J Clin Neurol.
2020 Jan;16(1):102-107. 10.3988/jcn.2020.16.1.102.
Intraoperative Motor Evoked Potential Improvement in Cervical Spondylotic Myelopathy: Comparison of Cortical Stimulation Parameters
- Affiliations
-
- 1Department of Neurology, National Neuroscience Institute, Singapore General Hospital, Singapore. lo.yew.long@singhealth.com.sg
- 2Department of Neurology, Duke-NUS Medical School, College Road, Singapore.
- 3Department of Orthopedic Surgery, Singapore General Hospital, Singapore.
- KMID: 2467791
- DOI: http://doi.org/10.3988/jcn.2020.16.1.102
Abstract
- BACKGROUND AND PURPOSE
Intraoperative monitoring of the motor pathways is a routine procedure for ensuring the integrity of descending motor tracts during spinal surgery. Intraoperative motor evoked potential improvement (MEPI) may be associated with a better postsurgical outcome in cervical spondylotic myelopathy (CSM). To compare the efficacy of two cortical stimulation parameters in eliciting MEPI intraoperatively during CSM surgery.
METHODS
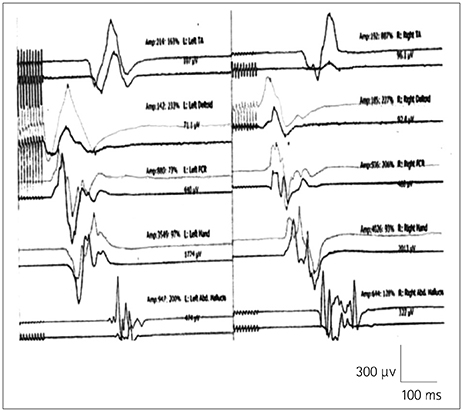
We studied 69 patients who underwent decompression surgery for CSM over a 9-month period using either 5 (Group 1) or 9 (Group 2) stimuli. MEPI was defined as the increase in the amplitude of MEPs from baseline at the end of CSM surgery just prior to skin closure.
RESULTS
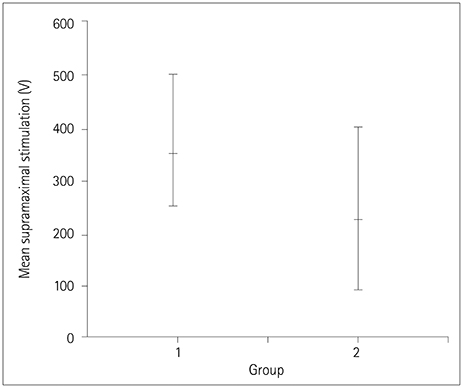
An MEPI of 100% from baseline was observed in 10 patients (53%) in Group 1 and 36 patients (72%) in Group 2. Comparisons of the baseline mean MEP amplitudes of muscles bilaterally between Groups 1 and 2 did not reveal any significant differences. Supramaximal stimulation showed that a significantly higher mean intensity was required for Group 1 than for Group 2.
CONCLUSIONS
MEPI is observed in a much larger proportion of cervical decompression surgery cases than previously thought. Intraoperative MEPI with longer-train cortical stimulation may reflect adequacy of decompression and provide additional guidance for the surgical procedure.
Keyword
MeSH Terms
Figure
Reference
-
1. Montgomery DM, Brower RS. Cervical spondylotic myelopathy. Clinical syndrome and natural history. Orthop Clin North Am. 1992; 23:487–493.2. Hochman M, Tuli S. Cervical spondylotic myelopathy: a review. Internet J Neurol. 2005; 4:24–42.
Article3. Dong Y, Holly LT, Albistegui-Dubois R, Yan X, Marehbian J, Newton JM, et al. Compensatory cerebral adaptations before and evolving changes after surgical decompression in cervical spondylotic myelopathy. J Neurosurg Spine. 2008; 9:538–551.
Article4. Holly LT, Dong Y, Albistegui-DuBois R, Marehbian J, Dobkin B. Cortical reorganization in patients with cervical spondylotic myelopathy. J Neurosurg Spine. 2007; 6:544–551.
Article5. Green A, Cheong PW, Fook-Chong S, Tiruchelvarayan R, Guo CM, Yue WM, et al. Cortical reorganization is associated with surgical decompression of cervical spondylotic myelopathy. Neural Plast. 2015; 2015:389531.
Article6. Lo YL, Chan LL, Lim W, Tan SB, Tan CT, Chen JL, et al. Systematic correlation of transcranial magnetic stimulation and magnetic resonance imaging in cervical spondylotic myelopathy. Spine (Phila Pa 1976). 2004; 29:1137–1145.
Article7. Lo YL, Chan LL, Lim W, Tan SB, Tan CT, Chen JL, et al. Transcranial magnetic stimulation screening for cord compression in cervical spondylosis. J Neurol Sci. 2006; 244:17–21.
Article8. Lo YL, Dan YF, Tan YE, Fook-Chong S, Tan SB, Tan CT, et al. Intraoperative monitoring study of ipsilateral motor evoked potentials in scoliosis surgery. Eur Spine J. 2006; 15:Suppl 5. 656–660.
Article9. Lo YL, Dan YF, Teo A, Tan YE, Yue WM, Raman S, et al. The value of bilateral ipsilateral and contralateral motor evoked potential monitoring in scoliosis surgery. Eur Spine J. 2008; 17:Suppl 2. S236–S238.
Article10. Lo YL, Teo A, Tan YE, Fook-Chong S, Guo CM, Yue WM, et al. Motor and somatosensory abnormalities are significant etiological factors for adolescent idiopathic scoliosis. J Neurol Sci. 2015; 359:117–123.
Article11. Wang S, Tian Y, Wang C, Lu X, Zhuang Q, Peng H, et al. Prognostic value of intraoperative MEP signal improvement during surgical treatment of cervical compressive myelopathy. Eur Spine J. 2016; 25:1875–1880.
Article12. Park MK, Lee SJ, Kim SB, Lee KW, Lee HJ, Han EY, et al. The effect of positive changes during intraoperative monitoring of the functional improvement in patients with cervical compressive myelopathy. Clin Interv Aging. 2018; 13:1211–1218.
Article13. Lo Y, Dan Y, Tan Y, Teo A, Tan S, Yue W, et al. Clinical and physiological effects of transcranial electrical stimulation position on motor evoked potentials in scoliosis surgery. Scoliosis. 2010; 5:3.
Article14. Lo YL, Dan YF, Tan YE, Nurjannah S, Tan SB, Tan CT, et al. Intraoperative motor-evoked potential monitoring in scoliosis surgery: comparison of desflurane/nitrous oxide with propofol total intravenous anesthetic regimens. J Neurosurg Anesthesiol. 2006; 18:211–214.
Article15. Burke D, Hicks RG. Surgical monitoring of motor pathways. J Clin Neurophysiol. 1998; 15:194–205.
Article16. Deletis V, Sala F. Intraoperative neurophysiological monitoring of the spinal cord during spinal cord and spine surgery: a review focus on the corticospinal tracts. Clin Neurophysiol. 2008; 119:248–264.
Article17. Haghighi SS, Gaines RW. Repetitive vs. single transcranial electrical stimulation for intraoperative monitoring of motor conduction in spine surgery. Mo Med. 2003; 100:262–265.18. Journée HL, Polak HE, De Kleuver M, Langeloo DD, Postma AA. Improved neuromonitoring during spinal surgery using double-train transcranial electrical stimulation. Med Biol Eng Comput. 2004; 42:110–113.
Article19. Fouyas IP, Statham PF, Sandercock PA. Cochrane review on the role of surgery in cervical spondylotic radiculomyelopathy. Spine (Phila Pa 1976). 2002; 27:736–747.
Article20. Wilson JR, Tetreault LA, Kim J, Shamji MF, Harrop JS, Mroz T, et al. State of the art in degenerative cervical myelopathy: an update on current clinical evidence. Neurosurgery. 2017; 80:S33–S45.
Article21. Gerling MC, Radcliff K, Isaacs R, Bianco K, Jalai CM, Worley NJ, et al. Two-year results of the prospective spine treatment outcomes study: an analysis of complication rates, predictors of their development, and effect on patient derived outcomes at 2 years for surgical management of cervical spondylotic myelopathy. World Neurosurg. 2017; 106:247–253.
Article22. Ryan K, Goncalves S, Bartha R, Duggal N. Motor network recovery in patients with chronic spinal cord compression: a longitudinal study following decompression surgery. J Neurosurg Spine. 2018; 28:379–388.
Article23. Kreitz T, Huang R, Beck D, Park AG, Hilibrand A. Prolonged preoperative weakness affects recovery of motor function after anterior cervical discectomy and fusion. J Am Acad Orthop Surg. 2018; 26:67–73.
Article24. Bhagavatula ID, Shukla D, Sadashiva N, Saligoudar P, Prasad C, Bhat DI. Functional cortical reorganization in cases of cervical spondylotic myelopathy and changes associated with surgery. Neurosurg Focus. 2016; 40:E2.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Motor Evoked Potential Study with Magnetic Stimulation In Ischemic Stroke Patients
- Comparison of Motor Evoked Potential Using Round Coil with That Using Butterfly Coil
- The Effect of Lorazepam on the Silent Period of the Motor Evoked Potential
- Limitation of Intraoperative Transcranial Electrical Stimulation-Motor Evoked Potential Monitoring During Brain Tumor Resection Adjacent to the Primary Motor Cortex
- Cervical Spondylotic Myelopathy: Natural Course and the Value of Diagnostic Techniques: WFNS Spine Committee Recommendations