J Vet Sci.
2017 Mar;18(1):101-104. 10.4142/jvs.2017.18.1.101.
Stage-specific embryonic antigen: determining expression in canine glioblastoma, melanoma, and mammary cancer cells
- Affiliations
-
- 1Department of Veterinary Clinical Sciences and Masonic Cancer Center, University of Minnesota, Minneapolis, MN 55455, USA. modiano@umn.edu wmlin925@126.com
- 2Department of Veterinary Medicine, College of Life Sciences, Longyan University, Longyan 364012, China.
- KMID: 2412594
- DOI: http://doi.org/10.4142/jvs.2017.18.1.101
Abstract
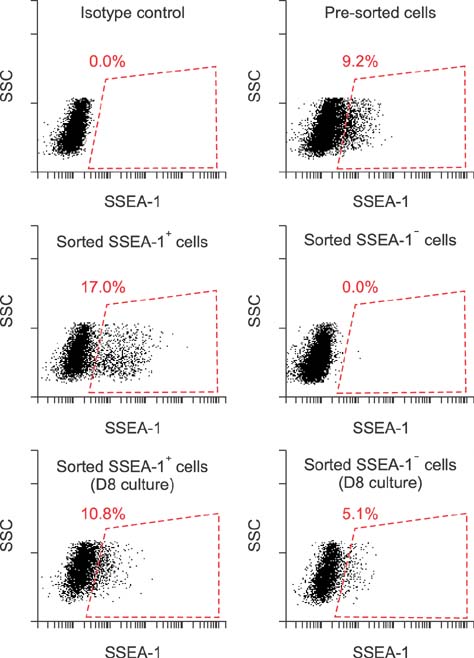
- The expression of stage-specific embryonic antigens (SSEAs) was determined in several types of canine cancer cells. Flow cytometry showed SSEA-1 expression in glioblastoma, melanoma, and mammary cancer cells, although none expressed SSEA-3 or SSEA-4. Expression of SSEA-1 was not detected in lymphoma, osteosarcoma, or hemangiosarcoma cell lines. Relatively stable SSEA-1 expression was observed between 24 and 72 h of culture. After 8 days in culture, sorted SSEA-1⻠and SSEA-1⺠cells re-established SSEA-1 expression to levels comparable to those observed in unsorted cells. Our results document, for the first time, the expression of SSEA-1 in several canine cancer cell lines.
Keyword
MeSH Terms
Figure
Reference
-
1. Elola MT, Capurro MI, Barrio MM, Coombs PJ, Taylor ME, Drickamer K, Mordoh J. Lewis x antigen mediates adhesion of human breast carcinoma cells to activated endothelium. Possible involvement of the endothelial scavenger receptor C-type lectin. Breast Cancer Res Treat. 2007; 101:161–174.
Article2. Hakomori S. Tumor-associated carbohydrate antigens defining tumor malignancy: basis for development of anti-cancer vaccines. Adv Exp Med Biol. 2001; 491:369–402.
Article3. Khanna C, Lindblad-Toh K, Vail D, London C, Bergman P, Barber L, Breen M, Kitchell B, McNeil E, Modiano JF, Niemi S, Comstock KE, Ostrander E, Westmoreland S, Withrow S. The dog as a cancer model. Nat Biotechnol. 2006; 24:1065–1066.
Article4. Koh S, Piedrahita JA. From “ES-like” cells to induced pluripotent stem cells: a historical perspective in domestic animals. Theriogenology. 2014; 81:103–111.
Article5. Lin WM, Karsten U, Goletz S, Cheng RC, Cao Y. Carbohydrate antigens as cancer-initiating cell markers. In : Shostak S, editor. Cancer Stem Cells: The Cutting Edge. Rijeka: InTechOpen;2011. p. 351–360.6. Nakagawa T, Endo Y, Watanabe M, Mochizuki M, Nishimura R, Sugano S, Sasaki N. Adhesional function of canine mammary gland tumor cells expressing sialyl Lewis X. J Vet Med Sci. 2009; 71:1225–1228.
Article7. Ni C, Huang J. Dynamic regulation of cancer stem cells and clinical challenges. Clin Transl Oncol. 2013; 15:253–258.
Article8. Noto Z, Yoshida T, Okabe M, Koike C, Fathy M, Tsuno H, Tomihara K, Arai N, Noguchi M, Nikaido T. CD44 and SSEA-4 positive cells in an oral cancer cell line HSC-4 possess cancer stem-like cell characteristics. Oral Oncol. 2013; 49:787–795.
Article9. Ohtsubo K, Marth JD. Glycosylation in cellular mechanisms of health and disease. Cell. 2006; 126:855–867.
Article10. Pruszak J, Sonntag KC, Aung MH, Sanchez-Pernaute R, Isacson O. Markers and methods for cell sorting of human embryonic stem cell-derived neural cell populations. Stem Cells. 2007; 25:2257–2268.
Article11. Rademacher TW, Parekh RB, Dwek RA. Glycobiology. Annu Rev Biochem. 1988; 57:785–838.
Article12. Son MJ, Woolard K, Nam DH, Lee J, Fine HA. SSEA-1 is an enrichment marker for tumor-initiating cells in human glioblastoma. Cell Stem Cell. 2009; 4:440–452.
Article13. Suila H, Pitkänen V, Hirvonen T, Heiskanen A, Anderson H, Laitinen A, Natunen S, Miller-Podraza H, Satomaa T, Natunen J, Laitinen S, Valmu L. Are globoseries glycosphingolipids SSEA-3 and -4 markers for stem cells derived from human umbilical cord blood? J Mol Cell Biol. 2011; 3:99–107.
Article14. Suzuki Y, Haraguchi N, Takahashi H, Uemura M, Nishimura J, Hata T, Takemasa I, Mizushima T, Ishii H, Doki Y, Mori M, Yamamoto H. SSEA-3 as a novel amplifying cancer cell surface marker in colorectal cancers. Int J Oncol. 2013; 42:161–167.
Article15. Takemitsu H, Zhao D, Yamamoto I, Harada Y, Michishita M, Arai T. Comparison of bone marrow and adipose tissue-derived canine mesenchymal stem cells. BMC Vet Res. 2012; 8:150.
Article16. Wen J, Tao W, Kuiatse I, Lin P, Feng Y, Jones RJ, Orlowski RZ, Zu Y. Dynamic balance of multiple myeloma clonogenic side population cell percentages controlled by environmental conditions. Int J Cancer. 2015; 136:991–1002.
Article17. Yagi H, Saito T, Yanagisawa M, Yu RK, Kato K. Lewis X-carrying N-glycans regulate the proliferation of mouse embryonic neural stem cells via the Notch signaling pathway. J Biol Chem. 2012; 287:24356–24364.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Alpha basic crystallin expression in canine mammary tumors
- Expression of CD133, CD44, CK7, and OCT4 in Animal Cancers
- Expression of embryonic stem cell markers in human term placenta
- Cytotoxicity of natural killer cells on canine mammary carcinoma cells
- Pigmented Mammary Paget Disease Misdiagnosed as Malignant Melanoma