J Bacteriol Virol.
2008 Dec;38(4):235-237. 10.4167/jbv.2008.38.4.235.
Purification of Protein Expressed from Three Different Regions of Norovirus (NoV)
- Affiliations
-
- 1Department of Food Science and Technology, Ewha Womans University, Seoul, Korea.
- 2Department of Bioindustry, College of Life and Environment, Daegu University, Daegu, Korea.
- 3Department of Microbiology, College of Medicine, the Catholic University of Korea, Seoul, Korea. paik@catholic.ac.kr
- 4Department of Environmental Health Research, National Institute of Environmental Research, Incheon, Korea.
- 5Catholic Hematopoietic Stem Cell Transplantation Center, the Catholic University of Korea, Seoul, Korea.
- KMID: 1483964
- DOI: http://doi.org/10.4167/jbv.2008.38.4.235
Abstract
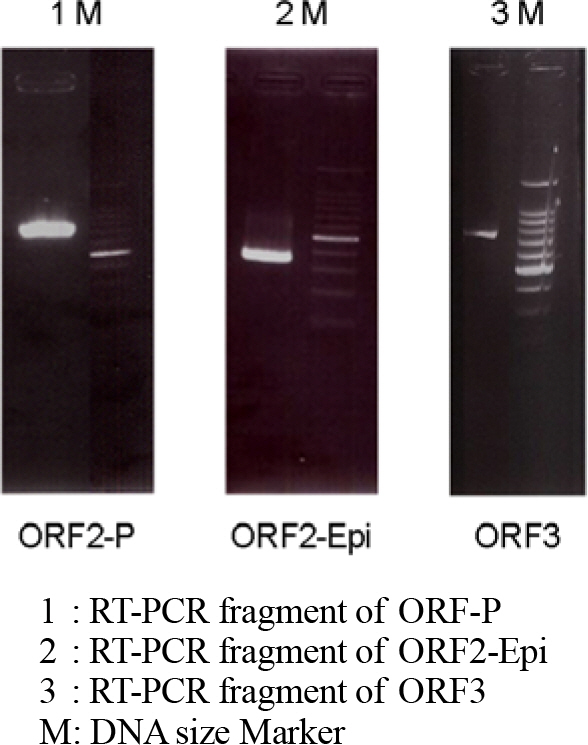
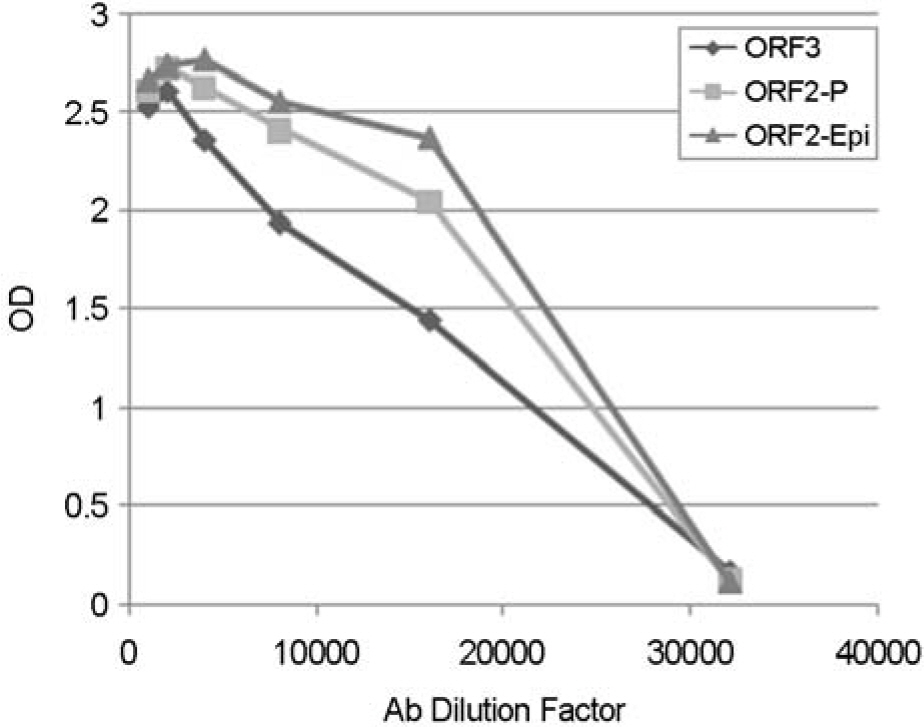
- Norovirus (NoV), which belongs to the family Caliciviridae, is one of the major causes of nonbacterial acute gastroenteritis in the world. In this study, we purified proteins from the epitope region of norovirus for development of the rapid diagnosis system using polyclonal antibodies. As antigens, parts of the ORF (open reading frame) 2, ORF2-P domain, ORF2-Epi, and ORF3 regions were selected and their expressions were induced. The antigenicity of the purified proteins was identified by Western blotting. Each of the purified proteins was injected into mice for the production of novel antibodies and after 3 months of immunization, sera from the mice were obtained. The polyclonal antibody titer was tested by enzyme-linked immunosorbent assay (ELISA) and antibody against ORF2-Epi showed the highest titer. Those polyclonal antibodies can be used in further immunoassay for the rapid detection of NoVs from food and clinical specimens.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Mouse Dual Ig Domain Containing Cell Adhesion Molecule Protein Expression and Purification Using the Baculovirus Expression Vector System
Seung-Won Park, Ji-Hyun Choi, Tae-Won Goo, Seong Ryul Kim, Gwang-Gill Lee, Seok-Woo Kang
J Bacteriol Virol. 2010;40(3):123-130. doi: 10.4167/jbv.2010.40.3.123.
Reference
-
1). Almanza H., Cubillos C., Angulo I., Mateos F., Coastón JR., van der Poel WH., Vinje J., Bárcena J., Mena I. Self-Assembly of the recombinant capsid protein of a swine norovirus into virus-like particles and evaluation of monoclonal antibodies cross-reactive with a human strain from genogroup II. J Clin Microbiol. 46:3971–3979. 2008.
Article2). Glass RI., Noel J., Ando T., Fankhauser R., Belliot G., Mounts A., Parashar UD., Bresee JS., Monroe SS. The epidemiology of enteric caliciviruses from humans: a reassessment using new diagnosis. J Infect Dis. 181:S254–S261. 2000.3). Le Guyader FS., Mittelholzer C., Haugarreau L., Hedlund KO., Alsterlund R., Pommepuy M., Svensson L. Detection of noroviruses in rasberries associated with a gastroenteritis outbreak. Int J Food Microbiol. 97:179–186. 2004.4). Okame M., Shiota T., Hansman G., Takagi M., Yagyu F., Takanashi S., Phan TG., Shimizu Y., Kohno H., Okitsu S., Ushijima H. Anti-norovirus polyclonal antibody and its potential for development of an antigen-ELISA. J Med Virol. 79:1180–1186. 2007.
Article5). Shiota T., Okame M., Takanashi S., Khamrin P., Takagi M., Satou K., Masuoka Y., Yagyu F., Shimizu Y., Kohno H., Mizuguchi M., Okitsu S., Ushijima H. Characterization of a broadly reactive monoclonal antibody against norovirus genogroups I and II: recognition of a novel conformational epitope. J Virol. 81:12298–12306. 2007.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Expression of human norovirus VP1 gene and VP1-specific monoclonal antibodies
- Recent advances in norovirus infection
- Genetic Characterization of Norovirus GII.3 Circulating in Korea
- An Norovirus Outbreak at a Local Festival in Chungnam Korea
- The Genetic Diversity of Norovirus from Children with Diarrhea in Gwangju Metropolitan City, Korea: 2008-2012