans, USA. Abstract.21. Fujimoto JG, Bouma B, Tearney GJ, Boppart SA, Pitris C, Brezinski ME, et al. New technology for high-speed and high-resolution optical coherence tomography. Ann N Y Acad Sci. 1998. 838:95–107.
Article22. Watanabe E, Yamashita Y, Maki A, Ito Y, Koizumi H. Non-invasive functional mapping with multi-channel near infrared spectroscopic topography in humans. Neurosci Lett. 1996. 205:41–44.
Article23. Kato H, Izumiyama M, Koizumi H, Takahashi A, Itoyama Y. Near-infrared spectroscopic topography as a tool to monitor motor reorganization after hemiparetic stroke: a comparison with functional MRI. Stroke. 2002. 33:2032–2036.
Article