J Breast Cancer.
2019 Sep;22(3):491-496. 10.4048/jbc.2019.22.e32.
Primary Histiocytic Sarcoma Presenting as a Breast Mass: A Case Report
- Affiliations
-
- 1Department of Pathology, College of Medicine, Hanyang University, Seoul, Korea. charm@hanyang.ac.kr
- 2Department of Surgery, College of Medicine, Hanyang University, Seoul, Korea.
- 3Department of Radiology, College of Medicine, Hanyang University, Seoul, Korea.
- 4Department of Nuclear Medicine, College of Medicine, Hanyang University, Seoul, Korea.
- KMID: 2458869
- DOI: http://doi.org/10.4048/jbc.2019.22.e32
Abstract
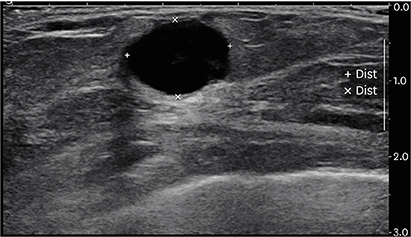
- Histiocytic sarcoma is a rare hematologic malignancy, with very few cases of primary histiocytic sarcoma of the breast described in English scientific literature. Herein, we describe a case of primary histiocytic sarcoma of the breast in a 75-year-old woman, with no clinical history of malignant tumors, who presented with a palpable solitary breast mass. Microscopically, the resected breast mass showed large pleomorphic cells, some multinucleated giant cells, and admixed inflammatory components. The pleomorphic tumor cells further showed a diffuse, noncohesive growth pattern, an abundant eosinophilic cytoplasm, and strong and diffuse immunoreactivity for cluster of differentiation (CD) 68 and CD163. Furthermore, a whole-body positron-emission tomography/computed tomography using deoxy-2-[¹â¸F]fluoro-D-glucose performed after surgery showed no other masses or lesions. After surgical excision, the patient was followed up, and no evidence of tumor recurrence or metastasis was noted.
MeSH Terms
Figure
Reference
-
1. Kommalapati A, Tella SH, Durkin M, Go RS, Goyal G. Histiocytic sarcoma: a population-based analysis of incidence, demographic disparities, and long-term outcomes. Blood. 2018; 131:265–268.
Article2. Trevisan F, Xavier CA, Pinto CA, Cattete FG, Stock FS, Martins ML. Case report of cutaneous histiocytic sarcoma: diagnostic and therapeutic dilemmas. An Bras Dermatol. 2013; 88:807–810.
Article3. Petkova NY, Hristoskova RG, Guenova ML, Raynov JI. A rare case of primary histiocytic sarcoma of the stomach. Folia Med (Plovdiv). 2018; 60:318–322.
Article4. May JM, Waddle MR, Miller DH, Stross WC, Kaleem TA, May BC, et al. Primary histiocytic sarcoma of the central nervous system: a case report with platelet derived growth factor receptor mutation and PD-L1/PD-L2 expression and literature review. Radiat Oncol. 2018; 13:167.
Article5. Yoshida C, Takeuchi M. Histiocytic sarcoma: identification of its histiocytic origin using immunohistochemistry. Intern Med. 2008; 47:165–169.
Article6. Hornick JL, Jaffe ES, Fletcher CD. Extranodal histiocytic sarcoma: clinicopathologic analysis of 14 cases of a rare epithelioid malignancy. Am J Surg Pathol. 2004; 28:1133–1144.7. Nangal JK, Kapoor A, Narayan S, Singhal MK, Beniwal S, Kumar HS. A case of CD68 negative histiocytic sarcoma of axilla masquerading as metastatic breast cancer. J Surg Case Rep. 2014; 2014:rju071.
Article8. Escandell I, Ramon MD, Sánchez S, Terradez L, Jorda E. Dermoscopic characteristics of a cutaneous histiocytic sarcoma in a young patient. J Am Acad Dermatol. 2017; 76:S5–7.
Article9. Skala SL, Lucas DR, Dewar R. Histiocytic sarcoma: review, discussion of transformation from B-cell lymphoma, and differential diagnosis. Arch Pathol Lab Med. 2018; 142:1322–1329.
Article10. Zhou J, Liu Y, Wang W, Xu FL. Histiocytic sarcoma of the neck: a case report. Mol Clin Oncol. 2018; 9:54–57.
Article11. Ansari J, Naqash AR, Munker R, El-Osta H, Master S, Cotelingam JD, et al. Histiocytic sarcoma as a secondary malignancy: pathobiology, diagnosis, and treatment. Eur J Haematol. 2016; 97:9–16.
Article12. Go H, Jeon YK, Huh J, Choi SJ, Choi YD, Cha HJ, et al. Frequent detection of BRAF(V600E) mutations in histiocytic and dendritic cell neoplasms. Histopathology. 2014; 65:261–272.
Article13. Liu Q, Tomaszewicz K, Hutchinson L, Hornick JL, Woda B, Yu H. Somatic mutations in histiocytic sarcoma identified by next generation sequencing. Virchows Arch. 2016; 469:233–241.
Article14. Facchetti F, Pileri SA, Lorenzi L, Tabanelli V, Rimsza L, Pittaluga S, et al. Histiocytic and dendritic cell neoplasms: what have we learnt by studying 67 cases. Virchows Arch. 2017; 471:467–489.
Article15. Pileri SA, Grogan TM, Harris NL, Banks P, Campo E, Chan JK, et al. Tumours of histiocytes and accessory dendritic cells: an immunohistochemical approach to classification from the International Lymphoma Study Group based on 61 cases. Histopathology. 2002; 41:1–29.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of Histiocytic Sarcoma Arising in Head and Neck Region with Rhabdoid Differentiation
- Histiocytic Sarcoma of Rectum: A Case Report
- Histiocytic Sarcoma of the Spleen: A Case Report and Review of the Literature
- Metastatic Granulocytic Sarcoma in the Breast: A Case Report
- Primary Angiosarcoma of the Breast: A Case Report