Tuberc Respir Dis.
2006 Feb;60(2):171-179.
Cross-resistance Between Rifampicin and Rifabutin and Its Relationship with rpoB Gene Mutations in Clinically Isolated MDR-TB Strains
- Affiliations
-
- 1National Masan Hospital, Masan, Korea. byoungju_kim@yahoo.com
- 2International Tuberculosis Research Center, Masan, Korea.
Abstract
-
BACKGROUND: Despite the emerging danger of MDR-TB to human beings, there have only been a limited number of drugs developed to treat MDR-TB since 1970. This study investigated the cross-resistance rate between rifampicin (RFP) and rifabutin (RBU) in order to determine the efficacy of rifabutin in treating MDR-TB. In addition, the results of rifabutin were correlated with the rpoB mutations, which are believed to be markers for MDR-TB and RFP resistance.
METHODS
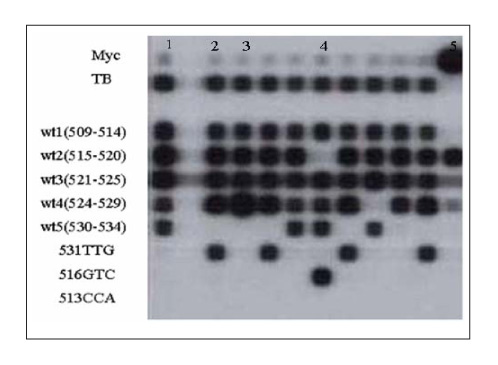
The MICs of RBU were tested against 126 clinical isolates of MDR-TB submitted to the clinical laboratory of National Masan TB Hospital in 2004. Five different concentrations (10-160 microgram/ml) were used for the MICs. The detection of the rpoB mutations was performed using a RFP resistance detection kit with a line probe assay(LiPA), which contains the oligonucleotide probes for 5 wide type and 3 specific mutations (513CCA, 516GTC, and 531TTG). The rpoB mutation was determined by direct sequencing.
RESULTS
The rate of cross-resistance between RFP and RBU was 70.5%(74/105) at 20 microgram/ml RBU(ed note: How much RFP?) Most mutations (86.3%) occurred in the 524~534 codons. The His526Gln, His526Leu, Leu533Pro, Gln513Glu, and Leu511Pro mutations(Ed note: Is this correct?) were associated with the susceptibilty to RBU.
CONCLUSION
Based on the cross-resistance rate between RFP and RBU, RBU may be used effectively in some MDR-TB patients. Therefore, a conventional drug susceptibility test for RBU and a determination of the critical concentration are needed. However, rpoB gene mutation test may be have limited clinical applications in detecting RBU resistance.
Keyword
MeSH Terms
Figure
Reference
-
1. Woodley CL, Kilburn JO. In vitro susceptibility of Mycobacterium avium complex and Mycobacterium tuberculosis strains to a spiro-piperidyl rifamycin. Am Rev Respir Dis. 1982. 126:586–587.2. O'Brien RJ, Lyle MA, Snider DE Jr. Rifabutin (ansamycin LM 427): a new rifamycin-S derivative for the treatment of mycobacterial disease. Rev Infect Dis. 1987. 9:519–530. s.3. O'Brien RJ, Geiter LJ, Lyle MA. Rifabutin (ansamycin LM 427) for the treatment of pulmonary Mycobacterium avium complex. Am Rev Respir Dis. 1990. 141:821–826.4. Ji B, Truffot-Pernot C, Lacroix C, Raviglione MC, O'Brien RJ, Olliaro P, et al. Effectiveness of rifampin, rifabutin, and rifapentine for preventive therapy of tuberculosis in mice. Am Rev Respir Dis. 1993. 148:1541–1546.5. Chien HP, Yu MC, Ong TF, Lin TP, Luh KT. In vitro activity of rifabutin and rifampin against clinical is olates of Mycobacterium tuberculosis in Taiwan. J Formos Med Assoc. 2000. 99:408–411.6. Uzun M, Erturan Z, Ang O. Investigation of cross-resistance between rifampin and rifabutin in Mycobacterium tuberculosis complex strains. Int J Tuberc Lung Dis. 2002. 6:164–165.7. Saribas Z, Kocagoz T, Alp A, Gunalp A. Rapid detection of rifampin resistance in Mycobacterium tuberculosis isolates by heteroduplex analysis and determination of rifamycin cross-resistance in rifampin-resistant isolates. J Clin Microbiol. 2003. 41:816–818.8. Senol G, Erbaycu A, Ozsoz A. Incidence of cross resistance between rifampicin and rifabutin in Mycobacterium tuberculosis strains in Izmir, Turkey. J Chemother. 2005. 17:380–384.9. Shim TS, Kim JS, Park MS, Lim CM, Lee SD, Koh YS, et al. Rifabutin suceptibility and rpoB gene mutations in multi-drug resistant Mycobacterium tuberculosis. Tuberc Respir Dis. 2000. 48:853–869.10. Lew WJ, Park YK, Kim HJ, Chang CH, Bai GH, Kim SK. The proportion of rifabutin-susceptible strains among rifampicin-resistant isolates and its specific rpoB mutations. Tuberc Respir Dis. 2005. 59:257–265.11. Yang B, Koga H, Ohno H, Ogawa K, Fukuda M, Hirakata Y, et al. Relationship between antimycobacterial activities of rifampicin, rifabutin and KRM-1648 and rpoB mutations of Mycobacterium tuberculosis. J Antimicrob Chemother. 1998. 42:621–628.12. Williams DL, Spring L, Collins L, Miller LP, Heifets LB, Gangaharam PRJ, et al. Contribution of rpoB mutations to development of rifamycin cross-resistance in Mycobacterium tuberculosis. Antimicrob Agents Chemother. 1998. 42:1853–1857.13. Cavusoglu C, Karaca-Derici Y, Bilgic A. In-vitro activity of rifabutin against rifampicin-resistant Mycobacterium tuberculosis isolates with known rpoB mutations. Clin Microbiol Infection. 2004. 10:662–665.14. Telenti A. Genetics of drug resistance in tuberculosis. Clin Chest Med. 1997. 18:55–64.15. de Beenhouwer H, Lhiang Z, Jannes G, Mijs W, Machtelinchx L, Rossau R, et al. Rapid detection of rifampicin resistance in sputum and biopsy specimens from tuberculosis patients by PCR and line probe assay. Tubercle Lung Dis. 1995. 76:425–430.16. Telenti A, Imboden P, Marchesi F, Schmidheini T, Bodmer T. Direct, automated detection of rifampin-resistant Mycobacterium tuberculosis by polymerase chain reaction and single-strand conformation polymorphism analysis. Antimicrob Agents Chemother. 1993. 37:2054–2058.17. Canetti G, Fox W, Khomenko A, Mahler H, Menon NK, Mitchison DA, et al. Advances in techniques of testing mycobacterial drug sensitivity, and the use of sensitivity tests in tuberculosis control programmes. Bull World Health Organ. 1969. 41:21–43.18. Bai GH. Anti-tuberculosis drug resistance in Korea. CDMR. 2005. 16:101–107.19. NCCLS. Susceptibility testing of mycobacteria, nocardiae, and other aerobic actinomycetes; Approved standard.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Proportion of Rifabutin-susceptible Strains among Rifampicin- resistant Isolates and Its Specific rpoB Mutations
- Rifabutin susceptibility and rpoB gene mutations in multi-drug resistant mycobacterium tuberculosis
- Detection of Point Mutations in the rpoB gene Related to Drug Susceptibility in Mycobacterium Tuberculosis using an Oligonucleotide Chip
- Comparison of PCR-Line Probe and PCR-SSCP Methods for the Detection of Rifampicin Resistant Mycobacterium Tuberculosis.
- Understanding Rifampicin Resistance in Tuberculosis through a Computational Approach