J Korean Med Assoc.
2003 Jan;46(1):21-28. 10.5124/jkma.2003.46.1.21.
The Advantage of UFT in the Patients with Stage IA & IB Lung Cancer after Complete Resection
- Affiliations
-
- 1Department of Thoracic Surgery, Yongdong Severance Hospital, Yonsei University College of Medicine, Korea. dylee@yumc.yonsei.ac.kr
- KMID: 2183061
- DOI: http://doi.org/10.5124/jkma.2003.46.1.21
Abstract
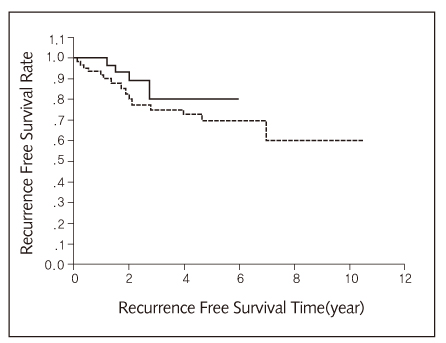
- Despite complete resection of tumor and mediastinal lymph node dissection, the post-operative survival rate of early stage I non-small cell carcinoma is not so good due to increased recurrences. So, we tried to use one of the chemotherapeutic agents after complete resection of lung carcinoma even though the pathologic final diagnosis is stage IA or IB in order to improve post-operative survivals. One-hundred and eight male and 23 female patients (mean age: 62 years, range: 28~83 years) participated in this clinical study from January 1992 to April 2002 at the Department of Thoracic and Cardiovascular Surgery, Yongdong Severance Hospital, Yonsei University, College of Medicine, Seoul, Korea. Twenty-nine patients were in stage IA and 102 were in stage IB. Among them, 3 patients died from surgical problems and 3 patients died from non surgical problems. Fifty-six patients were placed in the UFT treated group and 61 patients were in the control group. The 5-year survival rate of the patients treated with UFT was 88.6% and the 5-year survival rate of the patients in control group was 72.3% (p=0.0596). The results of our trial indicate that even patients with stage IA and IB non-small cell lung carcinoma would be benefited from orall UFT after complete surgical resection of the tumor.
Keyword
MeSH Terms
Figure
Reference
-
1. Rutman RJ, Cantarow A, Paschkis KE. Studies in 2-acetylaminofluorene carcinogenesis. III. The utilization of uracil-2-C14 by preneoplastic rat liver and rat hepatoma. Cancer Res. 1954. 14:119–123.2. Heidelberger C, Chaudhuri NK, Danneberg PB, et al. Fluorinated pyrimidines, a new class of tumor-inhibitory compounds. Nature. 1957. 179:663–666.
Article3. Fujii S, Kitano S, Ikenaka K, et al. Effect of coadministration of uracil or cytosine on the anti-tumor activity of clinical doses of 1-(2-tetrahydrofuryl)-5-fluorouracil and level of 5-fluorouracil in rodents. Gann. 1979. 70:209–214.4. Flehinger BJ, Kimmel M, Melamed MR. The effect of surgical treatment on survival from early lung cancer: implications for screening. Chest. 1992. 101:1013–1018.
Article5. Vrdoljak E, Mise K, Sapunar D, et al. Survival analysis of untreated patients with non-small-cell lung cancer. Chest. 1994. 106:1797–1800.
Article7. Sohn KH, Kwak YT, Cho KH, Kim JH, Lee DY, Lee HK, et al. A Survival Study of Surgically Treated Lung Cancer in Korea. J Korean Med Sci. 1991. 6(2):135–145.
Article8. Martini N, Melamed MR. Occult carcinoma of the lung. Ann Thorac Surg. 1980. 30:215–223.9. Padilla J, Calvo V, Penalver JC, et al. Surgical results and prognostic factors in early non-small-cell lung cancer. Ann Thorac Surg. 1997. 63:2324–2326.10. Ichinose Y, Hara N, Ohta M, et al. Is T factor of the TNM staging system a predominant prognostic factor in pathologic stage I non-small-cell lung cancer? A multivariate prognostic factor analysis of 151 patients. J Thorac Cardiovasc Surg. 1993. 106:90–94.
Article11. Ichinose Y, Yano T, Asoh H, et al. Prognostic factors obtained by a pathologic examination in completely resected non-small-cell lung cancer: an analysis in each pathologic stage. J Thorac Cardiovasc Surg. 1994. 107:1087–1094.
Article12. Macchiarini P, Fontanini G, Hardin JM, et al. Most peripheral, node-negative non-small-cell lung cancers have low proliferative rates and no intratumoral and pertumoral blood and lymphatic vessel invasion: rationale for treatment with wedge resection alone. J Thorac Cardiovasc Surg. 1992. 104:892–899.
Article13. Macchiarini P, Fontanini G, Hardin MJ, et al. Blood vessel invasion by tumor cells predicts recurrence in completely resected T1 N0 M0 non-small-cell lung cancer. J Thorac Cardiovasc Surg. 1993. 106:80–89.
Article15. Churchill ED, Sweet RH, Scannell JG, et al. Futher studies in the surgical management of carcinoma of the lung. J Thorac Surg. 1958. 36:301–308.16. Feld R, Rubinstein LV, Weisenberger TH, et al. Sites of recurrence in resected stage I non-small cell lung cancer: a guide for future studies. J Clin Oncol. 1984. 2:1352–1358.
Article17. D'Amico TA, Massey M, Herndon JE III, et al. A biologic risk model for stage I lung cancer: immunohistochemical analysis of 408 patients with the use of ten molecular markers. J Thorac Cardiovasc Surg. 1999. 117:736–743.18. Williams DE, Pariolero PC, Davis CS, et al. Survival of patients surgically treated for stage I lung cancer. J Thorac Cardiovasc Surg. 1981. 123:446–449.19. Kirby TJ, Mack MJ, Landreneau RJ, et al. Lobectomy-video-assisted thoracic surgery versus muscle-sparing thoracotomy: a randomized trial. J Thorac Cardiovasc Surg. 1995. 109:997–1002.
Article20. Martini N, Bains MS, Burt ME, et al. Incidence of local recurrence and second primary tumors in resected stage I lung cancer. J Thorac cardiovasc Surg. 1995. 109:120–129.
Article21. Thomas PA, Piantadosi S. The Lung Cancer Study Group. Postoperative T1 N0 non-small cell lung cancer. J Thorac Cardiovasc Surg. 1987. 94:349–354.
Article22. Ginsberg RJ, Rubinstein LV. Lung Cancer Study Group. Randomized trial of lobectomy versus limited resection for T1N0 non-small cell lung cancer. Ann Thorac Surg. 1995. 60:615–623.
Article26. Al-Kattan K, Sepsas E, Fountain SW, et al. Disease recurrence after resection for stage I lung cancer. Eur J Cardiovasc Surg. 1997. 12:380–384.
Article27. Naunheim KS, Virgo KS, Coplin MA, et al. Clinical surveillance testing after lung cancer operations. Ann Thorac Surg. 1995. 60:1612–1616.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Impact of Oral Administration of UFT for Postoperative Stage I Non-Small Cell Lung Cancer
- Comparison of Surgical Outcomes and Survival between Octogenarians and Younger Patients after Pulmonary Resection for Stage I Lung Cancer
- Impact of Postoperative Oral Administration of UFT for Completely Resected pT2N0 Non-Small Cell Lung Cancer
- Effect of Adjuvant Chemotherapy after Complete Resection for Pathologic Stage IB Lung Adenocarcinoma in High-Risk Patients as Defined by a New Recurrence Risk Scoring Model
- Risk Factor for Recurrence in Completely Resected Stage IB Non-small Cell Lung Cancer