Korean J Gastroenterol.
2020 Dec;76(6):282-296. 10.4166/kjg.2020.142.
Clinical Practice Guideline for the Management of Antithrombotic Agents in Patients Undergoing Gastrointestinal Endoscopy
- Affiliations
-
- 1Department of Gastroenterology, Hallym University Sacred Heart Hospital, Anyang, Korea
- 2Department of Internal Medicine, Gangneung Asan Hospital, University of Ulsan College of Medicine, Gangneung, Korea
- 3Department of Gastroenterology, Samsung Medical Center, Seoul, Korea
- 4Department of Internal Medicine, Seoul National University Hospital Healthcare System Gangnam Center, Seoul, Korea
- 5Department of Internal Medicine, Seoul National University Bundang Hospital, Seongnam, Korea
- 6Department of Gastroenterology, Asan Medical Center, Seoul, Korea
- 7National Evidence-based Healthcare Collaborating Agency, Seoul, Korea
- 8Department of Gastroenterology, Chosun University Hospital, Gwangju, Korea
- 9Department of Gastroenterology, Cha University Bundang Medical Center, Seongnam, Korea
- 10Department of Gastroenterology, Jeonbuk National Hospital, Jeonju, Korea
- 11Department of Gastroenterology, Daegu Catholic University School of Medicine, Daegu, Korea
- 12Department of Gastroenterology, Korea University Anam Hospital, Seoul, Korea
- KMID: 2509705
- DOI: http://doi.org/10.4166/kjg.2020.142
Abstract
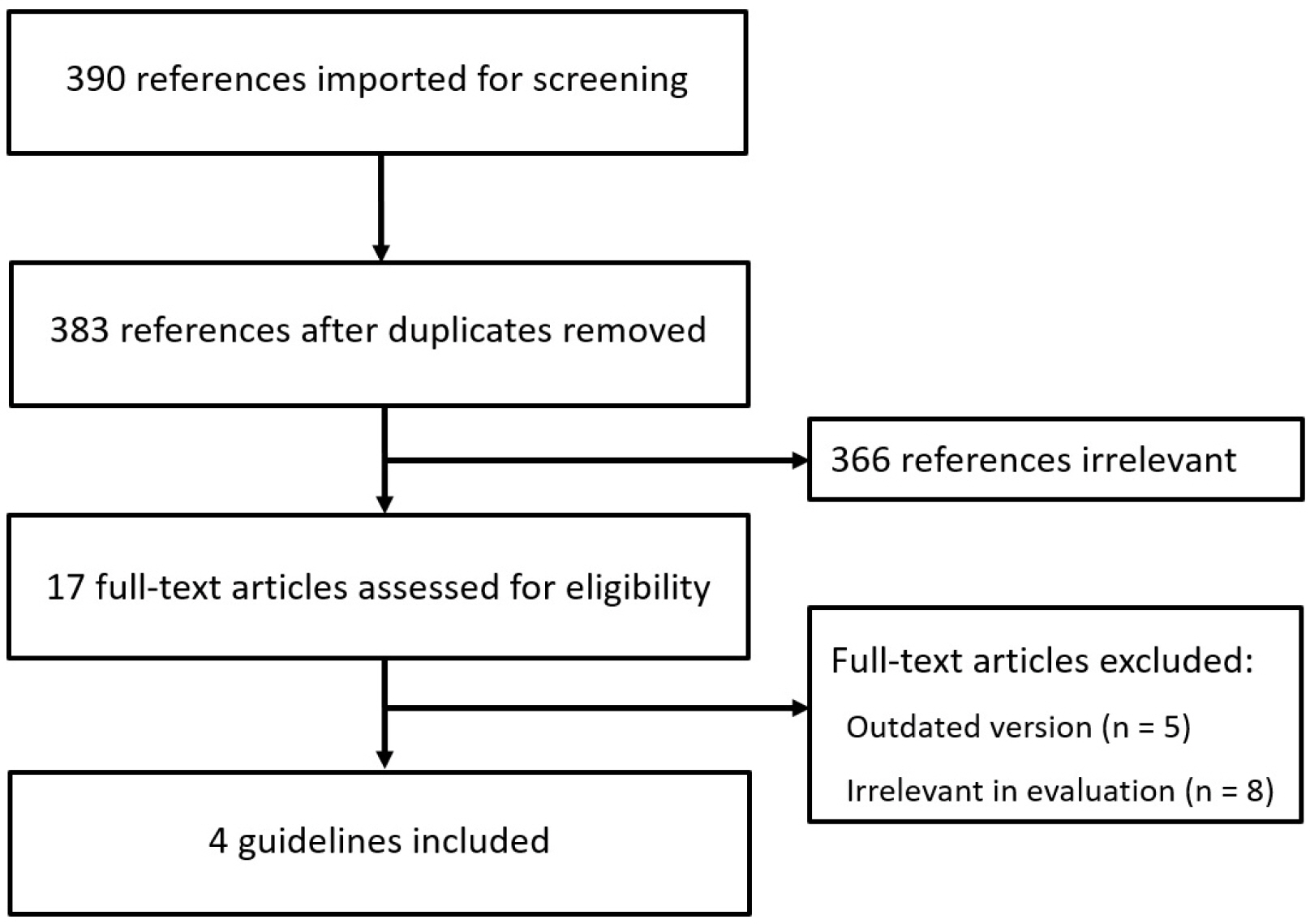
- Antithrombotic agents, including antiplatelet agents and anticoagulants, are increasingly used in South Korea. The management of patients using antithrombotic agents and requiring gastrointestinal endoscopy is an important clinical challenge. Although clinical practice guidelines (CPGs) for the management of patients receiving antithrombotic agents and undergoing gastrointestinal endoscopy have been developed in the Unites States, Europe, and Asia Pacific region, it is uncertain whether these guidelines can be adopted in South Korea. After reviewing current CPGs, we identified unmet needs and recognized significant discrepancies in the clinical practice among regions. This is the first CPG in Korea providing information that may assist endoscopists in the management of patients on antithrombotic agents who require diagnostic or elective therapeutic endoscopy. This guideline was developed through the adaptation process as an evidence-based method, with four guidelines retrieved by systematic review. Eligible guidelines were evaluated according to the Appraisal of Guidelines for Research and Evaluation II process, and 13 statements were established using a grading system. This guideline was reviewed by external experts before an official. It will be revised as necessary to cover changes in technology, evidence, or other aspects of clinical practice.
Keyword
Figure
Reference
-
1. Antithrombotic Trialists' Collaboration. 2002; Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ. 324:71–86. DOI: 10.1136/bmj.324.7329.71. PMID: 11786451. PMCID: PMC64503.
Article2. Baigent C, Blackwell L, et al. Antithrombotic Trialists' (ATT) Collaboration. 2009; Aspirin in the primary and secondary prevention of vascular disease: collaborative meta-analysis of individual participant data from randomised trials. Lancet. 373:1849–1860. DOI: 10.1016/S0140-6736(09)60503-1.3. Singer DE, Hughes RA, et al. Boston Area Anticoagulation Trial for Atrial Fibrillation Investigators. 1990; The effect of low-dose warfarin on the risk of stroke in patients with nonrheumatic atrial fibrillation. N Engl J Med. 323:1505–1511. DOI: 10.1056/NEJM199011293232201. PMID: 2233931.
Article4. van Walraven C, Hart RG, Singer DE, et al. 2002; Oral anticoagulants vs aspirin in nonvalvular atrial fibrillation: an individual patient meta-analysis. JAMA. 288:2441–2448. DOI: 10.1001/jama.288.19.2441. PMID: 12435257.5. Choi KS, Jung HY, Choi KD, et al. 2011; EMR versus gastrectomy for intramucosal gastric cancer: comparison of long-term outcomes. Gastrointest Endosc. 73:942–948. DOI: 10.1016/j.gie.2010.12.032. PMID: 21392757.
Article6. Pyo JH, Lee H, Min BH, et al. 2016; Long-term outcome of endoscopic resection vs. surgery for early gastric cancer: a non-inferioritymatched cohort study. Am J Gastroenterol. 111:240–249. DOI: 10.1038/ajg.2015.427. PMID: 26782817.7. Kim YG, Kong SH, Oh SY, et al. 2014; Effects of screening on gastric cancer management: comparative analysis of the results in 2006 and in 2011. J Gastric Cancer. 14:129–134. DOI: 10.5230/jgc.2014.14.2.129. PMID: 25061541. PMCID: PMC4105378.
Article8. Holbrook A, Schulman S, Witt DM, et al. 2012; Evidence-based management of anticoagulant therapy: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 141(Suppl 2):e152S–e184S. DOI: 10.1378/chest.141.4.1129b. PMCID: PMC3620915.9. Becker RC, Scheiman J, Dauerman HL, et al. 2009; Management of platelet-directed pharmacotherapy in patients with atherosclerotic coronary artery disease undergoing elective endoscopic gastrointestinal procedures. J Am Coll Cardiol. 54:2261–2276. DOI: 10.1016/j.jacc.2009.09.012. PMID: 19942393.
Article10. Fujimoto K, Fujishiro M, Kato M, et al. 2014; Guidelines for gastroenterological endoscopy in patients undergoing antithrombotic treatment. Dig Endosc. 26:1–14. DOI: 10.1111/den.12183. PMID: 24215155.
Article11. Acosta RD, Abraham NS, et al. ASGE Standards of Practice Committee. 2016; The management of antithrombotic agents for patients undergoing GI endoscopy. Gastrointest Endosc. 83:3–16. DOI: 10.1016/j.gie.2015.09.035. PMID: 26621548.
Article12. Veitch AM, Vanbiervliet G, Gershlick AH, et al. 2016; Endoscopy in patients on antiplatelet or anticoagulant therapy, including direct oral anticoagulants: British Society of Gastroenterology (BSG) and European Society of Gastrointestinal Endoscopy (ESGE) guidelines. Gut. 65:374–389. DOI: 10.1136/gutjnl-2015-311110. PMID: 26873868. PMCID: PMC4789831.
Article13. Chan FKL, Goh KL, Reddy N, et al. 2018; Management of patients on antithrombotic agents undergoing emergency and elective endoscopy: joint Asian Pacific Association of Gastroenterology (APAGE) and Asian Pacific Society for Digestive Endoscopy (APSDE) practice guidelines. Gut. 67:405–417. DOI: 10.1136/gutjnl-2017-315131. PMID: 29331946. PMCID: PMC5868286.
Article14. Brouwers MC, Kho ME, Browman GP, et al. 2010; AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 182:E839–E842. DOI: 10.1503/cmaj.090449. PMID: 20603348. PMCID: PMC3001530.
Article15. Higgins JPT, Altman DG, Sterne JAC. Higgins JPT, Green S, editors. c2011. Chapter 8: assessing risk of bias in included studies. Cochrane handbook for systematic reviews of interventions. Version 5.1.0 [Internet]. Cochrane;London: Available from: http://handbook-5-1.cochrane.org/. updated 2011 Mar.16. Sterne JA, Hernán MA, Reeves BC, et al. 2016; ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 355:i4919. DOI: 10.1136/bmj.i4919. PMID: 27733354. PMCID: PMC5062054.
Article17. Shea BJ, Grimshaw JM, Wells GA, et al. 2007; Development of AMSTAR: a measurement tool to assess the methodological quality of systematic reviews. BMC Med Res Methodol. 7:10. DOI: 10.1186/1471-2288-7-10. PMID: 17302989. PMCID: PMC1810543.
Article18. Guyatt GH, Oxman AD, Kunz R, et al. 2008; Incorporating considerations of resources use into grading recommendations. BMJ. 336:1170–1173. DOI: 10.1136/bmj.39504.506319.80. PMID: 18497416. PMCID: PMC2394579.
Article19. Ischemic Heart Disease Clinical Research Center. c2013. Recommendation for standard treatment for acute coronary syndrome [Internet]. Korean Society of Cardiology;Seoul: Available from: https://www.circulation.or.kr:4443/bbs/index.php?.code=m_info&category=&gubun=&page=1&number=263&-mode=view&keyfield=&key=. updated 2013 Mar 12; cited 2020 Jul 15.20. Levine GN, Bates ER, Bittl JA, et al. 2016; 2016 ACC/AHA guideline focused update on duration of dual antiplatelet therapy in patients with coronary artery disease: a report of the American College of Cardiology/American Heart Association Task Force on clinical practice guidelines: an update of the 2011 ACCF/AHA/SCAI guideline for percutaneous coronary intervention, 2011 ACCF/AHA guideline for coronary artery bypass graft surgery, 2012 ACC/AHA/ACP/AATS/PCNA/SCAI/STS guideline for the diagnosis and management of patients with stable ischemic heart disease, 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction, 2014 AHA/ACC guideline for the management of patients with non-ST-elevation acute coronary syndromes, and 2014 ACC/AHA guideline on perioperative cardiovascular evaluation and management of patients undergoing noncardiac surgery. Circulation. 134:e123–e155. DOI: 10.1161/CIR.0000000000000452.
Article21. Egholm G, Kristensen SD, Thim T, et al. 2016; Risk associated with surgery within 12 months after coronary drug-eluting stent implantation. J Am Coll Cardiol. 68:2622–2632. DOI: 10.1016/j.jacc.2016.09.967. PMID: 27978946.22. Hawn MT, Graham LA, Richman JS, Itani KM, Henderson WG, Maddox TM. 2013; Risk of major adverse cardiac events following noncardiacsurgery in patients with coronary stents. JAMA. 310:1462–1472. DOI: 10.1001/jama.2013.278787. PMID: 24101118.23. Holcomb CN, Graham LA, Richman JS, Itani KM, Maddox TM, Hawn MT. 2016; The incremental risk of coronary stents on postoperative adverse events: a matched cohort study. Ann Surg. 263:924–930. DOI: 10.1097/SLA.0000000000001246. PMID: 25894416.24. Valgimigli M, Bueno H, Byrne RA, et al. 2018; 2017 ESC focused update on dual antiplatelet therapy in coronary artery disease developed in collaboration with EACTS: The Task Force for dual anti-platelet therapy in coronary artery disease of the European Society of Cardiology (ESC) and of the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 39:213–260. DOI: 10.1093/eurheartj/ehx419. PMID: 28886622.25. Garcia DA, Regan S, Henault LE, et al. 2008; Risk of thromboembolism with short-term interruption of warfarin therapy. Arch Intern Med. 168:63–69. DOI: 10.1001/archinternmed.2007.23. PMID: 18195197.
Article26. Blacker DJ, Wijdicks EF, McClelland RL. 2003; Stroke risk in anti-coagulated patients with atrial fibrillation undergoing endoscopy. Neurology. 61:964–968. DOI: 10.1212/01.WNL.0000086817.54076.EB. PMID: 14557569.
Article27. Cappell MS, Abdullah M. 2000; Management of gastrointestinal bleeding induced by gastrointestinal endoscopy. Gastroenterol Clin North Am. 29:125–167. vi–vii. DOI: 10.1016/S0889-8553(05)70110-2.
Article28. Macrae FA, Tan KG, Williams CB. 1983; Towards safer colonoscopy: a report on the complications of 5000 diagnostic or therapeutic colonoscopies. Gut. 24:376–383. DOI: 10.1136/gut.24.5.376. PMID: 6601604. PMCID: PMC1419999.
Article29. Rogers BH, Silvis SE, Nebel OT, Sugawa C, Mandelstam P. 1975; Complications of flexible fiberoptic colonoscopy and polypectomy. Gastrointest Endosc. 22:73–77. DOI: 10.1016/S0016-5107(75)73705-7.30. Vu CK, Korman MG, Bejer I, Davis S. 1998; Gastrointestinal bleeding after cold biopsy. Am J Gastroenterol. 93:1141–1143. DOI: 10.1111/j.1572-0241.1998.346_e.x. PMID: 9672346.
Article31. Wexner SD, Garbus JE, Singh JJ. 2001; A prospective analysis of 13,580 colonoscopies. Reevaluation of credentialing guidelines. Surg Endosc. 15:251–261. DOI: 10.1007/s004640080147. PMID: 11344424.32. Parra-Blanco A, Kaminaga N, Kojima T, et al. 2000; Hemoclipping for postpolypectomy and postbiopsy colonic bleeding. Gastrointest Endosc. 51:37–41. DOI: 10.1016/S0016-5107(00)70384-1.
Article33. Sieg A, Hachmoeller-Eisenbach U, Eisenbach T. 2001; Prospective evaluation of complications in outpatient GI endoscopy: a survey among German gastroenterologists. Gastrointest Endosc. 53:620–627. DOI: 10.1067/mge.2001.114422. PMID: 11323588.
Article34. Ono S, Fujishiro M, Kodashima S, et al. 2012; Evaluation of safety of endoscopic biopsy without cessation of antithrombotic agents in Japan. J Gastroenterol. 47:770–774. DOI: 10.1007/s00535-012-0538-7. PMID: 22350697.
Article35. Whitson MJ, Dikman AE, von Althann C, et al. 2011; Is gastroduodenal biopsy safe in patients receiving aspirin and clopidogrel?: a prospective, randomized study involving 630 biopsies. J Clin Gastroenterol. 45:228–233. DOI: 10.1097/MCG.0b013e3181eb5efd. PMID: 20717045.36. Ono S, Fujishiro M, Hirano K, et al. 2009; Retrospective analysis on the management of anticoagulants and antiplatelet agents for scheduled endoscopy. J Gastroenterol. 44:1185–1189. DOI: 10.1007/s00535-009-0127-6. PMID: 19763388.
Article37. Shiffman ML, Farrel MT, Yee YS. 1994; Risk of bleeding after endoscopic biopsy or polypectomy in patients taking aspirin or other NSAIDS. Gastrointest Endosc. 40:458–462. DOI: 10.1016/S0016-5107(94)70210-1.
Article38. Hamada T, Yasunaga H, Nakai Y, et al. 2015; Bleeding after endoscopic sphincterotomy or papillary balloon dilation among users of antithrombotic agents. Endoscopy. 47:997–1004. DOI: 10.1055/s-0034-1392408. PMID: 26126160.
Article39. Bowles CJ, Leicester R, Romaya C, Swarbrick E, Williams CB, Epstein O. 2004; A prospective study of colonoscopy practice in the UK today: are we adequately prepared for national colorectal cancer screening tomorrow? Gut. 53:277–283. DOI: 10.1136/gut.2003.016436. PMID: 14724164. PMCID: PMC1774946.
Article40. Gavin DR, Valori RM, Anderson JT, Donnelly MT, Williams JG, Swarbrick ET. 2013; The national colonoscopy audit: a nationwide assessment of the quality and safety of colonoscopy in the UK. Gut. 62:242–249. DOI: 10.1136/gutjnl-2011-301848. PMID: 22661458.
Article41. Choung BS, Kim SH, Ahn DS, et al. 2014; Incidence and risk factors of delayed postpolypectomy bleeding: a retrospective cohort study. J Clin Gastroenterol. 48:784–789. DOI: 10.1097/MCG.0000000000000027. PMID: 24231934.42. Kim JH, Lee HJ, Ahn JW, et al. 2013; Risk factors for delayed post-polypectomy hemorrhage: a case-control study. J Gastroenterol Hepatol. 28:645–649. DOI: 10.1111/jgh.12132. PMID: 23369027.
Article43. Manocha D, Singh M, Mehta N, Murthy UK. 2012; Bleeding risk after invasive procedures in aspirin/NSAID users: polypectomy study in veterans. Am J Med. 125:1222–1227. DOI: 10.1016/j.amjmed.2012.05.030. PMID: 23164486.
Article44. Repici A, Hassan C, Vitetta E, et al. 2012; Safety of cold polypectomy for <10mm polyps at colonoscopy: a prospective multicenter study. Endoscopy. 44:27–31. DOI: 10.1055/s-0031-1291387. PMID: 22125197.45. Rutter MD, Nickerson C, Rees CJ, Patnick J, Blanks RG. 2014; Risk factors for adverse events related to polypectomy in the English bowel cancer screening programme. Endoscopy. 46:90–97. DOI: 10.1055/s-0033-1344987. PMID: 24477363.
Article46. Hui AJ, Wong RM, Ching JY, Hung LC, Chung SC, Sung JJ. 2004; Risk of colonoscopic polypectomy bleeding with anticoagulants and antiplatelet agents: analysis of 1657 cases. Gastrointest Endosc. 59:44–48. DOI: 10.1016/S0016-5107(03)02307-1.
Article47. Yousfi M, Gostout CJ, Baron TH, et al. 2004; Postpolypectomy lower gastrointestinal bleeding: potential role of aspirin. Am J Gastroenterol. 99:1785–1789. DOI: 10.1111/j.1572-0241.2004.30368.x. PMID: 15330919.
Article48. Lee SH, Shin SJ, Park DI, et al. 2012; Korean guideline for colonoscopic polypectomy. Clin Endosc. 45:11–24. DOI: 10.5946/ce.2012.45.1.11. PMID: 22741130. PMCID: PMC3363129.
Article49. Moon HS, Park SW, Kim DH, Kang SH, Sung JK, Jeong HY. 2014; Only the size of resected polyps is an independent risk factor for delayed postpolypectomy hemorrhage: a 10-year single-center case-control study. Ann Coloproctol. 30:182–185. DOI: 10.3393/ac.2014.30.4.182. PMID: 25210687. PMCID: PMC4155137.
Article50. Shalman D, Gerson LB. 2015; Systematic review with meta-analysis: the risk of gastrointestinal haemorrhage post-polypectomy in patients receiving anti-platelet, anti-coagulant and/or thienopyridine medications. Aliment Pharmacol Ther. 42:949–956. DOI: 10.1111/apt.13367. PMID: 26290157.
Article51. Sawhney MS, Salfiti N, Nelson DB, Lederle FA, Bond JH. 2008; Risk factors for severe delayed postpolypectomy bleeding. Endoscopy. 40:115–119. DOI: 10.1055/s-2007-966959. PMID: 18253906.
Article52. Bahin FF, Naidoo M, Williams SJ, et al. 2015; Prophylactic endoscopic coagulation to prevent bleeding after wide-field endoscopic mucosal resection of large sessile colon polyps. Clin Gastroenterol Hepatol. 13:724–730. e1-e2. DOI: 10.1016/j.cgh.2014.07.063. PMID: 25151254.
Article53. Burgess NG, Metz AJ, Williams SJ, et al. 2014; Risk factors for intra-procedural and clinically significant delayed bleeding after wide-field endoscopic mucosal resection of large colonic lesions. Clin Gastroenterol Hepatol. 12:651–661. e1-e3. DOI: 10.1016/j.cgh.2013.09.049. PMID: 24090728.
Article54. Metz AJ, Bourke MJ, Moss A, Williams SJ, Swan MP, Byth K. 2011; Factors that predict bleeding following endoscopic mucosal resection of large colonic lesions. Endoscopy. 43:506–511. DOI: 10.1055/s-0030-1256346. PMID: 21618150.
Article55. Cao Y, Liao C, Tan A, Gao Y, Mo Z, Gao F. 2009; Meta-analysis of endoscopic submucosal dissection versus endoscopic mucosal resection for tumors of the gastrointestinal tract. Endoscopy. 41:751–757. DOI: 10.1055/s-0029-1215053. PMID: 19693750.
Article56. Cho SJ, Choi IJ, Kim CG, et al. 2012; Aspirin use and bleeding risk after endoscopic submucosal dissection in patients with gastric neoplasms. Endoscopy. 44:114–121. DOI: 10.1055/s-0031-1291459. PMID: 22271021.
Article57. Dong J, Wei K, Deng J, et al. 2017; Effects of antithrombotic therapy on bleeding after endoscopic submucosal dissection. Gastrointest Endosc. 86:807–816. DOI: 10.1016/j.gie.2017.07.017. PMID: 28732709.
Article58. Koh R, Hirasawa K, Yahara S, et al. 2013; Antithrombotic drugs are risk factors for delayed postoperative bleeding after endoscopic submucosal dissection for gastric neoplasms. Gastrointest Endosc. 78:476–483. DOI: 10.1016/j.gie.2013.03.008. PMID: 23622974.
Article59. Kono Y, Obayashi Y, Baba Y, et al. 2018; Postoperative bleeding risk after gastric endoscopic submucosal dissection during antithrombotic drug therapy. J Gastroenterol Hepatol. 33:453–460. DOI: 10.1111/jgh.13872. PMID: 28696019.
Article60. Libânio D, Costa MN, Pimentel-Nunes P, Dinis-Ribeiro M. 2016; Risk factors for bleeding after gastric endoscopic submucosal dissection: a systematic review and meta-analysis. Gastrointest Endosc. 84:572–586. DOI: 10.1016/j.gie.2016.06.033. PMID: 27345132.
Article61. Lim JH, Kim SG, Kim JW, et al. 2012; Do antiplatelets increase the risk of bleeding after endoscopic submucosal dissection of gastric neoplasms? Gastrointest Endosc. 75:719–727. DOI: 10.1016/j.gie.2011.11.034. PMID: 22317881.
Article62. Sanomura Y, Oka S, Tanaka S, et al. 2014; Continued use of low-dose aspirin does not increase the risk of bleeding during or after endoscopic submucosal dissection for early gastric cancer. Gastric Cancer. 17:489–496. DOI: 10.1007/s10120-013-0305-3. PMID: 24142107. PMCID: PMC4072060.
Article63. Arimoto J, Higurashi T, Chiba H, et al. 2018; Continued use of a single antiplatelet agent does not increase the risk of delayed bleeding after colorectal endoscopic submucosal dissection. Dig Dis Sci. 63:218–227. DOI: 10.1007/s10620-017-4843-0. PMID: 29177848. PMCID: PMC5760603.
Article64. Ninomiya Y, Oka S, Tanaka S, et al. 2015; Risk of bleeding after endoscopic submucosal dissection for colorectal tumors in patients with continued use of low-dose aspirin. J Gastroenterol. 50:1041–1046. DOI: 10.1007/s00535-015-1053-4. PMID: 25682173.
Article65. Biondi-Zoccai GG, Lotrionte M, Agostoni P, et al. 2006; A systematic review and meta-analysis on the hazards of discontinuing or not adhering to aspirin among 50,279 patients at risk for coronary artery disease. Eur Heart J. 27:2667–2674. DOI: 10.1093/eurheartj/ehl334. PMID: 17053008.66. Maulaz AB, Bezerra DC, Michel P, Bogousslavsky J. 2005; Effect of discontinuing aspirin therapy on the risk of brain ischemic stroke. Arch Neurol. 62:1217–1220. DOI: 10.1001/archneur.62.8.1217. PMID: 16087761.
Article67. Oscarsson A, Gupta A, Fredrikson M, et al. 2010; To continue or discontinue aspirin in the perioperative period: a randomized, controlled clinical trial. Br J Anaesth. 104:305–312. DOI: 10.1093/bja/aeq003. PMID: 20150346.
Article68. Cotton PB, Lehman G, Vennes J, et al. 1991; Endoscopic sphincterotomy complications and their management: an attempt at consensus. Gastrointest Endosc. 37:383–393. DOI: 10.1016/S0016-5107(91)70740-2.
Article69. Freeman ML, Nelson DB, Sherman S, et al. 1996; Complications of endoscopic biliary sphincterotomy. N Engl J Med. 335:909–918. DOI: 10.1056/NEJM199609263351301. PMID: 8782497.
Article70. Hui CK, Lai KC, Yuen MF, Wong WM, Lam SK, Lai CL. 2002; Does withholding aspirin for one week reduce the risk of post-sphincterotomy bleeding? Aliment Pharmacol Ther. 16:929–936. DOI: 10.1046/j.1365-2036.2002.01251.x. PMID: 11966501.71. Hussain N, Alsulaiman R, Burtin P, et al. 2007; The safety of endoscopic sphincterotomy in patients receiving antiplatelet agents: a case-control study. Aliment Pharmacol Ther. 25:579–584. DOI: 10.1111/j.1365-2036.2006.03225.x. PMID: 17305758.72. Onal IK, Parlak E, Akdogan M, et al. 2013; Do aspirin and non-steroidal anti-inflammatory drugs increase the risk of post-sphincterotomy hemorrhage--a case-control study. Clin Res Hepatol Gastroenterol. 37:171–176. DOI: 10.1016/j.clinre.2012.04.010. PMID: 22677232.73. Nelson DB, Freeman ML. 1994; Major hemorrhage from endoscopic sphincterotomy: risk factor analysis. J Clin Gastroenterol. 19:283–287. DOI: 10.1097/00004836-199412000-00004. PMID: 7876506.
Article74. Hwang JH, Kang DH, Kim HW, Choi CW, Park SB. 2011; The safety of endoscopic sphincterotomy in patients taking aspirin. Korean J Med. 81:193–198.75. Feagins LA, Iqbal R, Harford WV, et al. 2013; Low rate of postpolypectomy bleeding among patients who continue thienopyridine therapy during colonoscopy. Clin Gastroenterol Hepatol. 11:1325–1332. DOI: 10.1016/j.cgh.2013.02.003. PMID: 23403011.
Article76. Gandhi S, Narula N, Mosleh W, Marshall JK, Farkouh M. 2013; Meta-analysis: colonoscopic post-polypectomy bleeding in patients on continued clopidogrel therapy. Aliment Pharmacol Ther. 37:947–952. DOI: 10.1111/apt.12292. PMID: 23530880.
Article77. Singh M, Mehta N, Murthy UK, Kaul V, Arif A, Newman N. 2010; Postpolypectomy bleeding in patients undergoing colonoscopy on uninterrupted clopidogrel therapy. Gastrointest Endosc. 71:998–1005. DOI: 10.1016/j.gie.2009.11.022. PMID: 20226452.
Article78. Lee MG, Kim J, Lee SH, et al. 2014; Effect of sustained use of platelet aggregation inhibitors on post-endoscopic sphincterotomy bleeding. Dig Endosc. 26:737–744. DOI: 10.1111/den.12271. PMID: 24673356.
Article79. Oh S, Kim SG, Kim J, et al. 2018; Continuous use of thienopyridine may be as safe as low-dose aspirin in endoscopic resection of gastric tumors. Gut Liver. 12:393–401. DOI: 10.5009/gnl17384. PMID: 29429155. PMCID: PMC6027827.
Article80. Eisenberg MJ, Richard PR, Libersan D, Filion KB. 2009; Safety of short-term discontinuation of antiplatelet therapy in patients with drug-eluting stents. Circulation. 119:1634–1642. DOI: 10.1161/CIRCULATIONAHA.108.813667. PMID: 19289638.
Article81. Hong SJ, Kim MJ, Kim JS, et al. 2019; Effect of perioperative antiplatelet therapy on outcomes in patients with drug-eluting stents undergoing elective noncardiac surgery. Am J Cardiol. 123:1414–1421. DOI: 10.1016/j.amjcard.2019.02.004. PMID: 30770090.
Article82. Choudari CP, Rajgopal C, Palmer KR. 1994; Acute gastrointestinal haemorrhage in anticoagulated patients: diagnoses and response to endoscopic treatment. Gut. 35:464–466. DOI: 10.1136/gut.35.4.464. PMID: 8174982. PMCID: PMC1374792.
Article83. Igarashi K, Takizawa K, Kakushima N, et al. 2017; Should antithrombotic therapy be stopped in patients undergoing gastric endoscopic submucosal dissection? Surg Endosc. 31:1746–1753. DOI: 10.1007/s00464-016-5167-4. PMID: 27530896.
Article84. Witt DM, Delate T, McCool KH, et al. 2009; Incidence and predictors of bleeding or thrombosis after polypectomy in patients receiving and not receiving anticoagulation therapy. J Thromb Haemost. 7:1982–1989. DOI: 10.1111/j.1538-7836.2009.03598.x. PMID: 19719825.
Article85. White RH, McKittrick T, Hutchinson R, Twitchell J. 1995; Temporary discontinuation of warfarin therapy: changes in the international normalized ratio. Ann Intern Med. 122:40–42. DOI: 10.7326/0003-4819-122-1-199501010-00006. PMID: 7985894.
Article86. Schulman S, Elbazi R, Zondag M, O'Donnell M. 2008; Clinical factors influencing normalization of prothrombin time after stopping warfarin: a retrospective cohort study. Thromb J. 6:15. DOI: 10.1186/1477-9560-6-15. PMID: 18925967. PMCID: PMC2586623.
Article87. Horiuchi A, Nakayama Y, Kajiyama M, Tanaka N, Sano K, Graham DY. 2014; Removal of small colorectal polyps in anticoagulated patients: a prospective randomized comparison of cold snare and conventional polypectomy. Gastrointest Endosc. 79:417–423. DOI: 10.1016/j.gie.2013.08.040. PMID: 24125514.88. Joung B, Lee JM, Lee KH, et al. 2018; 2018 Korean guideline of atrial fibrillation management. Korean Circ J. 48:1033–1080. DOI: 10.4070/kcj.2018.0339. PMID: 30403013. PMCID: PMC6221873.
Article89. Douketis JD, Spyropoulos AC, Kaatz S, et al. 2015; Perioperative bridging anticoagulation in patients with strial fibrillation. N Engl J Med. 373:823–833. DOI: 10.1056/NEJMoa1501035. PMID: 26095867. PMCID: PMC4931686.90. Friedland S, Sedehi D, Soetikno R. 2009; Colonoscopic polypectomy in anticoagulated patients. World J Gastroenterol. 15:1973–1976. DOI: 10.3748/wjg.15.1973. PMID: 19399929. PMCID: PMC2675087.
Article91. Timothy SK, Hicks TC, Opelka FG, Timmcke AE, Beck DE. 2001; Colonoscopy in the patient requiring anticoagulation. Dis Colon Rectum. 44:1845–1848. discussion, 1848-1849. DOI: 10.1007/BF02234465. PMID: 11742172.
Article92. Ara N, Iijima K, Maejima R, et al. 2015; Prospective analysis of risk for bleeding after endoscopic biopsy without cessation of antithrombotics in Japan. Dig Endosc. 27:458–464. DOI: 10.1111/den.12407. PMID: 25425518.93. Kato M, Uedo N, Hokimoto S, et al. 2018; Guidelines for gastroenterological endoscopy in patients undergoing antithrombotic treatment: 2017 appendix on anticoagulants including direct oral anticoagulants. Dig Endosc. 30:433–440. DOI: 10.1111/den.13184. PMID: 29733468.94. Lee JH, Lim HE, Lim WH, et al. 2019; The 2018 Korean heart rhythm society practical guidelines on the use of non-vitamin K-antagonist oral anticoagulants: bleeding control and perioperative management. Korean J Med. 94:40–56. DOI: 10.3904/kjm.2019.94.1.40.
Article95. Trociz IF, Tillmann C, Liesenfeld KH, Schäfer HG, Stangier J. 2007; Population pharmacokinetic analysis of the new oral thrombin inhibitor dabigatran etexilate (BIBR 1048) in patients undergoing primary elective total hip replacement surgery. J Clin Pharmacol. 47:371–382. DOI: 10.1177/0091270006297228. PMID: 17322149.96. Heidbuchel H, Verhamme P, Alings M, et al. 2017; Updated European Heart Rhythm Association practical guide on the use of non-vitamin-K antagonist anticoagulants in patients with non-valvular atrial fibrillation: executive summary. Eur Heart J. 38:2137–2149. DOI: 10.1093/eurheartj/ehw058. PMID: 27282612. PMCID: PMC5837231.
Article97. Dunn A. 2006; Perioperative management of oral anticoagulation: when and how to bridge. J Thromb Thrombolysis. 21:85–89. DOI: 10.1007/s11239-006-5582-9. PMID: 16475048.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Management of antithrombotic agents for gastrointestinal endoscopy
- Management of Patients on Antithrombotic Agents Undergoing Endoscopy
- Clinical Practice Guideline for the Management of Antithrombotic Agents in Patients Undergoing Gastrointestinal Endoscopy
- Management of antithrombotic agents and current issues in patients undergoing endoscopic submucosal dissection
- IDEN Consensus on Management of Antithrombotic Agents in Patients Undergoing Gastrointestinal Endoscopy