Ann Surg Treat Res.
2019 Jul;97(1):27-35. 10.4174/astr.2019.97.1.27.
Urinary transglutaminase 2 as a potent biomarker to predict interstitial fibrosis and tubular atrophy of kidney allograft during early posttransplant period in deceased donor kidney transplantation
- Affiliations
-
- 1Division of Kidney and Pancreas Transplantation, Department of Surgery, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. sshin@amc.seoul.kr
- 2Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- 3Department of Clinical Epidemiology and Biostatistics, Asan Medical Center, Seoul, Korea.
- KMID: 2450987
- DOI: http://doi.org/10.4174/astr.2019.97.1.27
Abstract
- PURPOSE
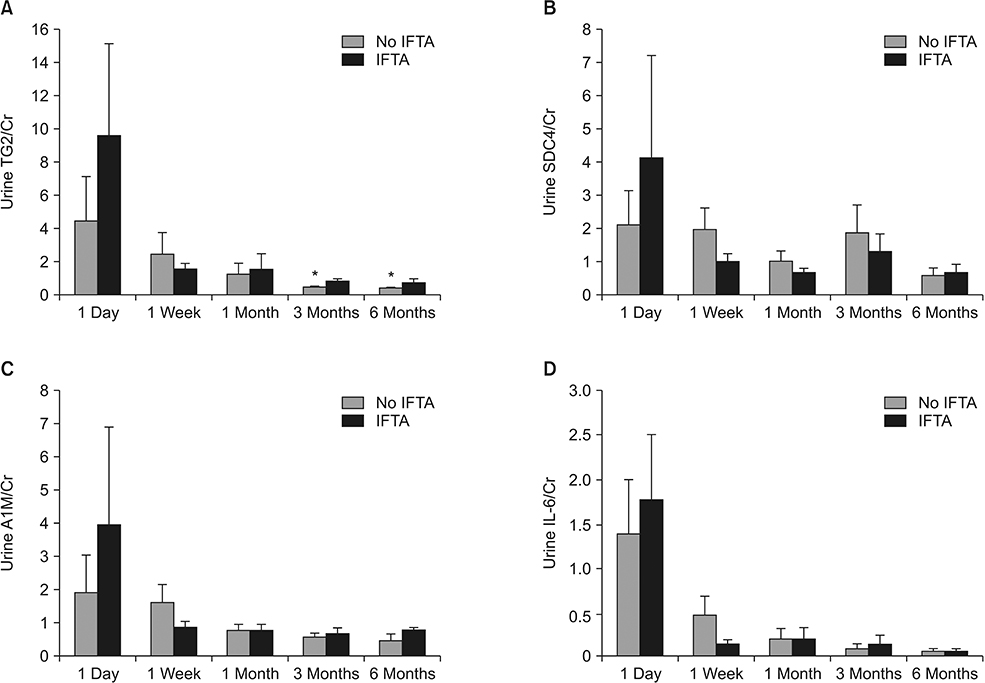
Transglutaminase type 2 (TG2) is an extracellular matrix crosslinking enzyme with a pivotal role in kidney fibrosis. We tested whether quantification of urinary TG2 may represent a noninvasive method to estimate the severity of kidney allograft fibrosis.
METHODS
We prospectively collected urine specimens from 18 deceased donor kidney transplant recipients at 1-day, 7-day, 1-month, 3-month, and 6-month posttransplant. In addition, kidney allograft tissue specimens at 0-day and 6-month posttransplant were sampled to analyze the correlation of urinary TG2 and kidney allograft fibrosis.
RESULTS
Thirteen recipients had increased interstitial fibrosis and tubular atrophy (IFTA) scores at the 6-month protocol biopsy (IFTA group). The mean level of urinary TG2 in the IFTA group was higher compared to that of 5 other recipients without IFTA (no IFTA group). Conversely, the mean level of urinary syndecan-4 in the IFTA group was lower than levels in patients without IFTA. In the IFTA group, double immunofluorescent staining revealed that TG2 intensity was significantly upregulated and colocalizations of TG2/heparin sulfate proteoglycan and nuclear syndecan-4 were prominent, usually around tubular structures.
CONCLUSION
Urinary TG2 in early posttransplant periods is a potent biomarker for kidney allograft inflammation or fibrosis.
MeSH Terms
Figure
Cited by 1 articles
-
Retroperitoneal
Viscum album extract instillation in patients with a large amount of drainage after kidney transplantation
Young Jun Park, Sang Kyun Mok, Jang Yong Kim, Sang Seob Yun, Sun Cheol Park
Ann Surg Treat Res. 2021;101(6):368-373. doi: 10.4174/astr.2021.101.6.368.
Reference
-
1. Joosten SA, Sijpkens YW, van Kooten C, Paul LC. Chronic renal allograft rejection: pathophysiologic considerations. Kidney Int. 2005; 68:1–13.
Article2. Williams WW, Taheri D, Tolkoff-Rubin N, Colvin RB. Clinical role of the renal transplant biopsy. Nat Rev Nephrol. 2012; 8:110–121.
Article3. Furness PN, Taub N. Convergence of European Renal Transplant Pathology Assessment Procedures (CERTPAP) Project. International variation in the interpretation of renal transplant biopsies: report of the CERTPAP Project. Kidney Int. 2001; 60:1998–2012.4. Haas M, Sis B, Racusen LC, Solez K, Glotz D, Colvin RB, et al. Banff 2013 meeting report: inclusion of c4d-negative antibody-mediated rejection and antibody-associated arterial lesions. Am J Transplant. 2014; 14:272–283.
Article5. Christians U, Klawitter J, Klawitter J. Biomarkers in transplantation--proteomics and metabolomics. Ther Drug Monit. 2016; 38:Suppl 1. S70–S74.
Article6. Schaub S, Rush D, Wilkins J, Gibson IW, Weiler T, Sangster K, et al. Proteomic-based detection of urine proteins associated with acute renal allograft rejection. J Am Soc Nephrol. 2004; 15:219–227.
Article7. Scarpellini A, Huang L, Burhan I, Schroeder N, Funck M, Johnson TS, et al. Syndecan-4 knockout leads to reduced extracellular transglutaminase-2 and protects against tubulointerstitial fibrosis. J Am Soc Nephrol. 2014; 25:1013–1027.
Article8. Racca MA, Novoa PA, Rodríguez I, Della Vedova AB, Pellizas CG, Demarchi M, et al. Renal dysfunction and intragraft proMMP9 activity in renal transplant recipients with interstitial fibrosis and tubular atrophy. Transpl Int. 2015; 28:71–78.
Article9. Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009; 150:604–612.
Article10. Ix JH, Biggs ML, Mukamal K, Djousse L, Siscovick D, Tracy R, et al. Urine collagen fragments and CKD progression-the cardiovascular health study. J Am Soc Nephrol. 2015; 26:2494–2503.
Article11. Ix JH, Katz R, Bansal N, Foster M, Weiner DE, Tracy R, et al. Urine fibrosis markers and risk of allograft failure in kidney transplant recipients: a case-cohort ancillary study of the FAVORIT trial. Am J Kidney Dis. 2017; 69:410–419.
Article12. Reese PP, Hall IE, Weng FL, Schroppel B, Doshi MD, Hasz RD, et al. Associations between deceased-donor urine injury biomarkers and kidney transplant outcomes. J Am Soc Nephrol. 2016; 27:1534–1543.
Article13. Teppo AM, Tornroth T, Honkanen E, Gronhagen-Riska C. Urinary amino-terminal propeptide of type III procollagen (PIIINP) as a marker of interstitial fibrosis in renal transplant recipients. Transplantation. 2003; 75:2113–2119.
Article14. Teppo AM, Honkanen E, Finne P, Tornroth T, Gronhagen-Riska C. Increased urinary excretion of alpha1-microglobulin at 6 months after transplantation is associated with urinary excretion of transforming growth factor-beta1 and indicates poor long-term renal outcome. Transplantation. 2004; 78:719–724.15. Ho J, Rush DN, Krokhin O, Antonovici M, Gao A, Bestland J, et al. Elevated urinary matrix metalloproteinase-7 detects underlying renal allograft inflammation and injury. Transplantation. 2016; 100:648–654.
Article16. Hricik DE, Nickerson P, Formica RN, Poggio ED, Rush D, Newell KA, et al. Multicenter validation of urinary CXCL9 as a risk-stratifying biomarker for kidney transplant injury. Am J Transplant. 2013; 13:2634–2644.
Article17. Jones RA, Kotsakis P, Johnson TS, Chau DY, Ali S, Melino G, et al. Matrix changes induced by transglutaminase 2 lead to inhibition of angiogenesis and tumor growth. Cell Death Differ. 2006; 13:1442–1453.
Article18. Balajthy Z, Csomos K, Vamosi G, Szanto A, Lanotte M, Fesus L. Tissue-transglutaminase contributes to neutrophil granulocyte differentiation and functions. Blood. 2006; 108:2045–2054.
Article19. Stephens P, Grenard P, Aeschlimann P, Langley M, Blain E, Errington R, et al. Crosslinking and G-protein functions of transglutaminase 2 contribute differentially to fibroblast wound healing responses. J Cell Sci. 2004; 117(Pt 15):3389–3403.
Article20. Johnson TS, El-Koraie AF, Skill NJ, Baddour NM, El Nahas AM, Njloma M, et al. Tissue transglutaminase and the progression of human renal scarring. J Am Soc Nephrol. 2003; 14:2052–2062.
Article21. Verderio EA, Johnson TS, Griffin M. Transglutaminases in wound healing and inflammation. Prog Exp Tumor Res. 2005; 38:89–114.
Article22. Oh K, Park HB, Byoun OJ, Shin DM, Jeong EM, Kim YW, et al. Epithelial transglutaminase 2 is needed for T cell interleukin-17 production and subsequent pulmonary inflammation and fibrosis in bleomycin-treated mice. J Exp Med. 2011; 208:1707–1719.
Article23. Shweke N, Boulos N, Jouanneau C, Vandermeersch S, Melino G, Dussaule JC, et al. Tissue transglutaminase contributes to interstitial renal fibrosis by favoring accumulation of fibrillar collagen through TGF-beta activation and cell infiltration. Am J Pathol. 2008; 173:631–642.24. Shin S, Cho YP, Jun H, Park H, Hong HN, Kwon TW. Transglutaminase type 2 in human abdominal aortic aneurysm is a potential factor in the stabilization of extracellular matrix. J Vasc Surg. 2013; 57:1362–1370.
Article25. Scarpellini A, Germack R, Lortat-Jacob H, Muramatsu T, Billett E, Johnson T, et al. Heparan sulfate proteoglycans are receptors for the cell-surface trafficking and biological activity of transglutaminase-2. J Biol Chem. 2009; 284:18411–18423.
Article26. Belkin AM. Extracellular TG2: emerging functions and regulation. FEBS J. 2011; 278:4704–4716.
Article27. Richardson TP, Trinkaus-Randall V, Nugent MA. Regulation of heparan sulfate proteoglycan nuclear localization by fibronectin. J Cell Sci. 2001; 114(Pt 9):1613–1623.
Article28. Wee YM, Lee HW, Choi MY, Jung HR, Choi JY, Kwon HW, et al. A composite of urinary biomarkers for differentiating between tubulointerstitial inflammation and interstitial fibrosis/tubular atrophy in kidney allografts. Ann Hepatobiliary Pancreat Surg. 2018; 22:310–320.
Article29. Price JW. Creatinine normalization of workplace urine drug tests: does it make a difference? J Addict Med. 2013; 7:129–132.30. Zhao F, Song S, Liu W, Keep RF, Xi G, Hua Y. Red blood cell lysis and brain tissue-type transglutaminase upregulation in a hippocampal model of intracerebral hemorrhage. Acta Neurochir Suppl. 2011; 111:101–105.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Multicenter, prospective observational study to identify and validate a composite of urinary exosomal biomarkers for kidney allograft tubulointerstitial fibrosis
- Clinical relevance of postoperative proteinuria for prediction of early renal outcomes after kidney transplantation
- Cysteine as a potential donor urinary biomarker for donor acute kidney injury and recipient early graft function
- Medullary histology may help to predict Banff scores in allograft kidneys
- A composite of urinary biomarkers for differentiating between tubulointerstitial inflammation and interstitial fibrosis/tubular atrophy in kidney allografts