Lab Anim Res.
2016 Sep;32(3):135-143. 10.5625/lar.2016.32.3.135.
Assessment of collagen antibody-induced arthritis in BALB/c mice using bioimaging analysis and histopathological examination
- Affiliations
-
- 1Department of Biomedical Laboratory Science, Namseoul University, Cheonan, Korea. kang@nsu.ac.kr
- 2Department of Health Administration, Namseoul University, Cheonan, Korea.
- KMID: 2353588
- DOI: http://doi.org/10.5625/lar.2016.32.3.135
Abstract
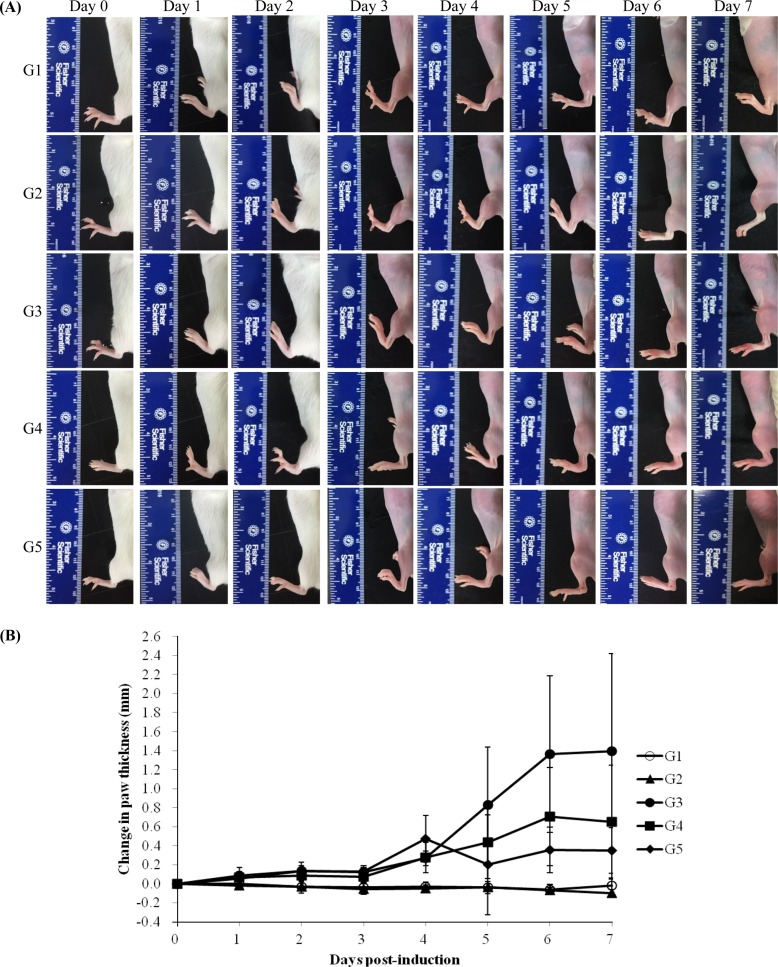
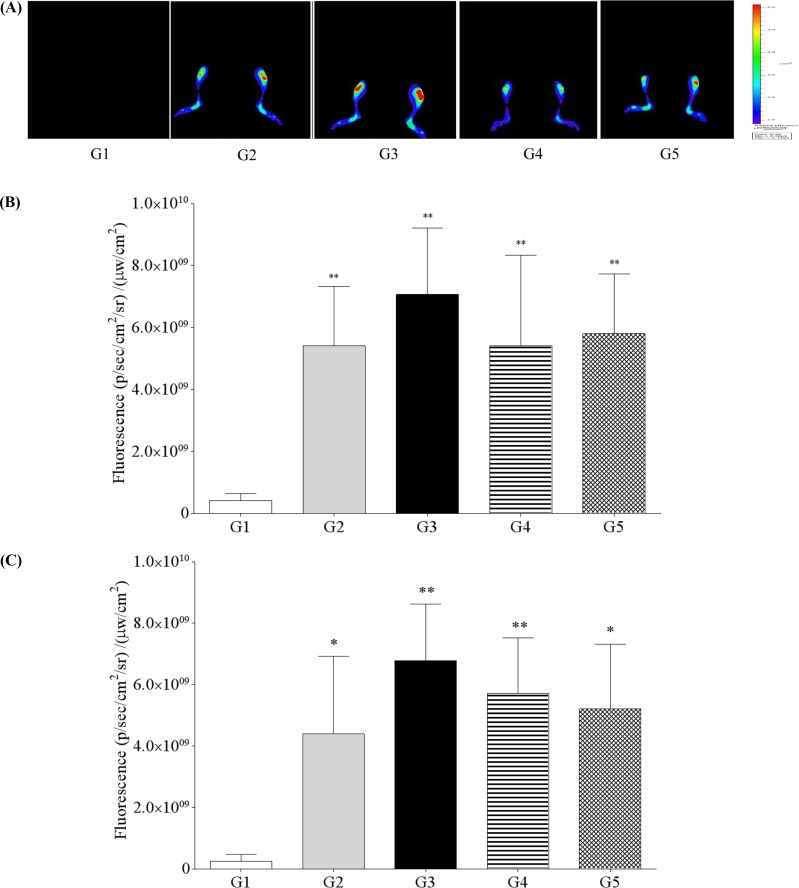
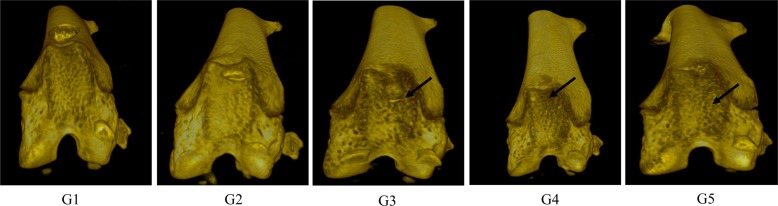
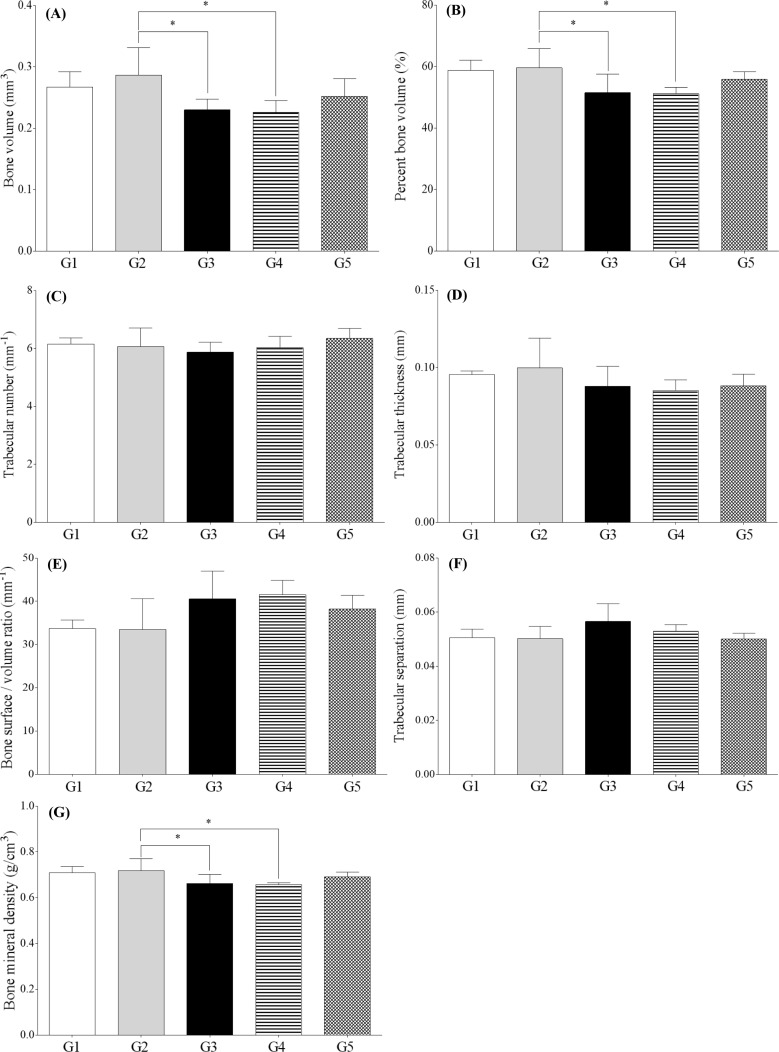
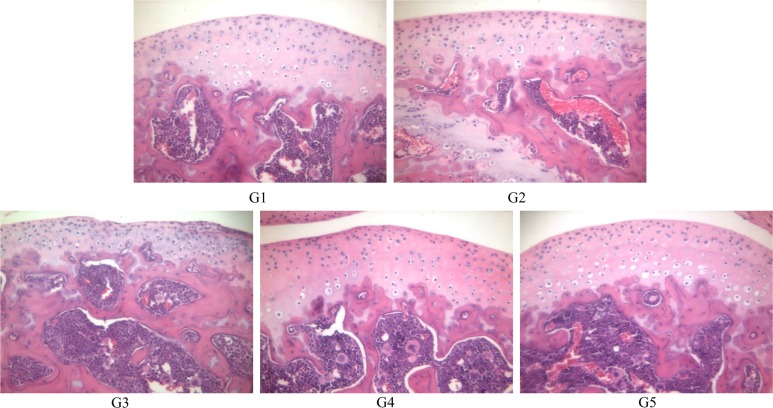
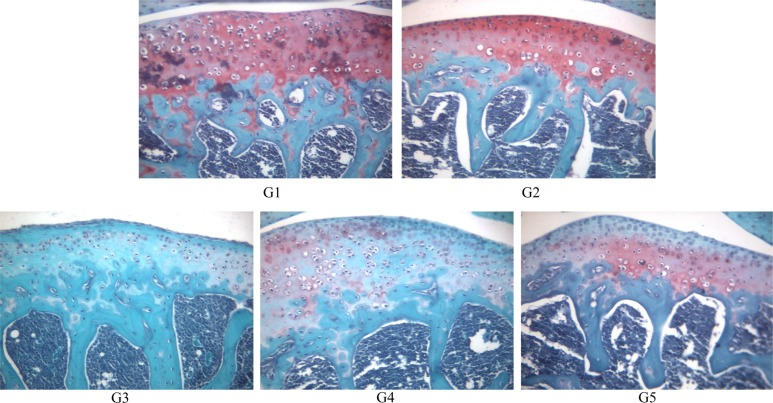
- The aim of this study was to examine the therapeutic potential of sulfasalazine and prednisolone in a mouse collagen antibody-induced arthritis (CAIA) model. Twenty-five male BALB/c mice were randomly divided into five groups: group 1 (G1): control, group 2 (G2): probe control, group 3 (G3): CAIA, group 4 (G4): CAIA+sulfasalazine (10 mg/kg, oral), and group 5 (G5): CAIA+prednisolone (100 mg/kg, oral). Fluorescence bioimaging was performed in vivo 24 and 48 h after treatment with a fluorescence probe (OsteoSense® 680 EX), and all mice were sacrificed. The hind knee joints were fixed in 10% neutral phosphate-buffered formalin, and micro-computed tomography (micro-CT) and histopathological analyses were performed. The paw thickness increased in a time-dependent manner in G3 mice, but trended toward a decrease in both G4 and G5 mice. Fluorescence intensity increased in G3 mice at 24 and 48 h after fluorescence probe treatment, but the fluorescence intensity in G4 and G5 mice was lower than that in G3. Micro-CT analyses showed that the joint surfaces of G3 mice had a rough and irregular articular appearance, but the occurrence of these irregularities was lower in G4 and G5. Hematoxylin and eosin and Safranin O-fast green staining confirmed that destruction of the cartilage and bony structures, synovial hyperplasia, and inflammatory cell infiltration all occurred in G3, and that the occurrence of these phenomena was lower in G4 and G5 than in G3. Taken together, these results suggest that sulfasalazine and prednisolone can reduce acute rheumatoid arthritis in mice.
Keyword
MeSH Terms
Figure
Reference
-
1. Scott DL, Wolfe F, Huizinga TW. Rheumatoid arthritis. Lancet. 2010; 376(9746):1094–1108. PMID: 20870100.
Article2. Khachigian LM. Collagen antibody-induced arthritis. Nat Protoc. 2006; 1(5):2512–2516. PMID: 17406499.
Article3. Nandakumar KS, Svensson L, Holmdahl R. Collagen type II-specific monoclonal antibody-induced arthritis in mice: description of the disease and the influence of age, sex, and genes. Am J Pathol. 2003; 163(5):1827–1837. PMID: 14578183.4. Hutamekalin P, Saito T, Yamaki K, Mizutani N, Brand DD, Waritani T, Terato K, Yoshino S. Collagen antibody-induced arthritis in mice: development of a new arthritogenic 5-clone cocktail of monoclonal anti-type II collagen antibodies. J Immunol Methods. 2009; 343(1):49–55. PMID: 19330909.
Article5. de Jong M, Essers J, van Weerden WM. Imaging preclinical tumour models: improving translational power. Nat Rev Cancer. 2014; 14(7):481–493. PMID: 24943811.
Article6. Chen Y, Liang CP, Liu Y, Fischer AH, Parwani AV, Pantanowitz L. Review of advanced imaging techniques. J Pathol Inform. 2012; 3:22. PMID: 22754737.
Article7. Weissleder R, Tung CH, Mahmood U, Bogdanov A Jr. In vivo imaging of tumors with protease-activated near-infrared fluorescent probes. Nat Biotechnol. 1999; 17(4):375–378. PMID: 10207887.
Article8. Lakey RL, Cawston TE. Sulfasalazine blocks the release of proteoglycan and collagen from cytokine stimulated cartilage and down-regulates metalloproteinases. Rheumatology (Oxford). 2009; 48(10):1208–1212. PMID: 19690126.
Article9. Mihara K, Almansa C, Smeets RL, Loomans EE, Dulos J, Vink PM, Rooseboom M, Kreutzer H, Cavalcanti F, Boots AM, Nelissen RL. A potent and selective p38 inhibitor protects against bone damage in murine collagen-induced arthritis: a comparison with neutralization of mouse TNFalpha. Br J Pharmacol. 2008; 154(1):153–164. PMID: 18297096.10. Bendele A. Animal models of rheumatoid arthritis. J Musculoskelet Neuronal Interact. 2001; 1(4):377–385. PMID: 15758488.11. Knoerzer DB, Donovan MG, Schwartz BD, Mengle-Gaw LJ. Clinical and histological assessment of collagen-induced arthritis progression in the diabetes-resistant BB/Wor rat. Toxicol Pathol. 1997; 25(1):13–19. PMID: 9061845.
Article12. Ying X, Monticello TM. Modern imaging technologies in toxicologic pathology: An overview. Toxicol Pathol. 2006; 34(7):815–826. PMID: 17178685.
Article13. Peterson JD, Labranche TP, Vasquez KO, Kossodo S, Melton M, Rader R, Listello JT, Abrams MA, Misko TP. Optical tomographic imaging discriminates between disease-modifying anti-rheumatic drug (DMARD) and non-DMARD efficacy in collagen antibody-induced arthritis. Arthritis Res Ther. 2010; 12(3):R105. PMID: 20509880.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Fluorescence bioimaging analysis of collagen antibody-induced arthritis in male mice
- Detection of acetaminophen-induced liver damage by fluorescence bioimaging, serum biochemistry and histopathological examination
- The Role of Lipocortin-1 in the Corticosteroid Action in Collagen Induced Arthritis
- Effect of Methotrexate on Collagen-Induced Arthritis Assessed by Micro-Computed Tomography and Histopathological Examination in Female Rats
- The Effect of Acanthopanax koreanum Extract on the Induction of Collagen Induced Arthritis for DBA/1J Mice