Cancer Res Treat.
2020 Jul;52(3):730-738. 10.4143/crt.2019.598.
Clinical Outcomes of Immune Checkpoint Blocker Therapy for Malignant Melanoma in Korean Patients: Potential Clinical Implications for a Combination Strategy Involving Radiotherapy
- Affiliations
-
- 1Department of Radiation Oncology, Yonsei University College of Medicine, Seoul, Korea
- 2Department of Radiation Oncology, Inha University Hospital, Incheon, Korea
- 3Department of Dermatology, Yonsei University College of Medicine, Seoul, Korea
- 4Division of Medical Oncology, Department of Internal Medicine, Yonsei University College of Medicine, Seoul, Korea
- KMID: 2504455
- DOI: http://doi.org/10.4143/crt.2019.598
Abstract
- Purpose
We investigated the clinical efficacy of immune checkpoint blocker (ICB) therapy for metastatic or advanced melanoma in Korean patients. As well, we assessed whether the effects of ICBs can be enhanced by combination therapy with palliative radiotherapy (RT).
Materials and Methods
We retrospectively reviewed the records of 127 patients with metastatic melanoma who received ICB with or without palliative RT between 2014 and 2018. The melanoma subtypes were classified as follows: chronic sun-damaged (CSD), acral, mucosal, and uveal. The primary endpoint was the objective response rate (ORR).
Results
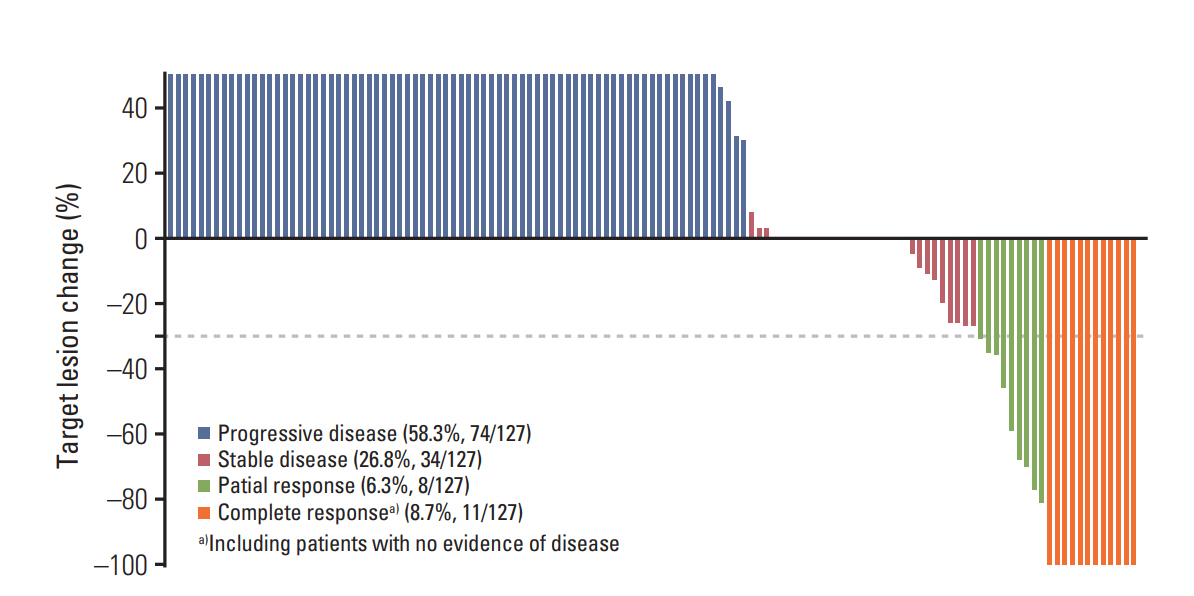
The overall ORR was 15%, with 11 complete and eight partial responses. ORRs for CSD, acral/mucosal, and uveal melanomas were 50%, 16.5%, and 0%, respectively (p=0.009). In addition to the subtype, stage at treatment, total tumor burden at treatment, and ICB type were significantly associated with ORR (all p < 0.05). Palliative RT was administered in 44% of patients during the treatment, and it did not affect ORR. Clinical responders to ICB therapy exhibited significantly higher 1-year progression-free and overall survival rates than nonresponders.
Conclusion
ORR for ICB monotherapy in Korean patients with melanoma is relatively modest compared with that in Western patients because the non-CSD subtypes are predominant in the Korean population. Our findings regarding combination therapy with ICB provided a rationale for the initiation of our phase II study (NCT04017897).
Keyword
Figure
Reference
-
References
1. Postow MA, Chesney J, Pavlick AC, Robert C, Grossmann K, McDermott D, et al. Nivolumab and ipilimumab versus ipilimumab in untreated melanoma. N Engl J Med. 2015; 372:2006–17.
Article2. Hodi FS, Chesney J, Pavlick AC, Robert C, Grossmann KF, McDermott DF, et al. Combined nivolumab and ipilimumab versus ipilimumab alone in patients with advanced melanoma: 2-year overall survival outcomes in a multicentre, randomised, controlled, phase 2 trial. Lancet Oncol. 2016; 17:1558–68.
Article3. Bradford PT, Goldstein AM, McMaster ML, Tucker MA. Acral lentiginous melanoma: incidence and survival patterns in the United States, 1986-2005. Arch Dermatol. 2009; 145:427–34.4. Curtin JA, Fridlyand J, Kageshita T, Patel HN, Busam KJ, Kutzner H, et al. Distinct sets of genetic alterations in melanoma. N Engl J Med. 2005; 353:2135–47.
Article5. Shoushtari AN, Munhoz RR, Kuk D, Ott PA, Johnson DB, Tsai KK, et al. The efficacy of anti-PD-1 agents in acral and mucosal melanoma. Cancer. 2016; 122:3354–62.
Article6. Hayward NK, Wilmott JS, Waddell N, Johansson PA, Field MA, Nones K, et al. Whole-genome landscapes of major melanoma subtypes. Nature. 2017; 545:175–80.
Article7. Ribas A, Wolchok JD. Cancer immunotherapy using checkpoint blockade. Science. 2018; 359:1350–5.
Article8. Wei Y, Du Q, Jiang X, Li L, Li T, Li M, et al. Efficacy and safety of combination immunotherapy for malignant solid tumors: a systematic review and meta-analysis. Crit Rev Oncol Hematol. 2019; 138:178–89.
Article9. Pfirschke C, Engblom C, Rickelt S, Cortez-Retamozo V, Garris C, Pucci F, et al. Immunogenic chemotherapy sensitizes tumors to checkpoint blockade therapy. Immunity. 2016; 44:343–54.
Article10. Fukumura D, Kloepper J, Amoozgar Z, Duda DG, Jain RK. Enhancing cancer immunotherapy using antiangiogenics: opportunities and challenges. Nat Rev Clin Oncol. 2018; 15:325–40.
Article11. Demaria S, Golden EB, Formenti SC. Role of local radiation therapy in cancer immunotherapy. JAMA Oncol. 2015; 1:1325–32.
Article12. Vanpouille-Box C, Alard A, Aryankalayil MJ, Sarfraz Y, Diamond JM, Schneider RJ, et al. DNA exonuclease Trex1 regulates radiotherapy-induced tumour immunogenicity. Nat Commun. 2017; 8:15618.
Article13. Shargel L, Wu-Pong S, Yu AB. Applied biopharmaceutics and pharmacokinetics. 7th ed. New York: McGraw-Hill Education;2015.14. Seymour L, Bogaerts J, Perrone A, Ford R, Schwartz LH, Mandrekar S. iRECIST: guidelines for response criteria for use in trials testing immunotherapeutics. Lancet Oncol. 2017; 18:e143–52.
Article15. Somarouthu B, Lee SI, Urban T, Sadow CA, Harris GJ, Kambadakone A. Immune-related tumour response assessment criteria: a comprehensive review. Br J Radiol. 2018; 91:20170457.
Article16. Tas F, Erturk K. Acral lentiginous melanoma is associated with certain poor prognostic histopathological factors but may not be correlated with nodal involvement, recurrence, and a worse survival. Pathobiology. 2018; 85:227–31.
Article17. Bello DM, Chou JF, Panageas KS, Brady MS, Coit DG, Carvajal RD, et al. Prognosis of acral melanoma: a series of 281 patients. Ann Surg Oncol. 2013; 20:3618–25.
Article18. Roh MR, Kim J, Chung KY. Treatment and outcomes of melanoma in acral location in Korean patients. Yonsei Med J. 2010; 51:562–8.
Article19. Maeda T, Yoshino K, Nagai K, Oaku S, Kato M, Hiura A, et al. Efficacy of nivolumab monotherapy against acral lentiginous melanoma and mucosal melanoma in Asian patients. Br J Dermatol. 2019; 180:1230–1.
Article20. Reits EA, Hodge JW, Herberts CA, Groothuis TA, Chakraborty M, Wansley EK, et al. Radiation modulates the peptide repertoire, enhances MHC class I expression, and induces successful antitumor immunotherapy. J Exp Med. 2006; 203:1259–71.
Article21. Lhuillier C, Rudqvist NP, Elemento O, Formenti SC, Demaria S. Radiation therapy and anti-tumor immunity: exposing immunogenic mutations to the immune system. Genome Med. 2019; 11:40.
Article22. Twyman-Saint Victor C, Rech AJ, Maity A, Rengan R, Pauken KE, Stelekati E, et al. Radiation and dual checkpoint blockade activate non-redundant immune mechanisms in cancer. Nature. 2015; 520:373–7.
Article23. Hiniker SM, Reddy SA, Maecker HT, Subrahmanyam PB, Rosenberg-Hasson Y, Swetter SM, et al. A prospective clinical trial combining radiation therapy with systemic immunotherapy in metastatic melanoma. Int J Radiat Oncol Biol Phys. 2016; 96:578–88.
Article24. Koller KM, Mackley HB, Liu J, Wagner H, Talamo G, Schell TD, et al. Improved survival and complete response rates in patients with advanced melanoma treated with concurrent ipilimumab and radiotherapy versus ipilimumab alone. Cancer Biol Ther. 2017; 18:36–42.
Article25. Mowery YM, Patel K, Chowdhary M, Rushing CN, Roy Choudhury K, Lowe JR, et al. Retrospective analysis of safety and efficacy of anti-PD-1 therapy and radiation therapy in advanced melanoma: a bi-institutional study. Radiother Oncol. 2019; 138:114–20.
Article26. Antonia SJ, Villegas A, Daniel D, Vicente D, Murakami S, Hui R, et al. Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. N Engl J Med. 2018; 379:2342–50.
Article27. McGee HM, Daly ME, Azghadi S, Stewart SL, Oesterich L, Schlom J, et al. Stereotactic ablative radiation therapy induces systemic differences in peripheral blood immunophenotype dependent on irradiated site. Int J Radiat Oncol Biol Phys. 2018; 101:1259–70.
Article28. Couty E, Vallard A, Sotton S, Ouni S, Garcia MA, Espenel S, et al. Safety assessment of anticancer drugs in association with radiotherapy in metastatic malignant melanoma: a real-life report: radiation/systemic drug combo in metastatic melanoma. Cancer Chemother Pharmacol. 2019; 83:881–92.29. Amin NP, Zainib M, Parker SM, Agarwal M, Mattes MD. Multi-institutional report on toxicities of concurrent nivolumab and radiation therapy. Adv Radiat Oncol. 2018; 3:399–404.
Article30. ClinicalTrials.gov. The combination of anti-PD-1 with radiotherapy in previously untreated metastatic melanoma [Internet]. Bethesda, MD: U.S. National Library of Medicine;2019. [cited 2019 Aug 19]. Available from: https://clinicaltrials.gov/ct2/show/NCT04017897?cond=NCT04017897&rank=1.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Radiotherapy and immune checkpoint blockades: a snapshot in 2016
- A case of malignant melanoma of the urinary badder
- Therapeutic Strategies for Drug-resistant Melanoma and Their Clinical Implications
- A Case of Mondor's Disease Developed during Radiotherapy after Resection of Malignant Melanoma
- Immune Checkpoint Inhibitor with or without Radiotherapy in Melanoma Patients with Brain Metastases: A Systematic Review and Meta-Analysis