Cancer Res Treat.
2018 Oct;50(4):1175-1185. 10.4143/crt.2017.494.
Prognosis of Pancreatic Cancer Patients with Synchronous or Metachronous Malignancies from Other Organs Is Better than Those with Pancreatic Cancer Only
- Affiliations
-
- 1Department of Pathology, Hanyang University College of Medicine, Seoul, Korea.
- 2Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. smhong28@gmail.com
- 3Department of Oncology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- 4Department of Surgery, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- 5Department of Radiation Oncology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- 6Department of Gastroenterology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- KMID: 2424790
- DOI: http://doi.org/10.4143/crt.2017.494
Abstract
- PURPOSE
Pancreatic cancer associated double primary tumors are rare and their clinicopathologic characteristics are not well elucidated.
MATERIALS AND METHODS
Clinicopathologic factors of 1,352 primary pancreatic cancers with or without associated double primary tumors were evaluated.
RESULTS
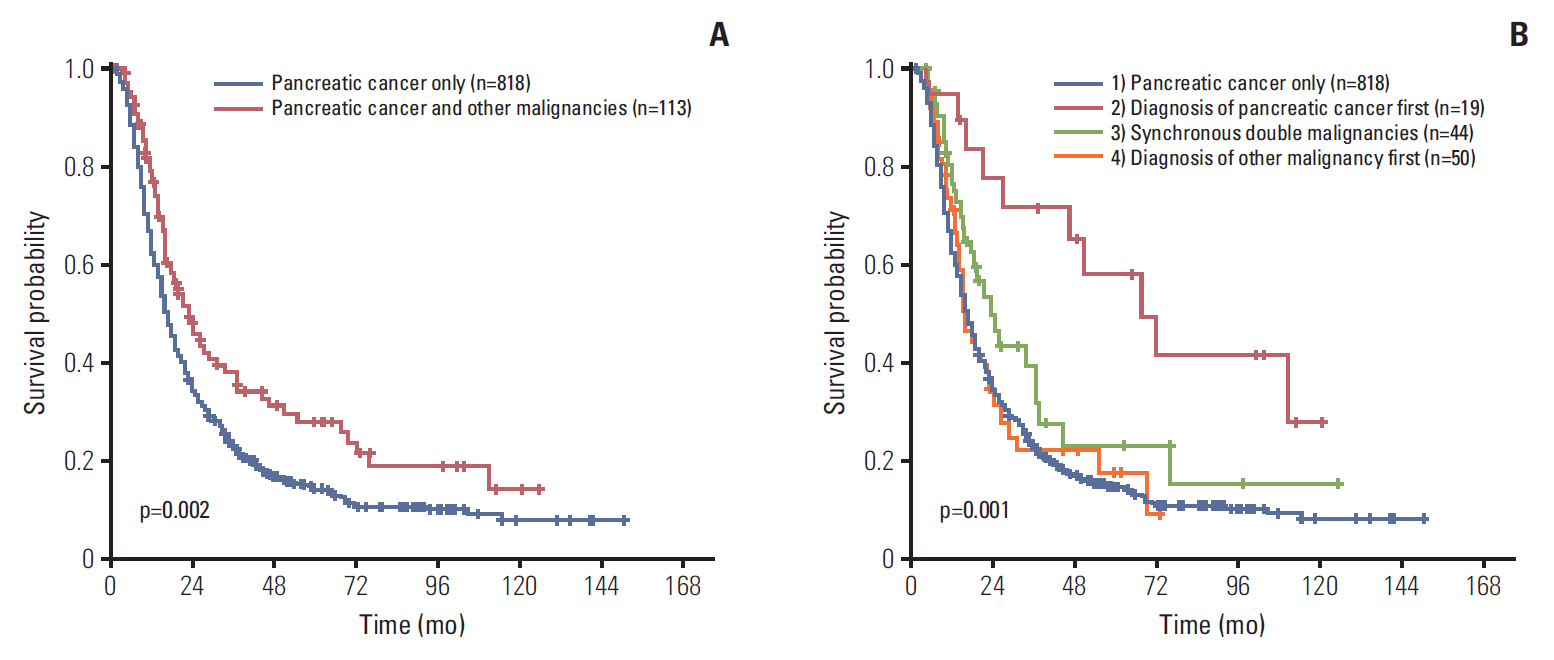
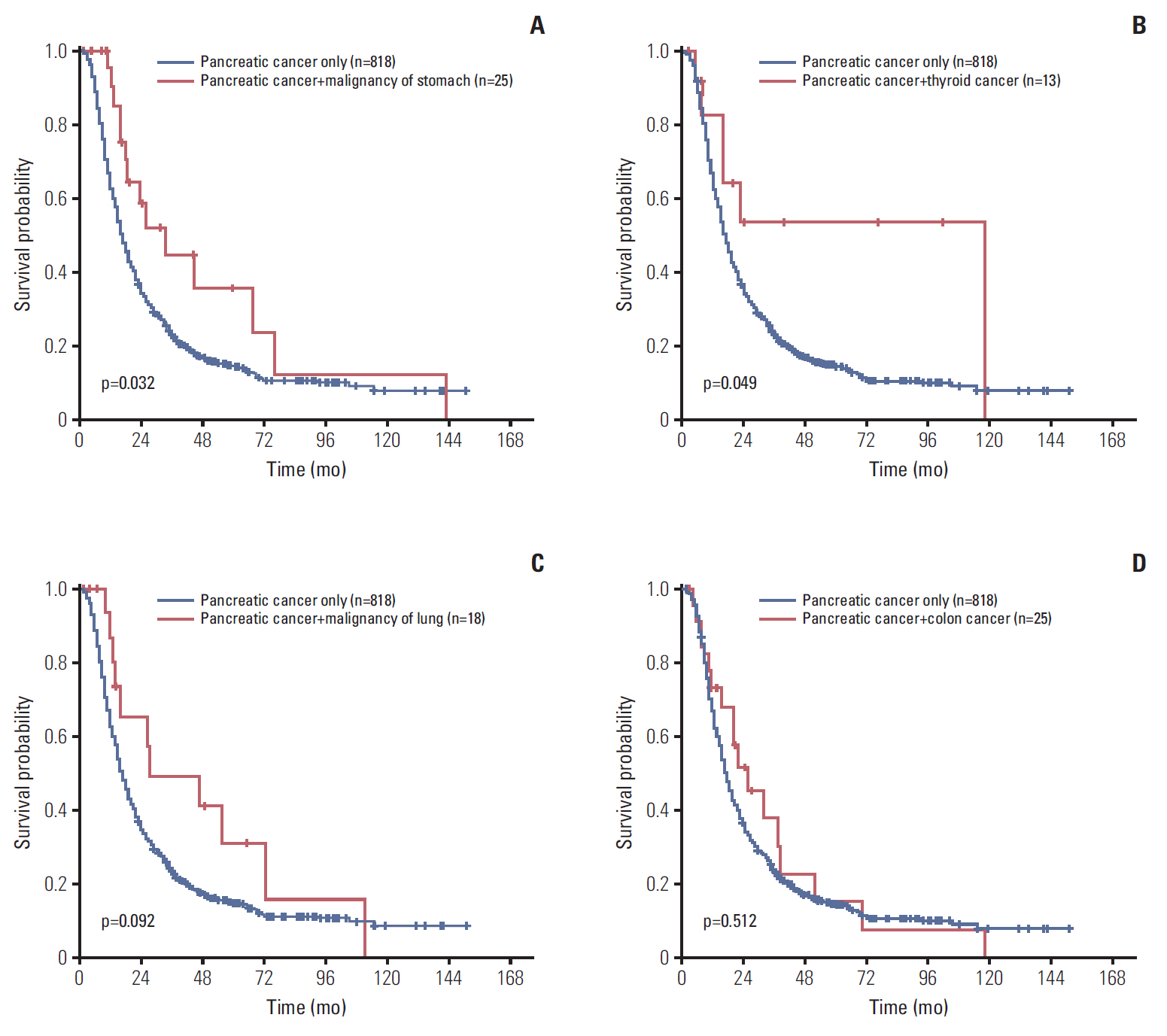
Of resected primary pancreatic cancers, 113 (8.4%) had associated double primary tumors, including 26 stomach, 25 colorectal, 18 lung, and 13 thyroid cancers. The median interval between the diagnoses of pancreatic cancer and associated double primary tumors was 0.5 months. Overall survival (OS) of pancreatic cancer patients with associated double primary tumors was longer than those with pancreatic cancer only (median, 23.1 months vs. 17.0 months; p=0.002). Patients whose pancreatic cancers were resected before the diagnosis of metachronous tumors had a better OS than patients whose pancreatic cancer resected after the diagnosis of metachronous tumors (48.9 months and 13.5 months, p=0.001) or those whose pancreatic cancers were resected synchronously with non-pancreas tumors (19.1 months, p=0.043). The OS of pancreatic cancer patients with stomach (33.9 months, p=0.032) and thyroid (117.8 months, p=0.049) cancers was significantly better than those with pancreas cancer only (17.0 months).
CONCLUSION
About 8% of resected pancreatic cancers had associated double primary tumors, and those from the colorectum, stomach, lung, and thyroid were common. Patients whose pancreatic cancer was resected before the diagnosis of metachronous tumors had better OS than those resected after the diagnosis of metachronous tumors or those resected synchronously.
MeSH Terms
Figure
Cited by 1 articles
-
A Newly Developed Pancreatic Adenocarcinoma in a Patient with Advanced Thyroid Cancer under Long-Term Sorafenib Use
Min Ji Kim, Han-Sang Baek, Sung Hak Lee, Dong-Jun Lim
Int J Thyroidol. 2021;14(2):175-179. doi: 10.11106/ijt.2021.14.2.175.
Reference
-
References
1. Bosman FT, Carneiro F, Hruban RH, Theise ND. WHO classification of tumors of the digestive system. 4th ed. Lyon: IARC Press;2010.2. Donahue TR, Reber HA. Surgical management of pancreatic cancer: pancreaticoduodenectomy. Semin Oncol. 2015; 42:98–109.3. Bali MA, Metens T, Denolin V, Delhaye M, Demetter P, Closset J, et al. Tumoral and nontumoral pancreas: correlation between quantitative dynamic contrast-enhanced MR imaging and histopathologic parameters. Radiology. 2011; 261:456–66.
Article4. Balachandran A, Bhosale PR, Charnsangavej C, Tamm EP. Imaging of pancreatic neoplasms. Surg Oncol Clin N Am. 2014; 23:751–88.
Article5. Bartsch DK, Gress TM, Langer P. Familial pancreatic cancer: current knowledge. Nat Rev Gastroenterol Hepatol. 2012; 9:445–53.6. Takai E, Yachida S, Shimizu K, Furuse J, Kubo E, Ohmoto A, et al. Germline mutations in Japanese familial pancreatic cancer patients. Oncotarget. 2016; 7:74227–35.
Article7. Cho JH, Bang S, Park SW, Chung JB, Song SY. BRCA2 mutations as a universal risk factor for pancreatic cancer has a limited role in Korean ethnic group. Pancreas. 2008; 36:337–40.
Article8. Eriguchi N, Aoyagi S, Hara M, Okuda K, Tamae T, Fukuda S, et al. Synchronous or metachronous double cancers of the pancreas and other organs: report on 12 cases. Surg Today. 2000; 30:718–21.
Article9. Gerdes B, Ziegler A, Ramaswamy A, Wild A, Langer P, Bartsch DK. Multiple primaries in pancreatic cancer patients: indicator of a genetic predisposition? Int J Epidemiol. 2000; 29:999–1003.
Article10. Moertel CG. Multiple primary malignant neoplasms: historical perspectives. Cancer. 1977; 40(4 Suppl):1786–92.11. Edge SB, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti AE. AJCC cancer staging handbook. 7th ed. New York: Springer;2010.12. Tagawa T, Ito K, Fukuzawa K, Okamoto T, Fujinaga A, Kawasaki T, et al. Surgical outcomes of non-small cell lung cancer in patients with a history of pancreaticobiliary cancer. Anticancer Res. 2017; 37:3307–9.
Article13. Ghothim M, Havlik R, Skalicky P, Klos D, Vrba R, Straznicka J, et al. Synchronous cancer duplicities of pancreas and stomach/kidney and their surgical treatment. Rozhl Chir. 2015; 94:251–5.14. Muller SA, Pahernik S, Hinz U, Martin DJ, Wente MN, Hackert T, et al. Renal tumors and second primary pancreatic tumors: a relationship with clinical impact? Patient Saf Surg. 2012; 6:18.
Article15. Kamisawa T, Isawa T, Egawa N, Tsuruta K, Okamoto A, Kawamura T, et al. Study on pancreatic cancer associated with other primary malignancies. Suizo. 1993; 8:164–9.16. Makino TK, Takahashi S, Maruyama H, Kohara T, Yokose Y, Emi Y, et al. Clinicopathology of pancreatic cancer analyzed from annual of the pathological autopsy cases in Japan. Tan To Sui. 1984; 5:761–8.17. Yoshimori M, Tajiri H, Nakamura K, Kishi K, Ozaki H. Clinical significance of multiple cancers including pancreatic cancer: report on 12 cases. Gan No Rinsho. 1982; 28:165–7.18. Maruchi N, Brian D, Ludwig J, Elveback LR, Kurland LT. Cancer of the pancreas in Olmsted County, Minnesota, 1935-1974. Mayo Clin Proc. 1979; 54:245–9.19. Cubilla A, Fitzgerald PJ. Pancreas cancer. I. Duct adenocarcinoma. A clinical-pathologic study of 380 patients. Pathol Annu. 1978; 13 Pt 1:241–89.20. Kasumi F, Togo S, Ota T, Takagi K, Kato H. Study of cases with double carcinoma of the stomach and pancreas. Gan No Rinsho. 1977; 23:1306–14.21. Moertel CG, Dockerty MB, Baggenstoss AH. Multiple primary malignant neoplasm. I. Introduction and presentation of data. Cancer. 1961; 14:221–30.22. Warren S, Gates O. Multiple primary malignant tumors: a survey of the literature and a statistical study. Am J Cancer. 1932; 16:1358–414.23. Jasperson KW, Tuohy TM, Neklason DW, Burt RW. Hereditary and familial colon cancer. Gastroenterology. 2010; 138:2044–58.
Article24. Jun SY, Lee EJ, Kim MJ, Chun SM, Bae YK, Hong SU, et al. Lynch syndrome-related small intestinal adenocarcinomas. Oncotarget. 2017; 8:21483–500.
Article25. Malesci A, Laghi L, Bianchi P, Delconte G, Randolph A, Torri V, et al. Reduced likelihood of metastases in patients with microsatellite-unstable colorectal cancer. Clin Cancer Res. 2007; 13:3831–9.
Article