Cancer Res Treat.
2018 Jan;50(1):255-264. 10.4143/crt.2017.062.
Comparison of Ion Personal Genome Machine Platforms for the Detection of Variants in BRCA1 and BRCA2
- Affiliations
-
- 1Department of Laboratory Medicine, Seoul National University Bundang Hospital, Seongnam, Korea. m91w95pf@snu.ac.kr
- 2Department of Laboratory Medicine, Seoul National University College of Medicine, Seoul, Korea.
- 3Eone-Diagnomics Genome Center, Incheon, Korea.
- KMID: 2403494
- DOI: http://doi.org/10.4143/crt.2017.062
Abstract
- PURPOSE
Transition to next generation sequencing (NGS) for BRCA1/BRCA2 analysis in clinical laboratories is ongoing but different platforms and/or data analysis pipelines give different results resulting in difficulties in implementation. We have evaluated the Ion Personal Genome Machine (PGM) Platforms (Ion PGM, Ion PGM Dx, Thermo Fisher Scientific) for the analysis of BRCA1/2.
MATERIALS AND METHODS
The results of Ion PGM with OTG-snpcaller, a pipeline based on Torrent mapping alignment program and Genome Analysis Toolkit, from 75 clinical samples and 14 reference DNA samples were compared with Sanger sequencing for BRCA1/BRCA2. Ten clinical samples and 14 reference DNA samples were additionally sequenced by Ion PGM Dx with Torrent Suite.
RESULTS
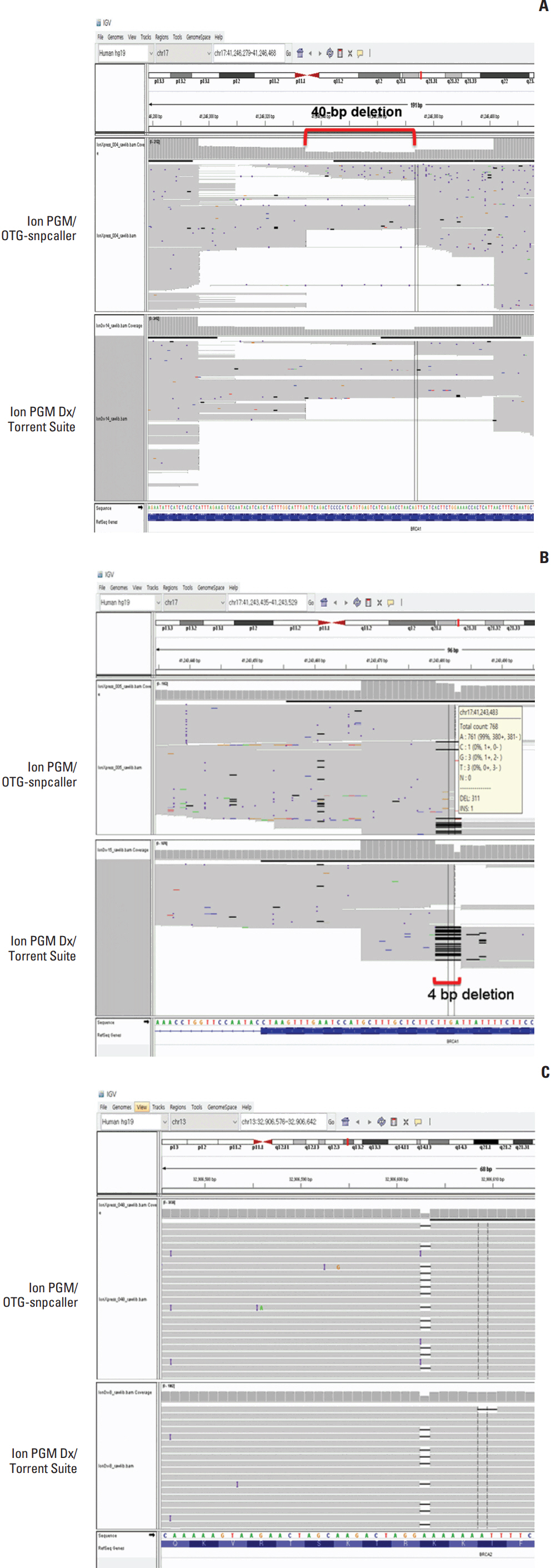
Fifty types of variants including 18 pathogenic or variants of unknown significance were identified from 75 clinical samples and known variants of the reference samples were confirmed by Sanger sequencing and/or NGS. One false-negative results were present for Ion PGM/OTG-snpcaller for an indel variant misidentified as a single nucleotide variant. However, eight discordant results were present for Ion PGM Dx/Torrent Suite with both false-positive and -negative results. A 40-bp deletion, a 4-bp deletion and a 1-bp deletion variant was not called and a false-positive deletion was identified. Four other variants were misidentified as another variant.
CONCLUSION
Ion PGM/OTG-snpcaller showed acceptable performance with good concordance with Sanger sequencing. However, Ion PGM Dx/Torrent Suite showed many discrepant results not suitable for use in a clinical laboratory, requiring further optimization of the data analysis for calling variants.
Keyword
Figure
Reference
-
References
1. Ford D, Easton DF, Stratton M, Narod S, Goldgar D, Devilee P, et al. Genetic heterogeneity and penetrance analysis of the BRCA1 and BRCA2 genes in breast cancer families. The Breast Cancer Linkage Consortium. Am J Hum Genet. 1998; 62:676–89.2. King MC, Marks JH, Mandell JB; New York Breast Cancer Study Group. Breast and ovarian cancer risks due to inherited mutations in BRCA1 and BRCA2. Science. 2003; 302:643–6.3. Hartmann LC, Lindor NM. The role of risk-reducing surgery in hereditary breast and ovarian cancer. N Engl J Med. 2016; 374:454–68.
Article4. National Comprehensive Cancer Network. NCCN guidelines, version 2. 2016. Breast and/or ovarian cancer genetic assessment. Fort Washington, PA: National Comprehensive Cancer Network;2016.5. Tafe LJ, Datto MB, Palomaki GE, Lacbawan FL; CAP/ACMG Biochemical and Molecular Genetics Resource Committee. Molecular testing for the BRCA1 and BRCA2 Ashkenazi Jewish founder mutations: a report on the College of American Pathologists proficiency testing surveys. Genet Med. 2015; 17:58–62.
Article6. Gerhardus A, Schleberger H, Schlegelberger B, Gadzicki D. Diagnostic accuracy of methods for the detection of BRCA1 and BRCA2 mutations: a systematic review. Eur J Hum Genet. 2007; 15:619–27.
Article7. Chan M, Ji SM, Yeo ZX, Gan L, Yap E, Yap YS, et al. Development of a next-generation sequencing method for BRCA mutation screening: a comparison between a high-throughput and a benchtop platform. J Mol Diagn. 2012; 14:602–12.8. Feliubadalo L, Lopez-Doriga A, Castellsague E, del Valle J, Menendez M, Tornero E, et al. Next-generation sequencing meets genetic diagnostics: development of a comprehensive workflow for the analysis of BRCA1 and BRCA2 genes. Eur J Hum Genet. 2013; 21:864–70.
Article9. Judkins T, Leclair B, Bowles K, Gutin N, Trost J, McCulloch J, et al. Development and analytical validation of a 25-gene next generation sequencing panel that includes the BRCA1 and BRCA2 genes to assess hereditary cancer risk. BMC Cancer. 2015; 15:215.
Article10. De Leeneer K, Hellemans J, De Schrijver J, Baetens M, Poppe B, Van Criekinge W, et al. Massive parallel amplicon sequencing of the breast cancer genes BRCA1 and BRCA2: opportunities, challenges, and limitations. Hum Mutat. 2011; 32:335–44.
Article11. Bosdet IE, Docking TR, Butterfield YS, Mungall AJ, Zeng T, Coope RJ, et al. A clinically validated diagnostic second-generation sequencing assay for detection of hereditary BRCA1 and BRCA2 mutations. J Mol Diagn. 2013; 15:796–809.
Article12. Tarabeux J, Zeitouni B, Moncoutier V, Tenreiro H, Abidallah K, Lair S, et al. Streamlined ion torrent PGM-based diagnostics: BRCA1 and BRCA2 genes as a model. Eur J Hum Genet. 2014; 22:535–41.
Article13. Dacheva D, Dodova R, Popov I, Goranova T, Mitkova A, Mitev V, et al. Validation of an NGS approach for diagnostic BRCA1/BRCA2 mutation testing. Mol Diagn Ther. 2015; 19:119–30.
Article14. Kluska A, Balabas A, Paziewska A, Kulecka M, Nowakowska D, Mikula M, et al. New recurrent BRCA1/2 mutations in Polish patients with familial breast/ovarian cancer detected by next generation sequencing. BMC Med Genomics. 2015; 8:19.
Article15. Trujillano D, Weiss ME, Schneider J, Koster J, Papachristos EB, Saviouk V, et al. Next-generation sequencing of the BRCA1 and BRCA2 genes for the genetic diagnostics of hereditary breast and/or ovarian cancer. J Mol Diagn. 2015; 17:162–70.
Article16. Park J, Jang W, Chae H, Kim Y, Chi HY, Kim M. Comparison of targeted next-generation and Sanger sequencing for the BRCA1 and BRCA2 mutation screening. Ann Lab Med. 2016; 36:197–201.
Article17. Strom CM, Rivera S, Elzinga C, Angeloni T, Rosenthal SH, Goos-Root D, et al. Development and validation of a next-generation sequencing assay for BRCA1 and BRCA2 variants for the clinical laboratory. PLoS One. 2015; 10:e0136419.
Article18. Yeo ZX, Chan M, Yap YS, Ang P, Rozen S, Lee AS. Improving indel detection specificity of the Ion Torrent PGM benchtop sequencer. PLoS One. 2012; 7:e45798.
Article19. Bragg LM, Stone G, Butler MK, Hugenholtz P, Tyson GW. Shining a light on dark sequencing: characterising errors in Ion Torrent PGM data. PLoS Comput Biol. 2013; 9:e1003031.
Article20. McKenna A, Hanna M, Banks E, Sivachenko A, Cibulskis K, Kernytsky A, et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 2010; 20:1297–303.
Article21. Zhu P, He L, Li Y, Huang W, Xi F, Lin L, et al. OTG-snpcaller: an optimized pipeline based on TMAP and GATK for SNP calling from ion torrent data. PLoS One. 2014; 9:e97507.
Article22. Robinson JT, Thorvaldsdottir H, Winckler W, Guttman M, Lander ES, Getz G, et al. Integrative genomics viewer. Nat Biotechnol. 2011; 29:24–6.
Article23. Quail MA, Smith M, Coupland P, Otto TD, Harris SR, Connor TR, et al. A tale of three next generation sequencing platforms: comparison of Ion Torrent, Pacific Biosciences and Illumina MiSeq sequencers. BMC Genomics. 2012; 13:341.
Article24. Enyedi MZ, Jaksa G, Pinter L, Sukosd F, Gyuris Z, Hajdu A, et al. Simultaneous detection of BRCA mutations and large genomic rearrangements in germline DNA and FFPE tumor samples. Oncotarget. 2016; 7:61845–59.
Article25. Lincoln SE, Kobayashi Y, Anderson MJ, Yang S, Desmond AJ, Mills MA, et al. A systematic comparison of traditional and multigene panel testing for hereditary breast and ovarian cancer genes in more than 1000 patients. J Mol Diagn. 2015; 17:533–44.
Article26. Yeo ZX, Wong JC, Rozen SG, Lee AS. Evaluation and optimisation of indel detection workflows for ion torrent sequencing of the BRCA1 and BRCA2 genes. BMC Genomics. 2014; 15:516.
Article27. Kang HP, Maguire JR, Chu CS, Haque IS, Lai H, Mar-Heyming R, et al. Design and validation of a next generation sequencing assay for hereditary BRCA1 and BRCA2 mutation testing. PeerJ. 2016; 4:e2162.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Immunohisochemical study of BRCA1, BRCA2, and poly (ADP-ribose) polymerase-1 in ovarian tumors
- Prognostic implications of ductal carcinoma in situ components in BRCA1/2-positive breast cancer: a retrospective cohort study
- Reclassification of BRCA1 and BRCA2 variants found in ovarian epithelial, fallopian tube, and primary peritoneal cancers
- Clinically Significant Unclassified Variants in BRCA1 and BRCA2 genes among Korean Breast Cancer Patients
- Performance Evaluation of BRCA1/2 Genetic Test Using Next-Generation Sequencing Based on Target Capture Method