Ann Surg Treat Res.
2020 Feb;98(2):89-95. 10.4174/astr.2020.98.2.89.
Is platelet-rich plasma improves the anastomotic healing in hyperthermic intraperitoneal chemotherapy with oxaliplatin: an experimental rat study
- Affiliations
-
- 1Department of General Surgery, University of Healthy Sciences, Samsun Research and Training Hospital, Samsun, Turkey. sonmezdr@gmail.com
- 2Department of Animal Nutrition and Nutritional Diseases, Faculty of Veterinary Medicine, Ondokuz Mayis University, Samsun, Turkey.
- 3Department of Biochemistry, Faculty of Medicine, Ondokuz Mayis University, Samsun, Turkey.
- 4Department of Pathology, University of Healthy Sciences, Samsun Research and Training Hospital, Samsun, Turkey.
- KMID: 2469108
- DOI: http://doi.org/10.4174/astr.2020.98.2.89
Abstract
- PURPOSE
Hyperthermic intraperitoneal chemotherapy (HIPEC) is a novel treatment option for peritoneal surface malignancies. Due to cytotoxic effects of chemotherapeutic agents, anastomosis healing can be impaired and lead to leakage rates higher than conventional intestinal surgery. In this experimental study, we aimed to investigate the effects of platelet-rich plasma (PRP) on colonic anastomosis in rats that received HIPEC with oxaliplatin.
METHODS
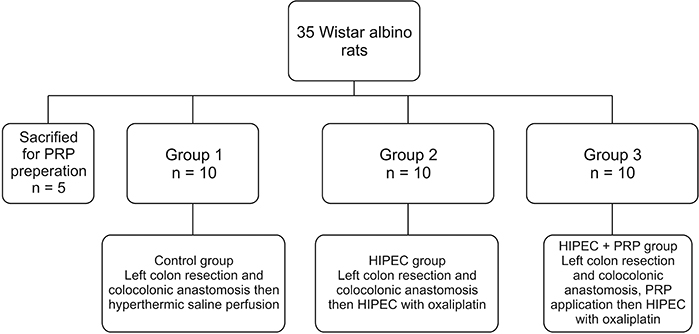
Thirty rats were divided into 3 groups. Group 1 was determined as control group and hyperthermic saline perfusion was performed after colon anastomosis. In group 2, colon anastomosis then hyperthermic oxaliplatin perfusion was performed. In the last group, the colonic anastomosis was enhanced by PRP gel and then hyperthermic oxaliplatin perfusion was performed. All the rats were reoperated on postoperative day 7 and anastomotic bursting pressure values were recorded. Tissue samples were taken for hydroxyproline assay and histopathological examination.
RESULTS
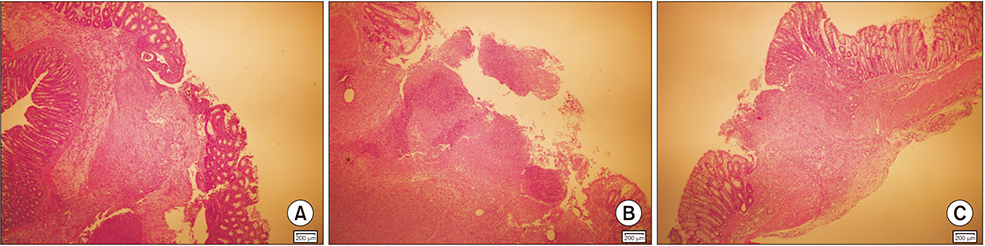
Control group had higher anastomotic bursting pressure value than group 2 and group 3 (P < 0.001). There were significant differences in anastomotic bursting pressure between groups 2 and 3 (P < 0.001). Group 2 had significantly lower hydroxyproline levels than group 3 and control group (P < 0.001). Histopathological examination revealed that PRP application reduced inflammatory response.
CONCLUSION
PRP application on colonic anastomosis improves anastomotic healing and can reduce anastomosis related complications and stoma creation; though further clinical studies are needed.
MeSH Terms
Figure
Reference
-
1. Aoyagi T, Terracina KP, Raza A, Takabe K. Current treatment options for colon cancer peritoneal carcinomatosis. World J Gastroenterol. 2014; 20:12493–12500.
Article2. Nadler A, McCart JA, Govindarajan A. Peritoneal carcinomatosis from colon cancer: a systematic review of the data for cytoreduction and intraperitoneal chemotherapy. Clin Colon Rectal Surg. 2015; 28:234–246.
Article3. Nagata H, Ishihara S, Hata K, Murono K, Kaneko M, Yasuda K, et al. Survival and prognostic factors for metachronous peritoneal metastasis in patients with colon cancer. Ann Surg Oncol. 2017; 24:1269–1280.
Article4. Bhatt A, Bhamre R, Rohila J, Kalikar V, Desouza A, Saklani A. Patients with extensive regional lymph node involvement (pN2) following potentially curative surgery for colorectal cancer are at increased risk for developing peritoneal metastases: a retrospective single-institution study. Colorectal Dis. 2019; 21:287–296.
Article5. Glehen O, Kwiatkowski F, Sugarbaker PH, Elias D, Levine EA, De Simone M, et al. Cytoreductive surgery combined with perioperative intraperitoneal chemotherapy for the management of peritoneal carcinomatosis from colorectal cancer: a multi-institutional study. J Clin Oncol. 2004; 22:3284–3292.
Article6. Verwaal VJ, van Ruth S, Witkamp A, Boot H, van Slooten G, Zoetmulder FA. Long-term survival of peritoneal carcinomatosis of colorectal origin. Ann Surg Oncol. 2005; 12:65–71.
Article7. Casado-Adam A, Alderman R, Stuart OA, Chang D, Sugarbaker PH. Gastrointestinal complications in 147 consecutive patients with peritoneal surface malignancy treated by cytoreductive surgery and perioperative intraperitoneal chemotherapy. Int J Surg Oncol. 2011; 2011:468698.
Article8. Hompes D, D’Hoore A, Van Cutsem E, Fieuws S, Ceelen W, Peeters M, et al. The treatment of peritoneal carcinomatosis of colorectal cancer with complete cytoreductive surgery and hyperthermic intraperitoneal peroperative chemotherapy (HIPEC) with oxaliplatin: a Belgian multicentre prospective phase II clinical study. Ann Surg Oncol. 2012; 19:2186–2194.
Article9. Gervais MK, Dube P, McConnell Y, Drolet P, Mitchell A, Sideris L. Cytoreductive surgery plus hyperthermic intraperitoneal chemotherapy with oxaliplatin for peritoneal carcinomatosis arising from colorectal cancer. J Surg Oncol. 2013; 108:438–443.
Article10. Riss S, Chandrakumaran K, Dayal S, Cecil TD, Mohamed F, Moran BJ. Risk of definitive stoma after surgery for peritoneal malignancy in 958 patients: comparative study between complete cytoreductive surgery and maximal tumor debulking. Eur J Surg Oncol. 2015; 41:392–395.
Article11. Whealon MD, Gahagan JV, Sujatha-Bhaskar S, O’Leary MP, Selleck M, Dumitra S, et al. Is fecal diversion needed in pelvic anastomoses during hyperthermic intraperitoneal chemotherapy (HIPEC)? Ann Surg Oncol. 2017; 24:2122–2128.
Article12. Yamaguchi R, Terashima H, Yoneyama S, Tadano S, Ohkohchi N. Effects of platelet-rich plasma on intestinal anastomotic healing in rats: PRP concentration is a key factor. J Surg Res. 2012; 173:258–266.
Article13. Sozutek A, Colak T, Cetinkunar S, Reyhan E, Irkorucu O, Polat G, et al. The effect of platelet-rich-plasma on the healing of left colonic anastomosis in a rat model of intra-abdominal sepsis. J Invest Surg. 2016; 29:294–301.
Article14. Alves R, Grimalt R. A review of platelet-rich plasma: history, biology, mechanism of action, and classification. Skin Appendage Disord. 2018; 4:18–24.
Article15. Verhofstad MH, Lange WP, van der Laak JA, Verhofstad AA, Hendriks T. Microscopic analysis of anastomotic healing in the intestine of normal and diabetic rats. Dis Colon Rectum. 2001; 44:423–431.
Article16. Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951; 193:265–275.
Article17. Strebel K, Nielsen SR, Biagini M, Qvist N. Effect of Humira® on intestinal anastomotic response in rabbits. J Invest Surg. 2015; 28:167–172.
Article18. Aghayeva A, Benlice C, Bilgin IA, Atukeren P, Dogusoy G, Demir F, et al. The effects of hyperthermic intraperitoneal chemoperfusion on colonic anastomosis: an experimental study in a rat model. Tumori. 2017; 103:307–313.
Article19. von Breitenbuch P, Piso P, Schlitt HJ. Safety of rectum anastomosis after cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. J Surg Oncol. 2018; 118:551–556.20. Deraco M, Glehen O, Helm CW, Morris DL, van der Speeten K. Cytoreductive surgery & perioperative chemotherapy for peritoneal surface malignancy: textbook and video atlas. Woodbury (CT): Cine-Med Publishing Inc.;2013.21. Braumann C, Muller V, Knies M, Aufmesser B, Schwenk W, Koplin G. Quality of life and need for care in patients with an ostomy: a survey of 2647 patients of the Berlin OStomy-Study (BOSS). Langenbecks Arch Surg. 2016; 401:1191–1201.
Article22. Doud AN, Levine EA, Fino NF, Stewart JH, Shen P, Votanopoulos KI. Stoma creation and reversal after cytoreductive surgery with hyperthermic intraperitoneal chemotherapy. Ann Surg Oncol. 2016; 23:503–510.
Article23. Yol S, Tekin A, Yilmaz H, Kucukkartallar T, Esen H, Caglayan O, et al. Effects of platelet rich plasma on colonic anastomosis. J Surg Res. 2008; 146:190–194.
Article24. Esat Duymus M, Temel S, Ozer H, Kemal Urhan M, Kaya F, Aslan F, et al. Comparison of the effects of plateletrich plasma prepared in various forms on the healing of dermal wounds in rats. Wounds. 2016; 28:99–108.25. Casella G, Soricelli E, Genco A, Ferrazza G, Basso N, Redler A. Use of platelet-rich plasma to reinforce the staple line during laparoscopic sleeve gastrectomy: feasibility study and preliminary outcome. J Laparoendosc Adv Surg Tech A. 2015; 25:222–227.
Article26. Hyman N, Manchester TL, Osler T, Burns B, Cataldo PA. Anastomotic leaks after intestinal anastomosis: it’s later than you think. Ann Surg. 2007; 245:254–258.27. Zhou B, Ren J, Ding C, Wu Y, Chen J, Wang G, et al. Protection of colonic anastomosis with platelet-rich plasma gel in the open abdomen. Injury. 2014; 45:864–868.
Article28. Schiff A, Roy S, Pignot M, Ghosh SK, Fegelman EJ. Diagnosis and management of intraoperative colorectal anastomotic leaks: a global retrospective patient chart review study. Surg Res Pract. 2017; 2017:3852731.
Article29. Daglioglu YK, Duzgun O, Sarici IS, Ulutas KT. Comparison of platelet rich plasma versus fibrin glue on colonic anastomoses in rats. Acta Cir Bras. 2018; 33:333–340.
Article30. Spartalis E, Prodromidou A, Spartalis M, Machairas N. Comment on ‘Role of platelet-rich fibrin on intestinal anastomosis wound healing in a rat’. Biomed Mater. 2018; 13:068001.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Platelet-rich Plasma on Burn Wounds according to Time of Application: An Experimental Study on Rats
- Platelet-rich plasma: a healing virtuoso
- Chemotherapy for ovarian cancer
- Use of Platelet-rich Plasma
- Current clinical applications of platelet-rich plasma in various gynecological disorders: An appraisal of theory and practice