Korean J Radiol.
2020 Feb;21(2):218-227. 10.3348/kjr.2019.0232.
B-Value Optimization in the Estimation of Intravoxel Incoherent Motion Parameters in Patients with Cervical Cancer
- Affiliations
-
- 1Department of Diagnostic Radiology, The University of Hong Kong, Hong Kong. eyplee77@hku.hk
- 2Institute of Diagnostic and Interventional Radiology, University Hospital of Zurich, Switzerland.
- KMID: 2471639
- DOI: http://doi.org/10.3348/kjr.2019.0232
Abstract
OBJECTIVE
This study aimed to find the optimal number of b-values for intravoxel incoherent motion (IVIM) imaging analysis, using simulated and in vivo data from cervical cancer patients.
MATERIALS AND METHODS
Simulated data were generated using literature pooled means, which served as reference values for simulations. In vivo data from 100 treatment-naïve cervical cancer patients with IVIM imaging (13 b-values, scan time, 436 seconds) were retrospectively reviewed. A stepwise b-value fitting algorithm calculated optimal thresholds. Feed forward selection determined the optimal subsampled b-value distribution for biexponential IVIM fitting, and simplified IVIM modeling using monoexponential fitting was attempted. IVIM parameters computed using all b-values served as reference values for in vivo data.
RESULTS
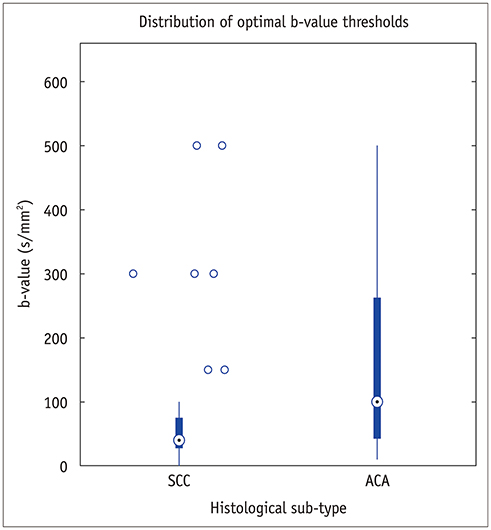
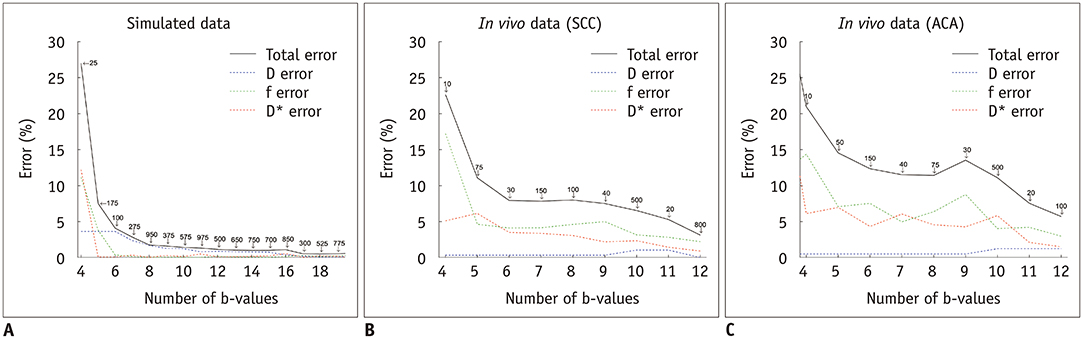
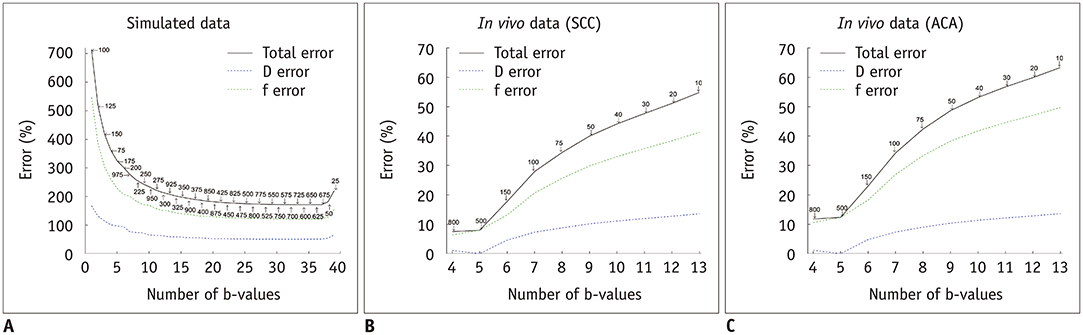
In simulations, parameters were accurately estimated with six b-values, or three b-values for simplified IVIM, respectively. In vivo data showed that the optimal threshold was 40 s/mm² for patients with squamous cell carcinoma and a subsampled acquisition of six b-values (scan time, 198 seconds) estimated parameters were not significantly different from reference parameters (individual parameter error rates of less than 5%). In patients with adenocarcinoma, the optimal threshold was 100 s/mm², but an optimal subsample could not be identified. Irrespective of the histological subtype, only three b-values were needed for simplified IVIM, but these parameters did not retain their discriminative ability.
CONCLUSION
Subsampling of six b-values halved the IVIM scan time without significant losses in accuracy and discriminative ability. Simplified IVIM is possible with only three b-values, at the risk of losing diagnostic information.
Keyword
MeSH Terms
Figure
Reference
-
1. McVeigh PZ, Syed AM, Milosevic M, Fyles A, Haider MA. Diffusion-weighted MRI in cervical cancer. Eur Radiol. 2008; 18:1058–1064.
Article2. Xue HD, Li S, Sun F, Sun HY, Jin ZY, Yang JX, et al. Clinical application of body diffusion weighted MR imaging in the diagnosis and preoperative N staging of cervical cancer. Chin Med Sci J. 2008; 23:133–137.
Article3. Patterson DM, Padhani AR, Collins DJ. Technology insight: water diffusion MRI—a potential new biomarker of response to cancer therapy. Nat Clin Pract Oncol. 2008; 5:220–233.
Article4. Koh DM, Takahara T, Imai Y, Collins DJ. Practical aspects of assessing tumors using clinical diffusion-weighted imaging in the body. Magn Reson Med Sci. 2007; 6:211–224.
Article5. Harry VN, Semple SI, Gilbert FJ, Parkin DE. Diffusion-weighted magnetic resonance imaging in the early detection of response to chemoradiation in cervical cancer. Gynecol Oncol. 2008; 111:213–220.
Article6. Kallehauge JF, Tanderup K, Haack S, Nielsen T, Muren LP, Fokdal L, et al. Apparent diffusion coefficient (ADC) as a quantitative parameter in diffusion weighted MR imaging in gynecologic cancer: dependence on b-values used. Acta Oncol. 2010; 49:1017–1022.
Article7. Lee EY, Yu X, Chu MM, Ngan HY, Siu SW, Soong IS, et al. Perfusion and diffusion characteristics of cervical cancer based on intraxovel incoherent motion MR imaging-a pilot study. Eur Radiol. 2014; 24:1506–1513.
Article8. Le Bihan D, Breton E, Lallemand D, Aubin ML, Vignaud J, Laval-Jeantet M. Separation of diffusion and perfusion in intravoxel incoherent motion MR imaging. Radiology. 1988; 168:497–505.
Article9. Lee EY, Hui ES, Chan KK, Tse KY, Kwong WK, Chang TY, et al. Relationship between intravoxel incoherent motion diffusion-weighted MRI and dynamic contrast-enhanced MRI in tissue perfusion of cervical cancers. J Magn Reson Imaging. 2015; 42:454–459.
Article10. Zhu L, Zhu L, Shi H, Wang H, Yan J, Liu B, et al. Evaluating early response of cervical cancer under concurrent chemo-radiotherapy by intravoxel incoherent motion MR imaging. BMC Cancer. 2016; 16:79.
Article11. Lee EYP, Perucho JAU, Vardhanabhuti V, He J, Siu SWK, Ngu SF, et al. Intravoxel incoherent motion MRI assessment of chemoradiation-induced pelvic bone marrow changes in cervical cancer and correlation with hematological toxicity. J Magn Reson Imaging. 2017; 46:1491–1498.
Article12. Zhu L, Wang H, Zhu L, Meng J, Xu Y, Liu B, et al. Predictive and prognostic value of intravoxel incoherent motion (IVIM) MR imaging in patients with advanced cervical cancers undergoing concurrent chemo-radiotherapy. Sci Rep. 2017; 7:11635.
Article13. Ogura A, Tamura T, Ozaki M, Doi T, Fujimoto K, Miyati T, et al. Apparent diffusion coefficient value is not dependent on magnetic resonance systems and field strength under fixed imaging parameters in brain. J Comput Assist Tomogr. 2015; 39:760–765.
Article14. Dale BM, Braithwaite AC, Boll DT, Merkle EM. Field strength and diffusion encoding technique affect the apparent diffusion coefficient measurements in diffusion-weighted imaging of the abdomen. Invest Radiol. 2010; 45:104–108.
Article15. Habermann CR, Gossrau P, Kooijman H, Graessner J, Cramer MC, Kaul MG, et al. Monitoring of gustatory stimulation of salivary glands by diffusion-weighted MR imaging: comparison of 1.5T and 3T. AJNR Am J Neuroradiol. 2007; 28:1547–1551.
Article16. Matsuoka A, Minato M, Harada M, Kubo H, Bandou Y, Tangoku A, et al. Comparison of 3.0-and 1.5-tesla diffusion-weighted imaging in the visibility of breast cancer. Radiat Med. 2008; 26:15–20.
Article17. Ogura A, Hatano I, Osakabe K, Yamaguchi N, Koyama D, Watanabe H. Importance of fractional b value for calculating apparent diffusion coefficient in DWI. AJR Am J Roentgenol. 2016; 207:1239–1243.
Article18. Freiman M, Voss SD, Mulkern RV, Perez-Rossello JM, Callahan MJ, Warfield SK. In vivo assessment of optimal b-value range for perfusion-insensitive apparent diffusion coefficient imaging. Med Phys. 2012; 39:4832–4839.19. Wurnig MC, Donati OF, Ulbrich E, Filli L, Kenkel D, Thoeny HC, et al. Systematic analysis of the intravoxel incoherent motion threshold separating perfusion and diffusion effects: proposal of a standardized algorithm. Magn Reson Med. 2015; 74:1414–1422.
Article20. Becker AS, Perucho JA, Wurnig MC, Boss A, Ghafoor S, Khong PL, et al. Assessment of cervical cancer with a parameter-free intravoxel incoherent motion imaging algorithm. Korean J Radiol. 2017; 18:510–518.
Article21. Pang Y, Turkbey B, Bernardo M, Kruecker J, Kadoury S, Merino MJ, et al. Intravoxel incoherent motion MR imaging for prostate cancer: an evaluation of perfusion fraction and diffusion coefficient derived from different b-value combinations. Magn Reson Med. 2013; 69:553–562.22. Koh DM, Collins DJ, Orton MR. Intravoxel incoherent motion in body diffusion-weighted MRI: reality and challenges. AJR Am J Roentgenol. 2011; 196:1351–1361.
Article23. Chen W, Zhang J, Long D, Wang Z, Zhu JM. Optimization of intra-voxel incoherent motion measurement in diffusion-weighted imaging of breast cancer. J Appl Clin Med Phys. 2017; 18:191–199.
Article24. Lemke A, Stieltjes B, Schad LR, Laun FB. Toward an optimal distribution of b values for intravoxel incoherent motion imaging. Magn Reson Imaging. 2011; 29:766–776.
Article25. Jambor I, Merisaari H, Aronen HJ, Järvinen J, Saunavaara J, Kauko T, et al. Optimization of b-value distribution for biexponential diffusion-weighted MR imaging of normal prostate. J Magn Reson Imaging. 2014; 39:1213–1222.
Article26. Dyvorne H, Jajamovich G, Kakite S, Kuehn B, Taouli B. Intravoxel incoherent motion diffusion imaging of the liver: optimal b-value subsampling and impact on parameter precision and reproducibility. Eur J Radiol. 2014; 83:2109–2113.
Article27. Sasaki M, Sumi M, Eida S, Katayama I, Hotokezaka Y, Nakamura T. Simple and reliable determination of intravoxel incoherent motion parameters for the differential diagnosis of head and neck tumors. PLoS One. 2014; 9:e112866.
Article28. Conklin J, Heyn C, Roux M, Cerny M, Wintermark M, Federau C. A simplified model for intravoxel incoherent motion perfusion imaging of the brain. AJNR Am J Neuroradiol. 2016; 37:2251–2257.
Article29. Pieper CC, Sprinkart AM, Meyer C, König R, Schild HH, Kukuk GM, et al. Evaluation of a simplified intravoxel incoherent motion (IVIM) analysis of diffusion-weighted imaging for prediction of tumor size changes and imaging response in breast cancer liver metastases undergoing radioembolization: a retrospective single center analysis. Medicine (Baltimore). 2016; 95:e3275.30. Pekar J, Moonen CTW, van Zijl PCM. On the precision of diffusion/perfusion imaging by gradient sensitization. Magn Reson Med. 1992; 23:122–129.
Article31. Concia M, Sprinkart AM, Penner AH, Brossart P, Gieseke J, Schild HH, et al. Diffusion-weighted magnetic resonance imaging of the pancreas: diagnostic benefit from an intravoxel incoherent motion model-based 3 b-value analysis. Invest Radiol. 2014; 49:93–100.32. Lin M, Yu X, Chen Y, Ouyang H, Wu B, Zheng D, et al. Contribution of mono-exponential, bi-exponential and stretched exponential model-based diffusion-weighted MR imaging in the diagnosis and differentiation of uterine cervical carcinoma. Eur Radiol. 2017; 27:2400–2410.
Article33. Zhou Y, Liu J, Liu C, Jia J, Li N, Xie L, et al. Intravoxel incoherent motion diffusion weighted MRI of cervical cancer—Correlated with tumor differentiation and perfusion. Magn Reson Imaging. 2016; 34:1050–1056.34. Wu Q, Wang Y, Shi L, Dong L, Liu M, Dou S, et al. Intravoxel incoherent motion diffusion-weighted magnetic resonance imaging of cervical cancer with different b-values. J Comput Assist Tomogr. 2017; 41:592–598.
Article35. Becker AS, Ghafoor S, Marcon M, Perucho JA, Wurnig MC, Wagner MW, et al. MRI texture features may predict differentiation and nodal stage of cervical cancer: a pilot study. Acta Radiol Open. 2017; 6:2058460117729574.
Article36. Lai AYT, Perucho JAU, Xu X, Hui ES, Lee EYP. Concordance of FDG PET/CT metabolic tumour volume versus DW-MRI functional tumour volume with T2-weighted anatomical tumour volume in cervical cancer. BMC Cancer. 2017; 17:825.
Article37. Le Bihan D, Turner R, MacFall JR. Effects of intravoxel incoherent motions (IVIM) in steady-state free precession (SSFP) imaging: application to molecular diffusion imaging. Magn Reson Med. 1989; 10:324–337.38. Sigmund EE, Cho GY, Kim S, Finn M, Moccaldi M, Jensen JH, et al. Intravoxel incoherent motion imaging of tumor microenvironment in locally advanced breast cancer. Magn Reson Med. 2011; 65:1437–1447.
Article39. Chandarana H, Lee VS, Hecht E, Taouli B, Sigmund EE. Comparison of biexponential and monoexponential model of diffusion weighted imaging in evaluation of renal lesions: preliminary experience. Invest Radiol. 2011; 46:285–291.40. Tan PN, Steinbach M, Kumar V. Introduction to data mining. 1st ed. Boston, MA: Pearson Addison Wesley;2005.41. Wurnig MC, Germann M, Boss A. Is there evidence for more than two diffusion components in abdominal organs?–A magnetic resonance imaging study in healthy volunteers. NMR Biomed. 2018; 31:e3852.42. Molmenti EP, Levy MF, Molmenti H, Casey D, Fasola CG, Hamilton WM, et al. Correlation between intraoperative blood flows and hepatic artery strictures in liver transplantation. Liver Transpl. 2002; 8:160–163.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Longitudinal Assessment of Intravoxel Incoherent Motion Diffusion Weighted Imaging in Evaluating the Radio-sensitivity of Nasopharyngeal Carcinoma Treated with Intensity-Modulated Radiation Therapy
- Comparative Study between ZOOMit and Conventional Intravoxel Incoherent Motion MRI for Assessing Parotid Gland Abnormalities in Patients with Early- or Mid-Stage Sjögren’s Syndrome
- Correlation of the Speed of Enhancement of Hepatic Hemangiomas with Intravoxel Incoherent Motion MR Imaging
- Intravoxel Incoherent Motion MR Imaging in the Head and Neck: Correlation with Dynamic Contrast-Enhanced MR Imaging and Diffusion-Weighted Imaging
- Assessment of Cervical Cancer with a Parameter-Free Intravoxel Incoherent Motion Imaging Algorithm