Blood Res.
2017 Mar;52(1):3-6. 10.5045/br.2017.52.1.3.
The Consortium for Improving Survival of Lymphoma (CISL): recent achievements and future perspective
- Affiliations
-
- 1Department of Oncology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- 2Division of Hematology/Oncology, Department of Internal Medicine, Hanyang University College of Medicine, Seoul, Korea.
- 3Division of Hematology/Oncology, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- KMID: 2375194
- DOI: http://doi.org/10.5045/br.2017.52.1.3
Abstract
- No abstract available.
MeSH Terms
Figure
Reference
-
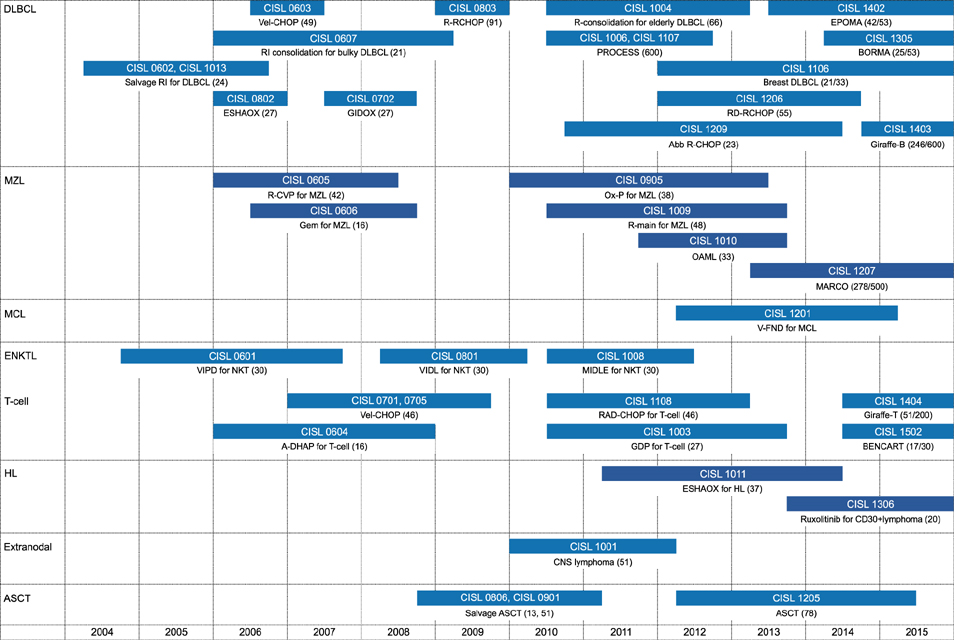
1. Suh C, Kim WS, Kim JS, Park BB. Review of the clinical research conducted by the Consortium for Improving Survival of Lymphoma of the Korean Society of Hematology Lymphoma Working Party. Blood Res. 2013; 48:171–177.
Article2. Kim S, Kim H, Kang H, et al. Clinical significance of cytogenetic aberrations in bone marrow of patients with diffuse large B-cell lymphoma: prognostic significance and relevance to histologic involvement. J Hematol Oncol. 2013; 6:76.
Article3. Hyun SY, Cheong JW, Kim SJ, et al. High-dose etoposide plus granulocyte colony-stimulating factor as an effective chemomobilization regimen for autologous stem cell transplantation in patients with non-Hodgkin lymphoma previously treated with CHOP-based chemotherapy: a study from the Consortium for Improving Survival of Lymphoma. Biol Blood Marrow Transplant. 2014; 20:73–79.
Article4. Lee SJ, Suh CW, Lee SI, et al. Clinical characteristics, pathological distribution, and prognostic factors in non-Hodgkin lymphoma of Waldeyer's ring: nationwide Korean study. Korean J Intern Med. 2014; 29:352–360.
Article5. Kang BW, Sohn SK, Moo JH, et al. Clinical features and treatment outcomes in patients with mantle cell lymphoma in Korea: Study by the Consortium for Improving Survival of Lymphoma. Blood Res. 2014; 49:15–21.
Article6. Lee S, Kim MJ, Kim JS, et al. Intraocular lymphoma in Korea: the Consortium for Improving Survival of Lymphoma (CISL) study. Blood Res. 2015; 50:242–247.
Article7. Jeong SH, Moon JH, Kim JS, et al. Multicenter analysis of treatment outcomes in adult patients with lymphoblastic lymphoma who received hyper-CVAD induction followed by hematopoietic stem cell transplantation. Ann Hematol. 2015; 94:617–625.
Article8. Hong J, Kim SJ, Ahn JS, et al. Treatment outcomes of rituximab plus hyper-CVAD in Korean patients with sporadic Burkitt or Burkitt-like lymphoma: Results of a multicenter analysis. Cancer Res Treat. 2015; 47:173–181.
Article9. Lee GW, Go SI, Kim SH, et al. Clinical outcome and prognosis of patients with primary sinonasal tract diffuse large B-cell lymphoma treated with rituximab-cyclophosphamide, doxorubicin, vincristine and prednisone chemotherapy: a study by the Consortium for Improving Survival of Lymphoma. Leuk Lymphoma. 2015; 56:1020–1026.
Article10. Koh MS, Kim WS, Kim SJ, et al. Is there role of additional chemotherapy after definitive local treatment for stage I/II marginal zone lymphoma?: Consortium for Improving Survival of Lymphoma (CISL) study. Int J Hematol. 2015; 102:420–425.
Article11. Kang HJ, Lee SS, Byun BH, et al. Repeated radioimmunotherapy with 131I-rituximab for patients with low-grade and aggressive relapsed or refractory B cell non-Hodgkin lymphoma. Cancer Chemother Pharmacol. 2013; 71:945–953.
Article12. Kim SJ, Kang HJ, Kim JS, et al. A phase I study of everolimus and CHOP in newly diagnosed peripheral T-cell lymphomas. Invest New Drugs. 2013; 31:1514–1521.
Article13. Kim SJ, Yang DH, Kim JS, et al. Concurrent chemoradiotherapy followed by L-asparaginase-containing chemotherapy, VIDL, for localized nasal extranodal NK/T cell lymphoma: CISL08-01 phase II study. Ann Hematol. 2014; 93:1895–1901.
Article14. Jung SH, Lee JJ, Kim WS, et al. Weekly rituximab consolidation following four cycles of R-CHOP induction chemotherapy in very elderly patients with diffuse large B-cell lymphoma: Consortium for improving survival of lymphoma study (CISL). Eur J Haematol. 2015; 94:504–510.
Article15. Park BB, Kim WS, Suh C, et al. Salvage chemotherapy of gemcitabine, dexamethasone, and cisplatin (GDP) for patients with relapsed or refractory peripheral T-cell lymphomas: a consortium for improving survival of lymphoma (CISL) trial. Ann Hematol. 2015; 94:1845–1851.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Review of the clinical research conducted by the Consortium for Improving Survival of Lymphoma of the Korean Society of Hematology Lymphoma Working Party
- Consortium for Improving Survival of Lymphoma (CISL): a model of multicenter collaboration for lymphoma studies in Korea
- Organ-specific considerations for marginal zone lymphomas in Korea, based on Consortium for Improving Survival of Lymphoma (CISL) and Korean clinical studies
- Intraocular lymphoma in Korea: the Consortium for Improving Survival of Lymphoma (CISL) study
- A Case of Primary Lymphoma Involving the Heart