Anat Cell Biol.
2018 Dec;51(4):266-273. 10.5115/acb.2018.51.4.266.
Ganglion cardiacum or juxtaductal body of human fetuses
- Affiliations
-
- 1Department of Anatomy and Institute of Medical Science, Chonbuk National University Medical School, Jeonju, Korea. 407kk@hanmail.net
- 2Department of Neurology, Wonkwang University School of Medicine and Hospital, Institute of Wonkwang Medical Science, Iksan, Korea.
- 3Department of Anatomy, Wuxi Medical School, Jiangnan University, Wuxi, China.
- 4Division of Internal Medicine, Asuka Hospital, Sapporo, Japan.
- 5Department of Anatomy, Akita University School of Medicine, Akita, Japan.
- KMID: 2430194
- DOI: http://doi.org/10.5115/acb.2018.51.4.266
Abstract
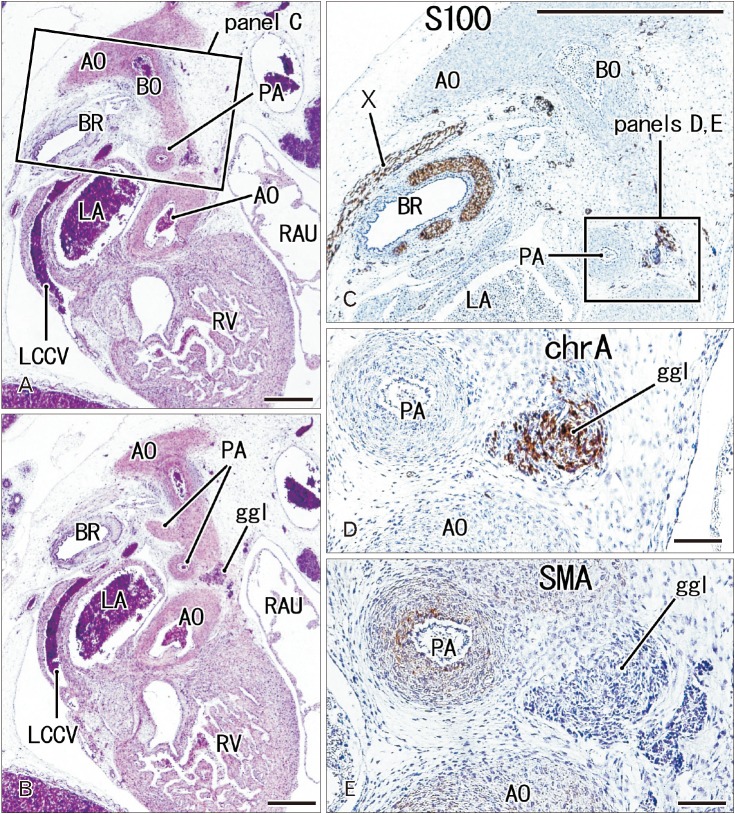
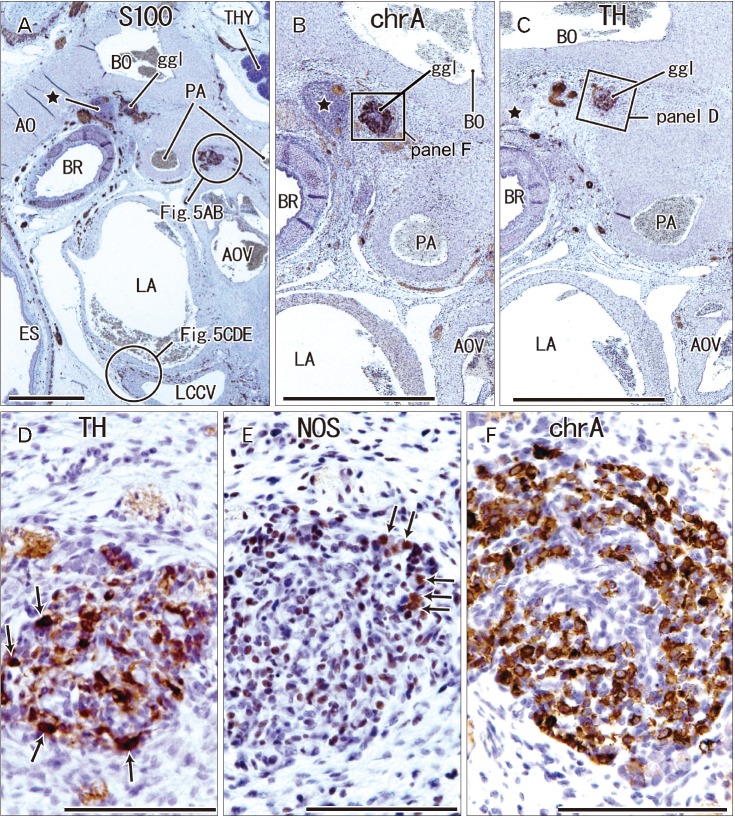
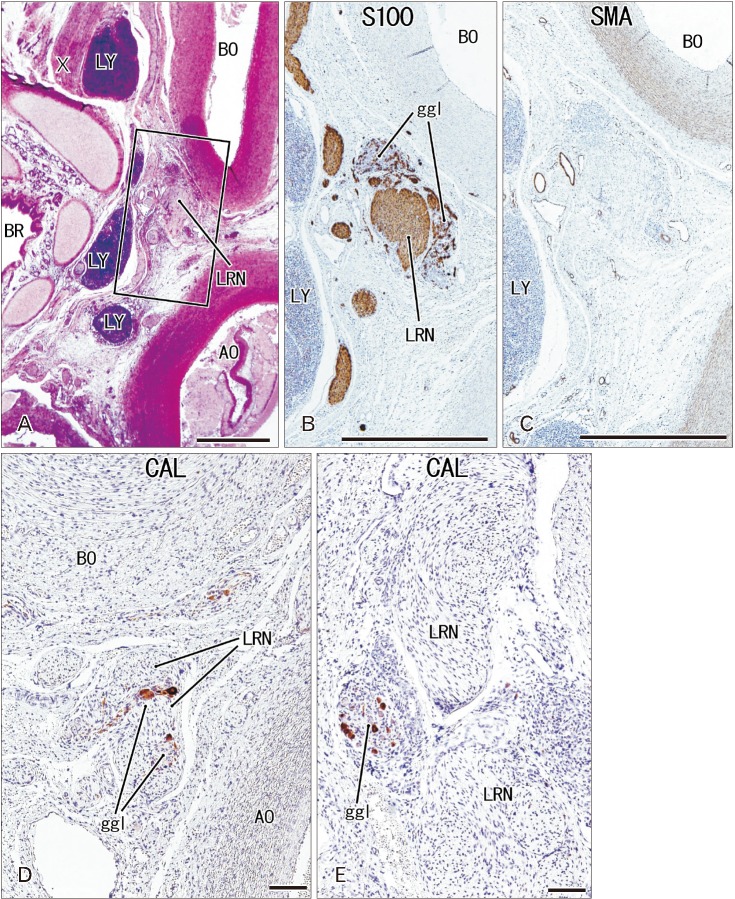
- The ganglion cardiacum or juxtaductal body is situated along the left recurrent laryngeal nerve in the aortic window and is an extremely large component of the cardiac nerve plexus. This study was performed to describe the morphologies of the ganglion cardiacum or juxtaductal body in human fetuses and to compare characteristics with intracardiac ganglion. Ganglia were immunostained in specimens from five fetuses of gestational age 12-16 weeks and seven fetuses of gestational age 28-34 weeks. Many ganglion cells in the ganglia were positive for tyrosine hydroxylase (TH; sympathetic nerve marker) and chromogranin A, while a few neurons were positive for neuronal nitric oxide synthase (NOS; parasympathetic nerve marker) or calretinin. Another ganglion at the base of the ascending aorta carried almost the same neuronal populations, whereas a ganglion along the left common cardinal vein contained neurons positive for chromogranin A and NOS but no or few TH-positive neurons, suggesting a site-dependent difference in composite neurons. Mixtures of sympathetic and parasympathetic neurons within a single ganglion are consistent with the morphology of the cranial base and pelvic ganglia. Most of the intracardiac neurons are likely to have a non-adrenergic non-cholinergic phenotype, whereas fewer neurons have a dual cholinergic/noradrenergic phenotype. However, there was no evidence showing that chromogranin A- and/or calretinin-positive cardiac neurons corresponded to these specific phenotypes. The present study suggested that the ganglion cardiacum was composed of a mixture of sympathetic and parasympathetic neurons, which were characterized the site-dependent differences in and near the heart.
Keyword
MeSH Terms
Figure
Reference
-
1. Toldt C. Anatomischer Atlas fűr Studierende und Arzte. Vol. 6. Berlin: Urban & Schwarzenberg;1903.2. Pauza DH, Skripka V, Pauziene N, Stropus R. Morphology, distribution, and variability of the epicardiac neural ganglionated subplexuses in the human heart. Anat Rec. 2000; 259:353–382. PMID: 10903529.3. Kawashima T. The autonomic nervous system of the human heart with special reference to its origin, course, and peripheral distribution. Anat Embryol (Berl). 2005; 209:425–438. PMID: 15887046.4. Taguchi K, Tsukamoto T, Murakami G. Anatomical studies of the autonomic nervous system in the human pelvis by the whole-mount staining method: left-right communicating nerves between bilateral pelvic plexuses. J Urol. 1999; 161:320–325. PMID: 10037431.5. Brack KE. The heart's ‘little brain’ controlling cardiac function in the rabbit. Exp Physiol. 2015; 100:348–353. PMID: 25833107.6. Pauziene N, Alaburda P, Rysevaite-Kyguoliene K, Pauza AG, Inokaitis H, Masaityte A, Rudokaite G, Saburkina I, Plisiene J, Pauza DH. Innervation of the rabbit cardiac ventricles. J Anat. 2016; 228:26–46. PMID: 26510903.7. Maifrino LB, Liberti EA, Castelucci P, De Souza RR. NADPH-diaphorase positive cardiac neurons in the atria of mice: a morphoquantitative study. BMC Neurosci. 2006; 7:10. PMID: 16451738.8. Rysevaite K, Saburkina I, Pauziene N, Vaitkevicius R, Noujaim SF, Jalife J, Pauza DH. Immunohistochemical characterization of the intrinsic cardiac neural plexus in whole-mount mouse heart preparations. Heart Rhythm. 2011; 8:731–738. PMID: 21232628.9. Richardson RJ, Grkovic I, Anderson CR. Immunohistochemical analysis of intracardiac ganglia of the rat heart. Cell Tissue Res. 2003; 314:337–350. PMID: 14523644.10. Steele PA, Gibbins IL, Morris JL, Mayer B. Multiple populations of neuropeptide-containing intrinsic neurons in the guinea-pig heart. Neuroscience. 1994; 62:241–250. PMID: 7816202.11. Hoover DB, Ganote CE, Ferguson SM, Blakely RD, Parsons RL. Localization of cholinergic innervation in guinea pig heart by immunohistochemistry for high-affinity choline transporters. Cardiovasc Res. 2004; 62:112–121. PMID: 15023558.12. Parsons RL, Locknar SA, Young BA, Hoard JL, Hoover DB. Presence and co-localization of vasoactive intestinal polypeptide with neuronal nitric oxide synthase in cells and nerve fibers within guinea pig intrinsic cardiac ganglia and cardiac tissue. Cell Tissue Res. 2006; 323:197–209. PMID: 16220273.13. Tan AY, Li H, Wachsmann-Hogiu S, Chen LS, Chen PS, Fishbein MC. Autonomic innervation and segmental muscular disconnections at the human pulmonary vein-atrial junction: implications for catheter ablation of atrial-pulmonary vein junction. J Am Coll Cardiol. 2006; 48:132–143. PMID: 16814659.14. Hoover DB, Isaacs ER, Jacques F, Hoard JL, Pagé P, Armour JA. Localization of multiple neurotransmitters in surgically derived specimens of human atrial ganglia. Neuroscience. 2009; 164:1170–1179. PMID: 19747529.15. Singh S, Johnson PI, Javed A, Gray TS, Lonchyna VA, Wurster RD. Monoamine- and histamine-synthesizing enzymes and neurotransmitters within neurons of adult human cardiac ganglia. Circulation. 1999; 99:411–419. PMID: 9918529.16. Singh S, Gray T, Wurster RD. Nitric oxide and carbon monoxide synthesizing enzymes and soluble guanylyl cyclase within neurons of adult human cardiac ganglia. Auton Neurosci. 2009; 145:93–98. PMID: 19106038.17. Katori Y, Jin ZW, Kawase T, Hong KH, Murakami G, Cho BH. Developmental changes in the distribution of calretinin-immunoreactive cells in human fetal nasal epithelium. Okajimas Folia Anat Jpn. 2010; 87:5–10. PMID: 20715566.18. Jin ZW, Cho KH, Jang HS, Murakami G, Rodriguez-Vazquez JF. Median sacral artery, sympathetic nerves, and the coccygeal body: a study using serial sections of human embryos and fetuses. Anat Rec (Hoboken). 2016; 299:819–827. PMID: 27149546.19. Licata RH. The human embryonic heart in the ninth week. Am J Anat. 1954; 94:73–125. PMID: 13124266.20. Orts Llorca F, Domenech Mateu JM, Puerta Fonolla J. Innervation of the sinu-atrial node and neighbouring regions in two human embryos. J Anat. 1979; 128(Pt 2):365–375. PMID: 438095.21. Jin ZW, Cho KH, Jang HS, Murakami G, Rodríguez-Vázquez JF, Yamamoto M, Abe SI. Coccygeal body revisited: an immunohistochemical study using donated elderly cadavers. Anat Rec (Hoboken). 2017; 300:1826–1837. PMID: 28545163.22. Hinata N, Hieda K, Sasaki H, Murakami G, Abe S, Matsubara A, Miyake H, Fujisawa M. Topohistology of sympathetic and parasympathetic nerve fibers in branches of the pelvic plexus: an immunohistochemical study using donated elderly cadavers. Anat Cell Biol. 2014; 47:55–65. PMID: 24693483.23. Kiyokawa H, Katori Y, Cho KH, Murakami G, Kawase T, Cho BH. Reconsideration of the autonomic cranial ganglia: an immunohistochemical study of mid-term human fetuses. Anat Rec (Hoboken). 2012; 295:141–149. PMID: 22095632.24. Roudenok V, Kühnel W. Distribution of vasoactive intestinal polypeptide-, calcitonin gene-related peptide-, somatostatin- and neurofilament-immunoreactivities in sympathetic ganglia of human fetuses and premature neonates. Ann Anat. 2001; 183:213–216. PMID: 11396789.25. Hoard JL, Hoover DB, Mabe AM, Blakely RD, Feng N, Paolocci N. Cholinergic neurons of mouse intrinsic cardiac ganglia contain noradrenergic enzymes, norepinephrine transporters, and the neurotrophin receptors tropomyosin-related kinase A and p75. Neuroscience. 2008; 156:129–142. PMID: 18674600.26. Magro G, Grasso S. Immunohisochemical identification and comparison of glial cell lineage in foetal, neonatal, adult and neoplastic human adrenal medulla. Histochem J. 1997; 29:293–299. PMID: 9184844.27. Inoue S, Cho BH, Song CH, Fujimiya M, Murakami G, Matsubara A. Migration and distribution of neural crest-derived cells in the human adrenal cortex at 9-16 weeks of gestation: an immunohistochemical study. Okajimas Folia Anat Jpn. 2010; 87:11–16. PMID: 20715567.28. Papka RE, Collins J, Copelin T, Wilson K. Calretinin-immunoreactive nerves in the uterus, pelvic autonomic ganglia, lumbosacral dorsal root ganglia and lumbosacral spinal cord. Cell Tissue Res. 1999; 298:63–74. PMID: 10555540.29. Liu CY, Zhang H, Christofi FL. Adenylyl cyclase co-distribution with the CaBPs, calbindin-D28 and calretinin, varies with cell type: assessment with the fluorescent dye, BODIPY forskolin, in enteric ganglia. Cell Tissue Res. 1998; 293:57–73. PMID: 9634598.30. Hasan W. Autonomic cardiac innervation: development and adult plasticity. Organogenesis. 2013; 9:176–193. PMID: 23872607.31. Terada T. Huge clusters of embryonic stem cells in human embryos: a morphologic study. Microsc Res Tech. 2014; 77:825–831. PMID: 25091607.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Ultrastructural Study on the Development of Ciliary Body in Human Fetuses
- An Ultrastructural Study on the Structural Development of the Cardiac Ganglion in the Human Fetuses
- Ultrastructural Study on Development of the Superior Cervical Ganglion of Human Fetuses
- Pulmonary Arterial Growth Pattern after Shunt Operation in Patients of Pulmonary Atresia with Ventricular Septal Defect Associated with Juxtaductal Stenosis
- Electron Microscopic Study on the Development of the Spinal Ganglion of Human Fetus