Yonsei Med J.
2019 Nov;60(11):1103-1107. 10.3349/ymj.2019.60.11.1103.
Invasive Pneumococcal Disease Caused by Non-Vaccine Type Multidrug-Resistant Streptococcus pneumoniae Transmitted by Close Contact in a Healthy Adult
- Affiliations
-
- 1Department of Internal Medicine, Jeju National University School of Medicine, Jeju, Korea. neosangtaek@naver.com
- 2Department of Laboratory Medicine, Jeju National University School of Medicine, Jeju, Korea.
- 3Department of Microbiology and Immunology, Jeju National University School of Medicine, Jeju, Korea. yomust7@gmail.com
- KMID: 2460228
- DOI: http://doi.org/10.3349/ymj.2019.60.11.1103
Abstract
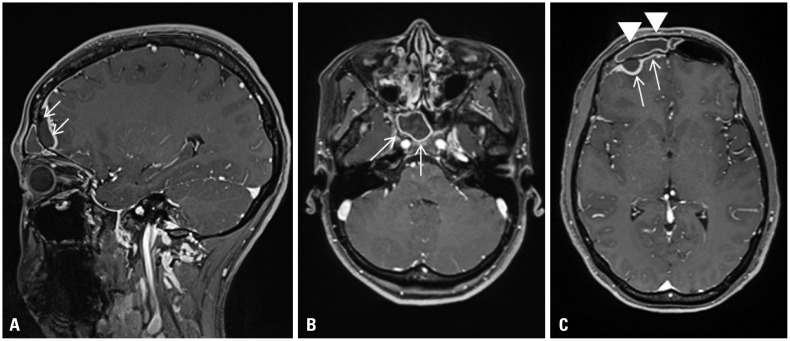
- The incidence of vaccine-type Streptococcus pneumoniae carriage and disease have declined in vaccinated children as well as in unvaccinated children and adults. However, diseases caused by non-vaccine type (NVT) S. pneumoniae are increasing. In this study, we report an invasive pneumococcal disease (IPD) caused by NVT multidrug-resistant (MDR) S. pneumoniae transmitted from a vaccinated infant to an unvaccinated healthy woman, and the clinical characteristics of this serotype. A 29-year-old previously healthy woman visited our hospital with fever and headache. She had been breastfeeding her baby for 8 months. She was diagnosed with brain abscess and sinusitis caused by S. pneumoniae. Although the patient had no previous exposure to antibiotics, antibiotic susceptibility test identified the pathogen as MDR. The patient's family members were examined using nasopharyngeal swabs for bacterial culture. The serotype of S. pneumoniae identified from the blood, abscess, and sputum of the patient was 15B/C. After investing the patient's family members, we found that the serotype from nasopharyngeal specimen of her baby was the same. We described an invasive MDR pneumococcal disease in an immunocompetent young adult in the community. IPD likely spread to the patient by close contact with her baby, who harbored S. pneumoniae of NVT. The spread of NVT S. pneumoniae in the post-vaccine era has increased in the community, and resistance pattern for S. pneumoniae of 15B/C changed compared to the pre-pneumococcal conjugate vaccine era. The spread of MDR pathogens causing IPD among family members should be monitored.
Keyword
MeSH Terms
Figure
Reference
-
1. Walker CLF, Rudan I, Liu L, Nair H, Theodoratou E, Bhutta ZA, et al. Global burden of childhood pneumonia and diarrhoea. Lancet. 2013; 381:1405–1416. PMID: 23582727.
Article2. Wessels MR. Streptococcal infections. In : Kasper DL, Fauci AS, Hauser SL, Longo DL, Loscalzo J, editors. Harrison's principles of internal medicine. 19th ed. New York: McGraw-Hill Education;2015. p. 963–970.3. Lee JK, Yun KW, Choi EH, Kim SJ, Lee SY, Lee HJ. Changes in the serotype distribution among antibiotic resistant carriage Streptococcus pneumoniae isolates in children after the introduction of the extended-valency pneumococcal conjugate vaccine. J Korean Med Sci. 2017; 32:1431–1439. PMID: 28776337.
Article4. Bonten MJ, Huijts SM, Bolkenbaas M, Webber C, Patterson S, Gault S, et al. Polysaccharide conjugate vaccine against pneumococcal pneumonia in adults. N Engl J Med. 2015; 372:1114–1125. PMID: 25785969.
Article5. Choi WS, Choi JH, Kwon KT, Seo K, Kim MA, Lee SO, et al. Revised adult immunization guideline recommended by the Korean society of infectious diseases, 2014. Infect Chemother. 2015; 47:68–79. PMID: 25844267.
Article6. Slotved HC, Dalby T, Hoffmann S. The effect of pneumococcal conjugate vaccines on the incidence of invasive pneumococcal disease caused by ten non-vaccine serotypes in Denmark. Vaccine. 2016; 34:769–774. PMID: 26772630.
Article7. Clinical and Laboratory Standards Institute (CLSI). Performance standards for antimicrobial susceptibility testing. CLSI supplement M100. 27th ed. Wayne (PA): CLSI;2017.8. Leung MH, Bryson K, Freystatter K, Pichon B, Edwards G, Charalambous BM, et al. Sequetyping: serotyping Streptococcus pneumoniae by a single PCR sequencing strategy. J Clin Microbiol. 2012; 50:2419–2427. PMID: 22553238.
Article9. Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol. 2013; 30:2725–2729. PMID: 24132122.
Article10. Givon-Lavi N, Fraser D, Porat N, Dagan R. Spread of Streptococcus pneumoniae and antibiotic-resistant S. pneumoniae from day-care center attendees to their younger siblings. J Infect Dis. 2002; 186:1608–1614. PMID: 12447737.11. Kim YK, LaFon D, Nahm MH. Indirect effects of pneumococcal conjugate vaccines in national immunization programs for children on adult pneumococcal disease. Infect Chemother. 2016; 48:257–266. PMID: 28032483.
Article12. Zuccotti G, Mameli C, Daprai L, Garlaschi ML, Dilillo D, Bedogni G, et al. Serotype distribution and antimicrobial susceptibilities of nasopharyngeal isolates of Streptococcus pneumoniae from healthy children in the 13-valent pneumococcal conjugate vaccine era. Vaccine. 2014; 32:527–534. PMID: 24342249.
Article13. Yoo JR, Heo ST, Oh H, Oh S, Kim YR, Lee KH. Changes in serotype of Streptococcus pneumoniae after the introduction of the 13-valent pneumococcal vaccine in a homogenous population on Jeju island. Infect Chemother. 2019; 51:67–72. PMID: 30941941.
Article14. Balsells E, Guillot L, Nair H, Kyaw MH. Serotype distribution of Streptococcus pneumoniae causing invasive disease in children in the post-PCV era: a systematic review and meta-analysis. PLoS One. 2017; 12:e0177113. PMID: 28486544.
Article15. Singleton RJ, Hennessy TW, Bulkow LR, Hammitt LL, Zulz T, Hurlburt DA, et al. Invasive pneumococcal disease caused by nonvaccine serotypes among Alaska native children with high levels of 7-valent pneumococcal conjugate vaccine coverage. JAMA. 2007; 297:1784–1792. PMID: 17456820.
Article16. Dube FS, Ramjith J, Gardner-Lubbe S, Nduru P, Robberts FJL, Wolter N, et al. Longitudinal characterization of nasopharyngeal colonization with Streptococcus pneumoniae in a South African birth cohort post 13-valent pneumococcal conjugate vaccine implementation. Sci Rep. 2018; 8:12497. PMID: 30131607.
Article17. Baek JY, Kim SH, Kang CI, Chung DR, Peck KR, Song JH, et al. Emergence of an extensively drug-resistant (XDR) Streptococcus pneumoniae serotype 15A by capsular switching. Int J Med Microbiol. 2018; 308:986–989. PMID: 30143394.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Invasive Pneumococcal Diseases in Korean Adults After the Introduction of Pneumococcal Vaccine into the National Immunization Program
- Indirect Effects of Pneumococcal Conjugate Vaccines in National Immunization Programs for Children on Adult Pneumococcal Disease
- Direct and Indirect Effects of Pneumococcal Protein Conjugate Vaccine
- Efficacy and effectiveness of pneumococcal conjugate vaccine in children
- Pneumococcal vaccine