Blood Res.
2021 Jun;56(2):65-71. 10.5045/br.2021.2021028.
Clinical impact of cell-free serum Epstein–Barr virus status in patients with newly diagnosed malignant lymphoma
- Affiliations
-
- 1Departments of Hematology/Oncology, Kyungpook National University Hospital, School of Medicine, Kyungpook National University, Daegu, Korea
- 2Departments of Laboratory Medicine, Kyungpook National University Hospital, School of Medicine, Kyungpook National University, Daegu, Korea
- 3Department of Hematology/Oncology, Chonnam National University Hwasun Hospital, School of Medicine, Chonnam National University, Hwasun, Korea
- KMID: 2517002
- DOI: http://doi.org/10.5045/br.2021.2021028
Abstract
- Background
We analyzed cell-free serum Epstein‒Barr virus (EBV) DNA to identify its prognostic role in patients with newly diagnosed lymphoma.
Methods
We retrospectively reviewed patients diagnosed with lymphoma between January 2014 and July 2020. Patients were enrolled according to the following criteria: i) pathologically confirmed lymphomas according to the World Health Organization criteria, ii) age over 18 years, iii) serum EBV DNA measurement using polymerase chain reaction prior to first-line therapy, and iv) receipt of curative standard chemotherapy. In total, 263 patients met these criteria and were included in this study.
Results
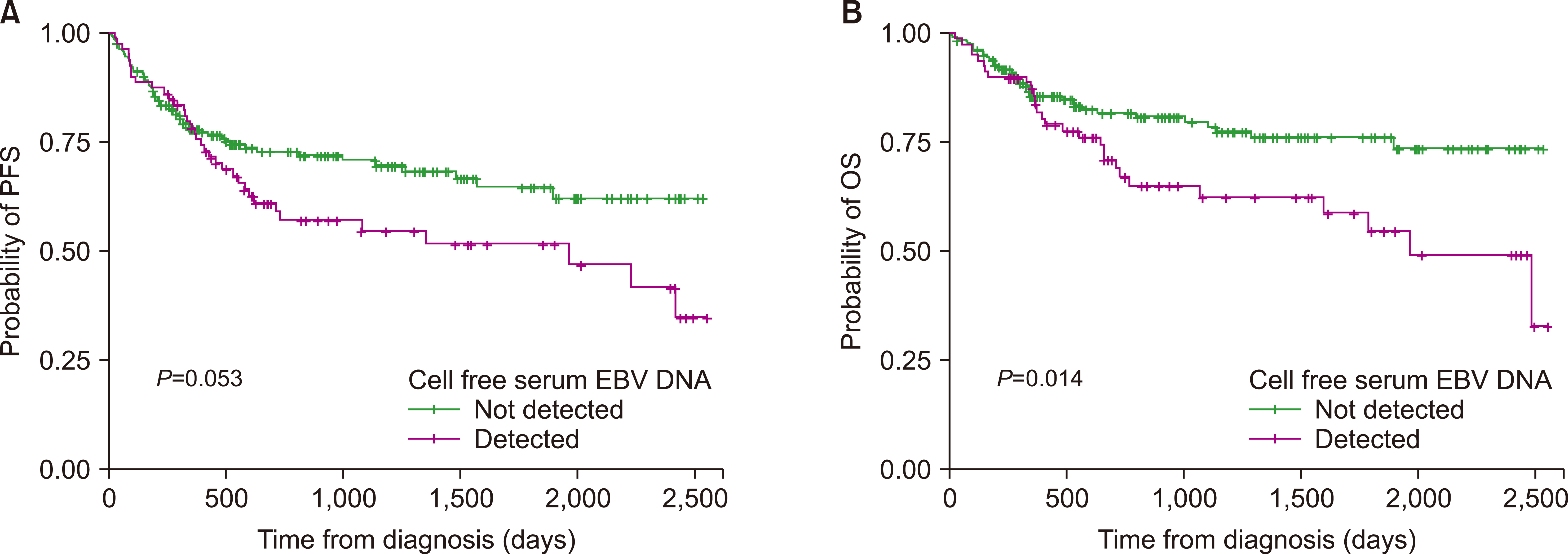
Serum EBV DNA was detected in 79 patients (30.0%). Patients with positive serum EBV tended to be older (P =0.090), and the proportion of T-cell lineage lymphomas was higher than that of B-cell lymphomas (P =0.003). EBV positivity was significantly associated with more advanced disease based on the Ann Arbor staging system (P =0.008) and the International Prognostic Index (P =0.009). EBV positivity was also associated with higher disease relapse (P =0.038) and death rates (P =0.005). EBV-positive lymphomas further showed inferior long-term survival outcomes in terms of progression-free survival (PFS) (P =0.053) and overall survival (OS) (P =0.014). In the subgroup analyses, serum EBV positivity was a significant prognostic factor for patients with B-cell lineage lymphomas in terms of PFS (P =0.003) and OS (P =0.033).
Conclusion
We demonstrated that cell-free serum EBV DNA status at the time of diagnosis has potential as a prognostic biomarker for patients with newly diagnosed malignant lymphomas.
Keyword
Figure
Reference
-
1. Odumade OA, Hogquist KA, Balfour HH Jr. 2011; Progress and problems in understanding and managing primary Epstein-Barr virus infections. Clin Microbiol Rev. 24:193–209. DOI: 10.1128/CMR.00044-10. PMID: 21233512. PMCID: PMC3021204.
Article2. Grywalska E, Rolinski J. 2015; Epstein-Barr virus-associated lymphomas. Semin Oncol. 42:291–303. DOI: 10.1053/j.seminoncol.2014.12.030. PMID: 25843733.
Article3. Delecluse HJ, Feederle R, O'Sullivan B, Taniere P. 2007; Epstein Barr virus-associated tumours: an update for the attention of the working pathologist. J Clin Pathol. 60:1358–64. DOI: 10.1136/jcp.2006.044586. PMID: 17873116. PMCID: PMC2095566.
Article4. Cancer Genome Atlas Research Network. 2014; Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 513:202–9. DOI: 10.1038/nature13480. PMID: 25079317. PMCID: PMC4170219.5. Jarrett RF, Stark GL, White J, et al. 2005; Impact of tumor Epstein-Barr virus status on presenting features and outcome in age-defined subgroups of patients with classic Hodgkin lymphoma: a population-based study. Blood. 106:2444–51. DOI: 10.1182/blood-2004-09-3759. PMID: 15941916.
Article6. Park S, Lee J, Ko YH, et al. 2007; The impact of Epstein-Barr virus status on clinical outcome in diffuse large B-cell lymphoma. Blood. 110:972–8. DOI: 10.1182/blood-2007-01-067769. PMID: 17400912.
Article7. Wang ZY, Liu QF, Wang H, et al. 2012; Clinical implications of plasma Epstein-Barr virus DNA in early-stage extranodal nasal-type NK/T-cell lymphoma patients receiving primary radiotherapy. Blood. 120:2003–10. DOI: 10.1182/blood-2012-06-435024. PMID: 22826562.
Article8. Kanakry JA, Li H, Gellert LL, et al. 2013; Plasma Epstein-Barr virus DNA predicts outcome in advanced Hodgkin lymphoma: correlative analysis from a large North American cooperative group trial. Blood. 121:3547–53. DOI: 10.1182/blood-2012-09-454694. PMID: 23386127. PMCID: PMC3643756.
Article9. Swerdlow SH, Campo E, Harris NL, editors. 2008. WHO classification of tumours of haematopoietic and lymphoid tissues. 4th ed. IARC Press;Lyon, France:10. Cheson BD, Fisher RI, Barrington SF, et al. 2014; Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol. 32:3059–68. DOI: 10.1200/JCO.2013.54.8800. PMID: 25113753. PMCID: PMC4979083.
Article11. Shannon-Lowe C, Rickinson AB, Bell AI. 2017; Epstein-Barr virus-associated lymphomas. Philos Trans R Soc Lond B Biol Sci. 372:20160271. DOI: 10.1098/rstb.2016.0271. PMID: 28893938. PMCID: PMC5597738.
Article12. Castillo JJ, Beltran BE, Miranda RN, Young KH, Chavez JC, Sotomayor EM. 2016; EBV-positive diffuse large B-cell lymphoma of the elderly: 2016 update on diagnosis, risk-stratification, and management. Am J Hematol. 91:529–37. DOI: 10.1002/ajh.24370. PMID: 27093913.
Article13. Haverkos BM, Huang Y, Gru A, et al. 2017; Frequency and clinical correlates of elevated plasma Epstein-Barr virus DNA at diagnosis in peripheral T-cell lymphomas. Int J Cancer. 140:1899–906. DOI: 10.1002/ijc.30566. PMID: 27943278. PMCID: PMC5323329.
Article14. Lu TX, Liang JH, Miao Y, et al. 2015; Epstein-Barr virus positive diffuse large B-cell lymphoma predict poor outcome, regardless of the age. Sci Rep. 5:12168. DOI: 10.1038/srep12168. PMID: 26202875. PMCID: PMC4511873.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of Epstein-Barr Virus-Positive Diffuse Large B-Cell Lymphoma Occurring in Thyroid Gland
- A Case of Epstein-Barr Virus-positive Diffuse, Large B-cell Lymphoma after Angioimmunoblastic T-cell Lymphoma
- Histopathologic Analysis of Malignant Lymphoma Involving the Skin and Its Relationship with the Epstein-Barr Virus
- Epstein-Barr Virus-Associated Vesiculopapular Eruption on the Face of a Patient with Natural Killer T Cell Lymphoma
- A Case of Epstein-Barr Virus-associated Primary Gastric T-cell Lymphoma with Rapidly Progressive Endoscopic Features and Clinical Course