Diabetes Metab J.
2022 Jan;46(1):81-92. 10.4093/dmj.2021.0016.
Effects of Teneligliptin on HbA1c levels, Continuous Glucose Monitoring-Derived Time in Range and Glycemic Variability in Elderly Patients with T2DM (TEDDY Study)
- Affiliations
-
- 1Division of Endocrinology and Metabolism, Department of Internal Medicine, Samsung Changwon Hospital, Sungkyunkwan University School of Medicine, Changwon, Korea
- 2Division of Endocrinology and Metabolism, Department of Internal Medicine, Seoul National University Hospital, Seoul, Korea
- 3Division of Endocrinology and Metabolism, Department of Internal Medicine, Chungnam National University College of Medicine, Daejeon, Korea
- 4Division of Endocrinology and Metabolism, Department of Internal Medicine, Chosun University College of Medicine, Gwangju, Korea
- 5Division of Endocrinology and Metabolism, Department of Medicine, Kyung Hee University Hospital at Gangdong, Kyung Hee University School of Medicine, Seoul, Korea
- 6Division of Endocrinology and Metabolism, Department of Internal Medicine, Dong-A University Medical Center, Dong-A University College of Medicine, Busan, Korea
- 7Handok Inc., Seoul, Korea
- 8Division of Endocrinology and Metabolism, Department of Internal Medicine, Cardiovascular and Metabolic Disease Center, Inje University Sanggye Paik Hospital, Inje University College of Medicine, Seoul, Korea
- 9Division of Endocrinology and Metabolism, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
- KMID: 2525128
- DOI: http://doi.org/10.4093/dmj.2021.0016
Abstract
- Background
To evaluate the effects of teneligliptin on glycosylated hemoglobin (HbA1c) levels, continuous glucose monitoring (CGM)-derived time in range, and glycemic variability in elderly type 2 diabetes mellitus patients.
Methods
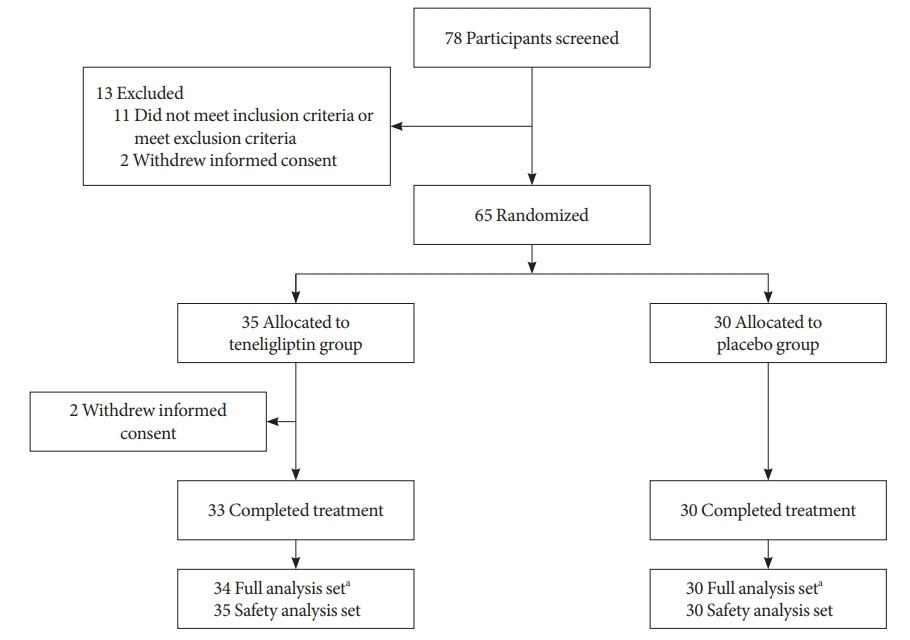
This randomized, double-blinded, placebo-controlled study was conducted in eight centers in Korea (clinical trial registration number: NCT03508323). Sixty-five participants aged ≥65 years, who were treatment-naïve or had been treated with stable doses of metformin, were randomized at a 1:1 ratio to receive 20 mg of teneligliptin (n=35) or placebo (n=30) for 12 weeks. The main endpoints were the changes in HbA1c levels from baseline to week 12, CGM metrics-derived time in range, and glycemic variability.
Results
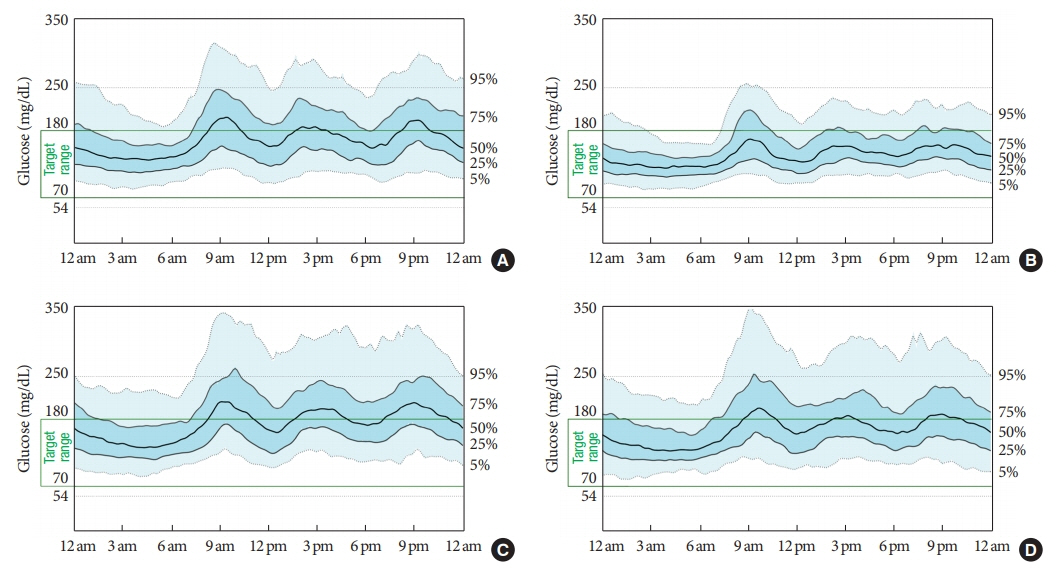
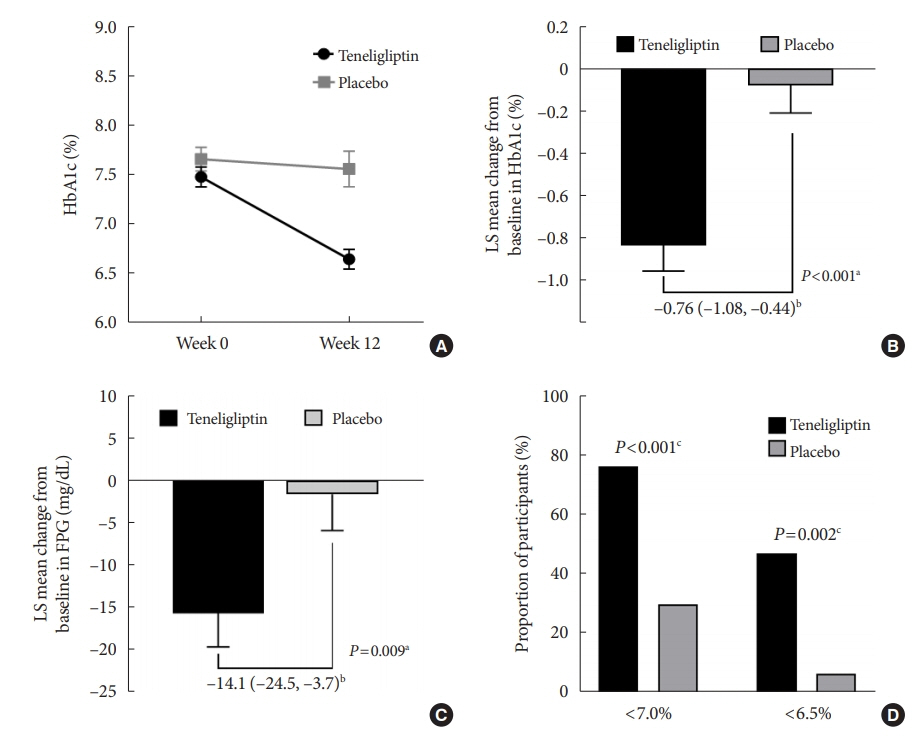
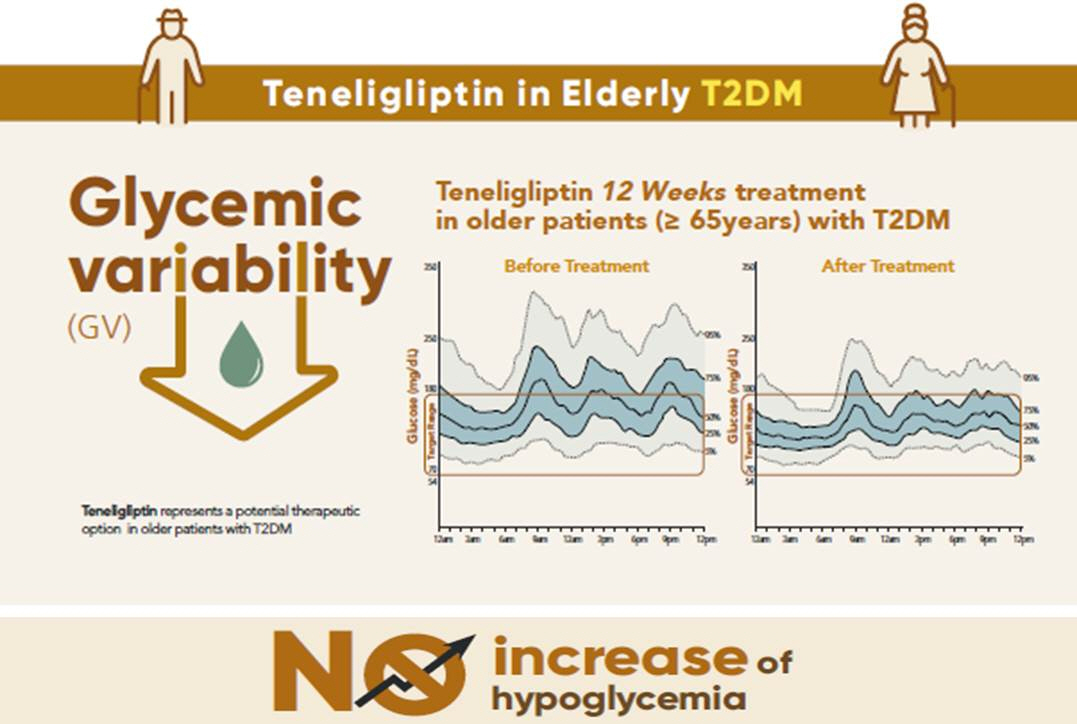
After 12 weeks, a significant reduction (by 0.84%) in HbA1c levels was observed in the teneligliptin group compared to that in the placebo group (by 0.08%), with a between-group least squares mean difference of –0.76% (95% confidence interval [CI], –1.08 to –0.44). The coefficient of variation, standard deviation, and mean amplitude of glycemic excursion significantly decreased in participants treated with teneligliptin as compared to those in the placebo group. Teneligliptin treatment significantly decreased the time spent above 180 or 250 mg/dL, respectively, without increasing the time spent below 70 mg/dL. The mean percentage of time for which glucose levels remained in the 70 to 180 mg/dL time in range (TIR70–180) at week 12 was 82.0%±16.0% in the teneligliptin group, and placebo-adjusted change in TIR70–180 from baseline was 13.3% (95% CI, 6.0 to 20.6).
Conclusion
Teneligliptin effectively reduced HbA1c levels, time spent above the target range, and glycemic variability, without increasing hypoglycemia in our study population.
Keyword
Figure
Cited by 1 articles
-
Association between Variability of Metabolic Risk Factors and Cardiometabolic Outcomes
Min Jeong Park, Kyung Mook Choi
Diabetes Metab J. 2022;46(1):49-62. doi: 10.4093/dmj.2021.0316.
Reference
-
1. International Diabetes Federation. IDF Diabetes Atlas. 9th ed. Brussels: International Diabetes Federation;2019.2. Jung CH, Son JW, Kang S, Kim WJ, Kim HS, Kim HS, et al. Diabetes fact sheets in Korea, 2020: an appraisal of current status. Diabetes Metab J. 2021; 45:1–10.
Article3. Bae JC. Trends of diabetes epidemic in Korea. Diabetes Metab J. 2018; 42:377–9.
Article4. Ha KH, Kim DJ. Trends in the diabetes epidemic in Korea. Endocrinol Metab (Seoul). 2015; 30:142–6.
Article5. Kirkman MS, Briscoe VJ, Clark N, Florez H, Haas LB, Halter JB, et al. Diabetes in older adults. Diabetes Care. 2012; 35:2650–64.
Article6. Yakaryılmaz FD, Ozturk ZA. Treatment of type 2 diabetes mellitus in the elderly. World J Diabetes. 2017; 8:278–85.
Article7. Lee PG, Halter JB. The pathophysiology of hyperglycemia in older adults: clinical considerations. Diabetes Care. 2017; 40:444–52.
Article8. Bremer JP, Jauch-Chara K, Hallschmid M, Schmid S, Schultes B. Hypoglycemia unawareness in older compared with middle-aged patients with type 2 diabetes. Diabetes Care. 2009; 32:1513–7.
Article9. Tanaka C, Saisho Y, Tanaka K, Kou K, Tanaka M, Meguro S, et al. Factors associated with glycemic variability in Japanese patients with diabetes. Diabetol Int. 2014; 5:36–42.
Article10. Gude F, Diaz-Vidal P, Rua-Perez C, Alonso-Sampedro M, Fernandez-Merino C, Rey-Garcia J, et al. Glycemic variability and its association with demographics and lifestyles in a general adult population. J Diabetes Sci Technol. 2017; 11:780–90.
Article11. Zhou Z, Sun B, Huang S, Zhu C, Bian M. Glycemic variability: adverse clinical outcomes and how to improve it? Cardiovasc Diabetol. 2020; 19:102.
Article12. Jung HS. Clinical implications of glucose variability: chronic complications of diabetes. Endocrinol Metab (Seoul). 2015; 30:167–74.
Article13. Battelino T, Danne T, Bergenstal RM, Amiel SA, Beck R, Biester T, et al. Clinical targets for continuous glucose monitoring data interpretation: recommendations from the international consensus on time in range. Diabetes Care. 2019; 42:1593–603.14. Lu J, Ma X, Zhou J, Zhang L, Mo Y, Ying L, et al. Association of time in range, as assessed by continuous glucose monitoring, with diabetic retinopathy in type 2 diabetes. Diabetes Care. 2018; 41:2370–6.
Article15. Lu J, Ma X, Shen Y, Wu Q, Wang R, Zhang L, et al. Time in range is associated with carotid intima-media thickness in type 2 diabetes. Diabetes Technol Ther. 2020; 22:72–8.
Article16. Yoo JH, Choi MS, Ahn J, Park SW, Kim Y, Hur KY, et al. Association between continuous glucose monitoring-derived time in range, other core metrics, and albuminuria in type 2 diabetes. Diabetes Technol Ther. 2020; 22:768–76.
Article17. Yang J, Yang X, Zhao D, Wang X, Wei W, Yuan H. Association of time in range, as assessed by continuous glucose monitoring, with painful diabetic polyneuropathy. J Diabetes Investig. 2021; 12:828–36.
Article18. Schott G, Martinez YV, Ediriweera de Silva RE, Renom-Guiteras A, Vogele A, Reeves D, et al. Effectiveness and safety of dipeptidyl peptidase 4 inhibitors in the management of type 2 diabetes in older adults: a systematic review and development of recommendations to reduce inappropriate prescribing. BMC Geriatr. 2017; 17(Suppl 1):226.
Article19. Lee S, Lee H, Kim Y, Kim E. Effect of DPP-IV inhibitors on glycemic variability in patients with T2DM: a systematic review and meta-analysis. Sci Rep. 2019; 9:13296.
Article20. Kim MK, Rhee EJ, Han KA, Woo AC, Lee MK, Ku BJ, et al. Efficacy and safety of teneligliptin, a dipeptidyl peptidase-4 inhibitor, combined with metformin in Korean patients with type 2 diabetes mellitus: a 16-week, randomized, double- blind, placebo-controlled phase III trial. Diabetes Obes Metab. 2015; 17:309–12.
Article21. Kadowaki T, Sasaki K, Ishii M, Matsukawa M, Ushirogawa Y. Efficacy and safety of teneligliptin 40 mg in type 2 diabetes: a pooled analysis of two phase III clinical studies. Diabetes Ther. 2018; 9:623–36.
Article22. Boyd CM, Darer J, Boult C, Fried LP, Boult L, Wu AW. Clinical practice guidelines and quality of care for older patients with multiple comorbid diseases: implications for pay for performance. JAMA. 2005; 294:716–24.
Article23. Herrera AP, Snipes SA, King DW, Torres-Vigil I, Goldberg DS, Weinberg AD. Disparate inclusion of older adults in clinical trials: priorities and opportunities for policy and practice change. Am J Public Health. 2010; 100 Suppl 1:S105–12.
Article24. Suh S, Kim JH. Glycemic variability: how do we measure it and why is it important? Diabetes Metab J. 2015; 39:273–82.
Article25. Vigersky RA, McMahon C. The relationship of hemoglobin A1C to time-in-range in patients with diabetes. Diabetes Technol Ther. 2019; 21:81–5.
Article26. Beck RW, Bergenstal RM, Cheng P, Kollman C, Carlson AL, Johnson ML, et al. The relationships between time in range, hyperglycemia metrics, and HbA1c. J Diabetes Sci Technol. 2019; 13:614–26.
Article27. Rodbard D. Glucose time in range, time above range, and time below range depend on mean or median glucose or HbA1c, glucose coefficient of variation, and shape of the glucose distribution. Diabetes Technol Ther. 2020; 22:492–500.
Article28. Monnier L, Colette C. Glycemic variability: should we and can we prevent it? Diabetes Care. 2008; 31 Suppl 2:S150–4.29. Saisho Y. Glycemic variability and oxidative stress: a link between diabetes and cardiovascular disease? Int J Mol Sci. 2014; 15:18381–406.30. Elumalai S, Karunakaran U, Moon JS, Won KC. High glucoseinduced PRDX3 acetylation contributes to glucotoxicity in pancreatic β-cells: prevention by teneligliptin. Free Radic Biol Med. 2020; 160:618–29.
Article31. Ceriello A, De Nigris V, Iijima H, Matsui T, Gouda M. The unique pharmacological and pharmacokinetic profile of teneligliptin: implications for clinical practice. Drugs. 2019; 79:733–50.
Article32. Kim YG, Hahn S, Oh TJ, Kwak SH, Park KS, Cho YM. Differences in the glucose-lowering efficacy of dipeptidyl peptidase-4 inhibitors between Asians and non-Asians: a systematic review and meta-analysis. Diabetologia. 2013; 56:696–708.
Article33. Diabetes Canada Clinical Practice Guidelines Expert Committee, Lipscombe L, Booth G, Butalia S, Dasgupta K, Eurich DT, et al. Pharmacologic glycemic management of type 2 diabetes in adults. Can J Diabetes. 2018; 42 Suppl 1:S88–103.34. Wu D, Li L, Liu C. Efficacy and safety of dipeptidyl peptidase-4 inhibitors and metformin as initial combination therapy and as monotherapy in patients with type 2 diabetes mellitus: a metaanalysis. Diabetes Obes Metab. 2014; 16:30–7.
Article35. Esposito K, Chiodini P, Maiorino MI, Capuano A, Cozzolino D, Petrizzo M, et al. A nomogram to estimate the HbA1c response to different DPP-4 inhibitors in type 2 diabetes: a systematic review and meta- analysis of 98 trials with 24 163 patients. BMJ Open. 2015; 5:e005892.36. Kwak SH, Hwang YC, Won JC, Bae JC, Kim HJ, Suh S, et al. Comparison of the effects of gemigliptin and dapagliflozin on glycaemic variability in type 2 diabetes: a randomized, openlabel, active-controlled, 12-week study (STABLE II study). Diabetes Obes Metab. 2020; 22:173–81.
Article37. Kim G, Lim S, Kwon HS, Park IB, Ahn KJ, Park CY, et al. Efficacy and safety of evogliptin treatment in patients with type 2 diabetes: a multicentre, active-controlled, randomized, doubleblind study with open-label extension (the EVERGREEN study). Diabetes Obes Metab. 2020; 22:1527–36.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Time in Range from Continuous Glucose Monitoring: A Novel Metric for Glycemic Control
- Glycemic Variability and Diabetes Mellitus
- HbA1c Variability and Micro- and Macrovascular Complications of Diabetes
- Glucose Profiles Assessed by Intermittently Scanned Continuous Glucose Monitoring System during the Perioperative Period of Metabolic Surgery
- Use of Flash Glucose Monitoring in Patients on Intensive Insulin Treatment