Clin Exp Vaccine Res.
2019 Jul;8(2):132-135. 10.7774/cevr.2019.8.2.132.
Rabies immune status of raccoon dogs residing in areas where rabies bait vaccine has been distributed
- Affiliations
-
- 1Viral Disease Research Division, Animal and Plant Quarantine Agency, Ministry of Agriculture, Food and Rural Affairs, Gimcheon, Korea. yangdk@korea.kr
- 2College of Veterinary Medicine, Kangwon National University, Chuncheon, Korea.
- KMID: 2455085
- DOI: http://doi.org/10.7774/cevr.2019.8.2.132
Abstract
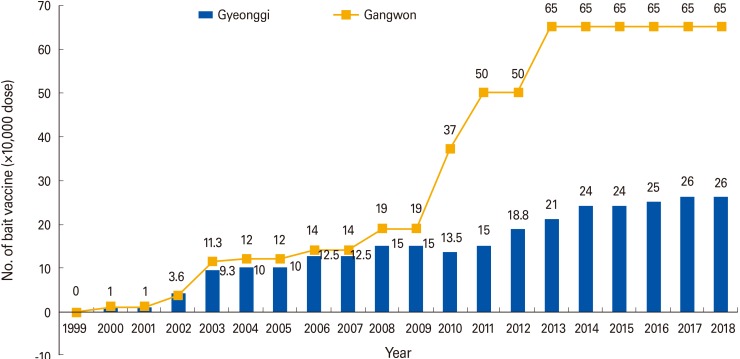
- Since 2000, large amounts of rabies bait vaccine have been distributed in two provinces where raccoon dog-mediated rabies has occurred. A total of 146 raccoon dogs were caught in Gangwon and Gyeonggi Provinces from January 2017 to June 2018, and raccoon dog blood samples were collected. Of the 146 raccoon dogs, 13.7% (20/146) had rabies antibodies. In Gyeonggi and Gangwon provinces, the rate of rabies antibody was 8.5% (5/59) and 17.2% (15/87), respectively. Considering these results, it would be desirable to improve the distribution method or use a new bait vaccine to prevent animal rabies in South Korea.
Keyword
MeSH Terms
Figure
Reference
-
1. Fooks AR, Banyard AC, Horton DL, Johnson N, McElhinney LM, Jackson AC. Current status of rabies and prospects for elimination. Lancet. 2014; 384:1389–1399. PMID: 24828901.
Article2. Bourhy H, Kissi B, Tordo N. Taxonomy and evolutionary studies on lyssaviruses with special reference to Africa. Onderstepoort J Vet Res. 1993; 60:277–282. PMID: 7777311.3. Kim CH, Lee CG, Yoon HC, et al. Rabies, an emerging disease in Korea. J Vet Med B Infect Dis Vet Public Health. 2006; 53:111–115. PMID: 16629721.
Article4. Yang DK, Kim HH, Cho IS. Strategies to maintain Korea’s animal rabies non-occurrence status. Clin Exp Vaccine Res. 2018; 7:87–92. PMID: 30112347.
Article5. Office International des Epizooties. Manual of diagnostic tests and vaccines for terrestrial animals (rabies). 7th ed. Paris: Office International des Epizooties;2012. p. 263–282.6. Maki J, Guiot AL, Aubert M, et al. Oral vaccination of wildlife using a vaccinia-rabies-glycoprotein recombinant virus vaccine (RABORAL V-RG((R))): a global review. Vet Res. 2017; 48:57. PMID: 28938920.
Article7. Ortmann S, Kretzschmar A, Kaiser C, et al. In vivo safety studies with SPBN GASGAS in the frame of oral vaccination of foxes and raccoon dogs against rabies. Front Vet Sci. 2018; 5:91. PMID: 29868616.
Article8. Rupprecht CE, Hanlon CA, Slate D. Oral vaccination of wildlife against rabies: opportunities and challenges in prevention and control. Dev Biol (Basel). 2004; 119:173–184. PMID: 15742629.9. Cliquet F, Aubert M. Elimination of terrestrial rabies in Western European countries. Dev Biol (Basel). 2004; 119:185–204. PMID: 15747421.10. Sterner RT, Meltzer MI, Shwiff SA, Slate D. Tactics and economics of wildlife oral rabies vaccination, Canada and the United States. Emerg Infect Dis. 2009; 15:1176–1184. PMID: 19757549.
Article11. Oh SY, Kim SA, Kim JY, Yoo HS, Lee KK, Shin NS. Detection of antibodies against the rabies virus in Korean raccoon dogs (Nyctereutes procyonoides koreensis). J Zoo Wildl Med. 2012; 43:174–176. PMID: 22448528.
Article12. Mainguy J, Fehlner-Gardiner C, Slate D, Rudd RJ. Oral rabies vaccination in raccoons: comparison of ONRAB(R) and RABORAL V-RG(R) vaccine-bait field performance in Quebec, Canada and Vermont, USA. J Wildl Dis. 2013; 49:190–193. PMID: 23307388.13. Hsu AP, Tseng CH, Barrat J, et al. Safety, efficacy and immunogenicity evaluation of the SAG2 oral rabies vaccine in Formosan ferret badgers. PLoS One. 2017; 12:e0184831. PMID: 28977009.
Article14. Lalosevic D, Lalosevic V, Lazarevic-Ivanc L, Knezevic I. BHK-21 cell culture rabies vaccine: immunogenicity of a candidate vaccine for humans. Dev Biol (Basel). 2008; 131:421–429. PMID: 18634504.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Antibody Response in Korean Raccoon Dogs Inoculated with Inactivated Rabies Vaccines
- Outbreaks and Control of Animal Rabies in Korea
- Safety and Immunogenicity of a Recombinant Rabies Virus Strain (ERAG3G) in Korean Raccoon Dogs
- Strategies to maintain Korea's animal rabies non-occurrence status
- Mass vaccination has led to the elimination of rabies since 2014 in South Korea