J Korean Soc Transplant.
2018 Dec;32(4):108-112. 10.4285/jkstn.2018.32.4.108.
Donor Specific Antibody Negative Antibody-Mediated Rejection after ABO Incompatible Liver Transplantation
- Affiliations
-
- 1Department of Surgery, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seongnam, Korea. choiyoungrok@gmail.com
- 2Department of Pathology, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seongnam, Korea.
- 3Department of Pathology, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea.
- KMID: 2430591
- DOI: http://doi.org/10.4285/jkstn.2018.32.4.108
Abstract
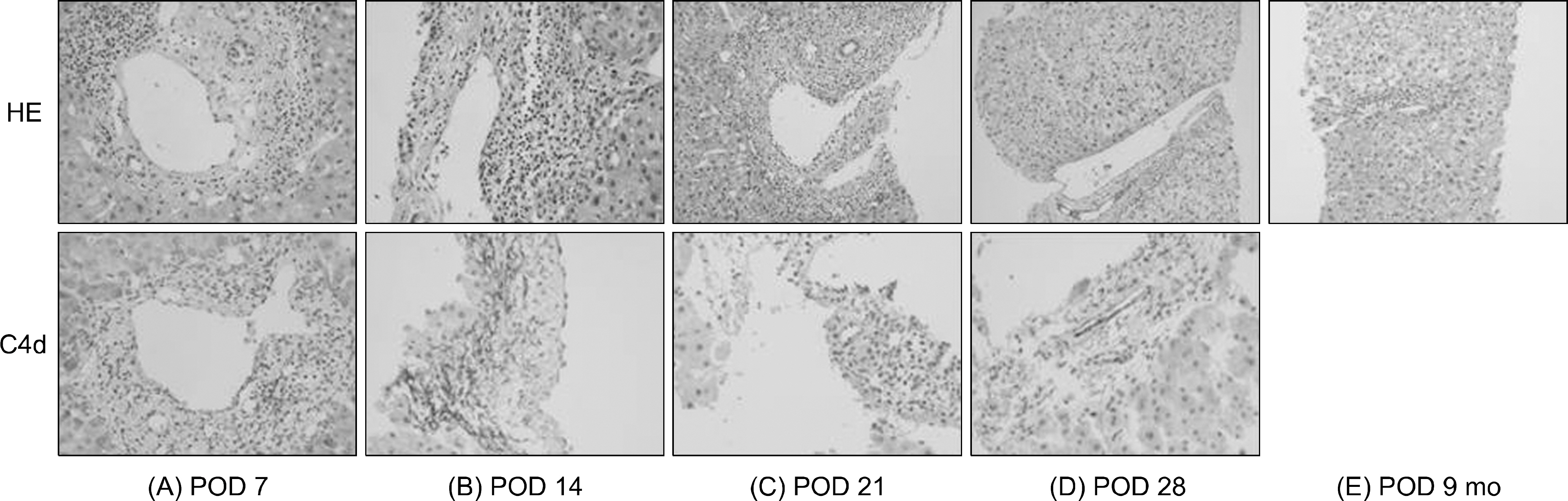
- Antibody-mediated rejection (AMR) is a major complication after ABO-incompatible liver transplantation. According to the 2016 Banff Working Group on Liver Allograft Criteria for the diagnosis of acute AMR, a positive serum donor specific antibody (DSA) is needed. On the other hand, the clinical significance of the histological findings of AMR in the absence of DSA is unclear. This paper describes a 57-year-old man (blood type, O+) who suffered from hepatitis B virus cirrhosis with hepatocellular carcinoma. Pre-operative DSA and cross-matching were negative. After transplantation, despite the improvement of the liver function, acute AMR was observed in the protocol biopsy on postoperative day 7; the cluster of differentiation 19+ (CD19+) count was 0% and anti-ABO antibody titers were 1:2. This paper presents the allograft injury like AMR in the absence of DSA after ABOi living donor liver transplantation with low titers of anti-ABO antibody and depleted serum CD19+ B cells.
Keyword
MeSH Terms
Figure
Reference
-
1). Cooper DK. A brief history of cross-species organ transplantation. Proc (Bayl Univ Med Cent). 2012. 25:49–57.
Article2). Gugenheim J., Samuel D., Reynes M., Bismuth H. Liver transplantation across ABO blood group barriers. Lancet. 1990. 336:519–23.
Article3). Usuda M., Fujimori K., Koyamada N., Fukumori T., Sekiguchi S., Kawagishi N, et al. Successful use of anti-CD20 mono-clonal antibody (rituximab) for ABO-incompatible living-related liver transplantation. Transplantation. 2005. 79:12–6.
Article4). Egawa H., Teramukai S., Haga H., Tanabe M., Fukushima M., Shimazu M. Present status of ABO-incompatible living donor liver transplantation in Japan. Hepatology. 2008. 47:143–52.
Article5). Demetris AJ., Bellamy C., Hubscher SG., O'Leary J., Randhawa PS., Feng S, et al. 2016 Comprehensive update of the Banff Working Group on liver allograft pathology: introduction of antibody-mediated rejection. Am J Transplant. 2016. 16:2816–35.6). Krishnan NS., Zehnder D., Briggs D., Higgins R. Human leukocyte antigen antibody incompatible renal transplantation. Indian J Nephrol. 2012. 22:409–14.
Article7). Song GW., Lee SG., Hwang S., Kim KH., Ahn CS., Moon DB, et al. ABO-incompatible adult living donor liver transplantation under the desensitization protocol with rituximab. Am J Transplant. 2016. 16:157–70.
Article8). Castillo-Rama M., Castro MJ., Bernardo I., Meneu-Diaz JC., Elola-Olaso AM., Calleja-Antolin SM, et al. Preformed antibodies detected by cytotoxic assay or multibead array decrease liver allograft survival: role of human leukocyte antigen compatibility. Liver Transpl. 2008. 14:554–62.
Article9). Kozlowski T., Rubinas T., Nickeleit V., Woosley J., Schmitz J., Collins D, et al. Liver allograft antibody-mediated rejection with demonstration of sinusoidal C4d staining and circulating donor-specific antibodies. Liver Transpl. 2011. 17:357–68.
Article10). O'Leary JG., Kaneku H., Jennings LW., Banuelos N., Susskind BM., Terasaki PI, et al. Preformed class II donor-specific antibodies are associated with an increased risk of early rejection after liver transplantation. Liver Transpl. 2013. 19:973–80.11). Taner T., Gandhi MJ., Sanderson SO., Poterucha CR., De Goey Sr., Stegall MD, et al. Prevalence, course and impact of HLA donor-specific antibodies in liver transplantation in the first year. Am J Transplant. 2012. 12:1504–10.
Article12). Kaneku H., O'Leary JG., Banuelos N., Jennings LW., Susskind BM., Klintmalm GB, et al. De novo donor-specific HLA antibodies decrease patient and graft survival in liver transplant recipients. Am J Transplant. 2013. 13:1541–8.13). Zou Y., Heinemann FM., Grosse-Wilde H., Sireci G., Wang Z., Lavingia B, et al. Detection of anti-MICA antibodies in patients awaiting kidney transplantation, during the post-transplant course, and in eluates from rejected kidney allografts by Luminex flow cytometry. Hum Immunol. 2006. 67:230–7.
Article14). Alten TA., Negm AA., Voigtlander T., Jaeckel E., Lehner F., Brauner C, et al. Safety and performance of liver biopsies in liver transplant recipients. Clin Transplant. 2014. 28:585–9.
Article15). O'Leary JG., Kaneku H., Demetris AJ., Marr JD., Shiller SM., Susskind BM, et al. Antibody-mediated rejection as a contributor to previously unexplained early liver allograft loss. Liver Transpl. 2014. 20:218–27.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Diagnosis of Acute Antibody-Mediated Rejection in ABO-Incompatible Liver Transplants
- Immunologic strategies and outcomes in ABO-incompatible living donor liver transplantation
- Overcoming high pre-transplant isoagglutinin titers using high-dose intravenous immunoglobulin, salvage plasmapheresis, and booster rituximab without splenectomy in ABO-incompatible living donor liver transplantation: a case report
- ABO-incompatible living donor liver transplantation with a simplified desensitization and immunosuppression protocol: a single center retrospective study
- Desensitization in HLA Incompatible Transplantation