Korean Circ J.
2023 Aug;53(8):535-547. 10.4070/kcj.2022.0348.
Comparison of Veno-arterial Extracorporeal Membrane Oxygenation Configurations for Patients Listed for Heart Transplantation
- Affiliations
-
- 1Department of Thoracic and Cardiovascular Surgery, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- 2Division of Cardiology, Department of Internal Medicine, Hallym Sacred Heart Hospital, Hallym University College of Medicine, Anyang, Korea
- 3Division of Cardiology, Department of Internal Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- 4Department of Clinical Epidemiology and Biostatistics, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- KMID: 2545150
- DOI: http://doi.org/10.4070/kcj.2022.0348
Abstract
- Background and Objectives
Veno-arterial extracorporeal membrane oxygenation (VAECMO) as a bridge to eventual heart transplantation (HT) is increasingly used worldwide. However, the effect of different VA-ECMO types on HT outcomes remains unclear.
Methods
This was a retrospective observational study of 111 patients receiving VA-ECMO and awaiting HT. We assessed 3 ECMO configuration groups: peripheral (n=76), central (n=12), and peripheral to central ECMO conversion (n=23). Cox proportional hazards regression and landmark analysis were conducted to analyze the effect of the ECMO configuration on HT and in-hospital mortality rates. We also evaluated adverse events during ECMO support.
Results
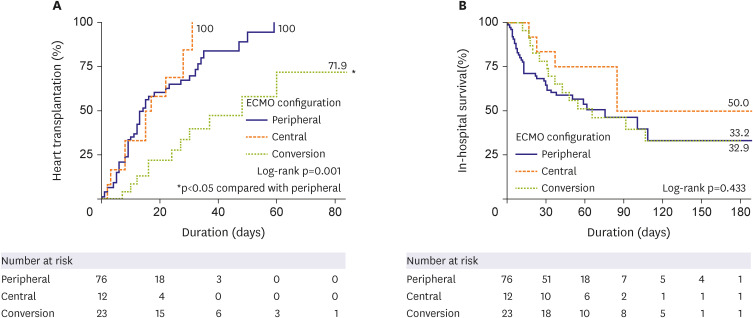
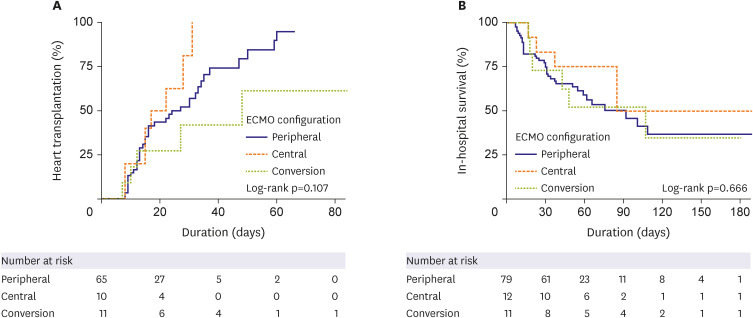
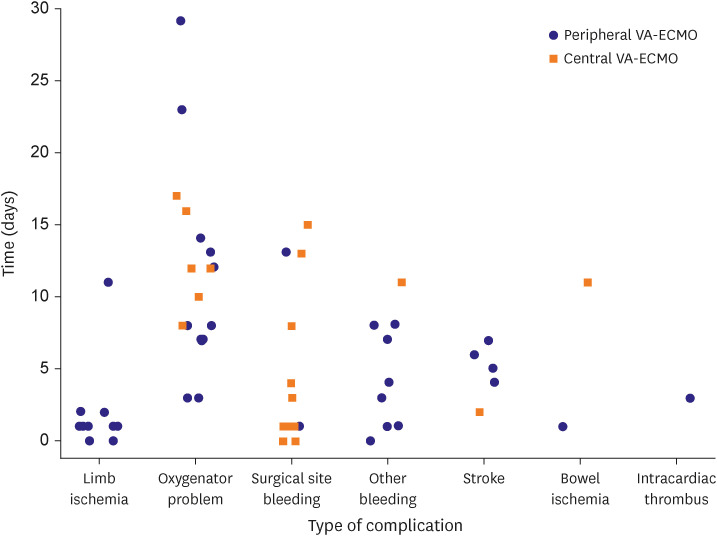
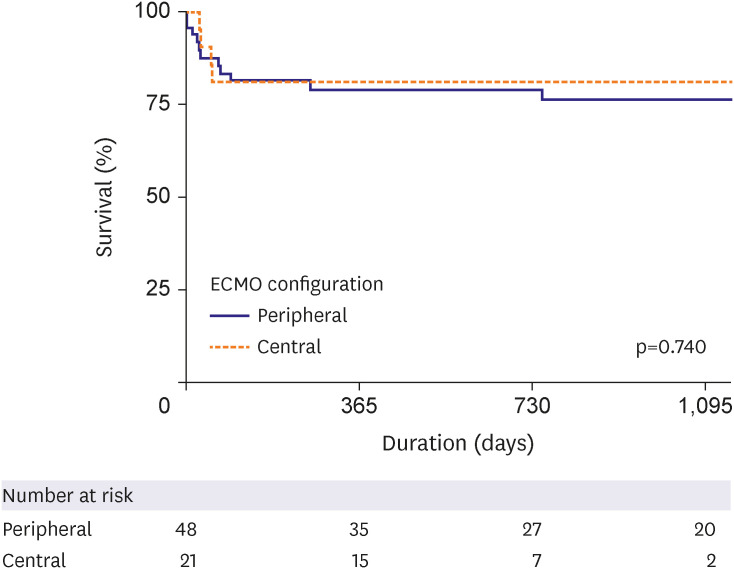
HT was performed in the peripheral (n=48, 63.2%), central (n=10, 83.3%), and conversion (n=11, 47.8%) ECMO groups (p=0.133) with a median interval of 10.5, 16, and 30 days, respectively (p<0.001). The cumulative incidence of HT was significantly lower in the conversion group (hazard ratio, 0.292, 95% confidence interval, 0.145–0.586, p=0.001). However, there was no difference in in-hospital mortality (log-rank p=0.433). In the landmark analysis, in-hospital mortality did not differ significantly among the 3 groups. Although we did note a trend toward lower HT in the conversion group, the difference was not statistically significant. Surgical site bleeding occurred mainly in the central, while limb ischemia occurred mainly in the peripheral groups.
Conclusions
We suggest that if patients are being stably supported with their initial ECMO configuration, whether it is central or peripheral, it should be maintained, and ECMO conversion should only be cautiously performed when necessary.
Figure
Cited by 1 articles
-
Editorial on Paper Titled Comparison of Veno-Arterial Extracorporeal Membrane Oxygenation Type in Patients Listed for Heart Transplantation
Jeong Hoon Yang
Korean Circ J. 2023;53(8):548-549. doi: 10.4070/kcj.2023.0168.
Reference
-
1. Khush KK, Hsich E, Potena L, et al. The international thoracic organ transplant registry of the International Society for Heart and Lung Transplantation: thirty-eighth adult heart transplantation report - 2021; focus on recipient characteristics. J Heart Lung Transplant. 2021; 40:1035–1049. PMID: 34419370.
Article2. Combes A, Price S, Slutsky AS, Brodie D. Temporary circulatory support for cardiogenic shock. Lancet. 2020; 396:199–212. PMID: 32682486.
Article3. Tehrani BN, Truesdell AG, Psotka MA, et al. A standardized and comprehensive approach to the management of cardiogenic shock. JACC Heart Fail. 2020; 8:879–891. PMID: 33121700.
Article4. DeFilippis EM, Clerkin K, Truby LK, et al. ECMO as a bridge to left ventricular assist device or heart transplantation. JACC Heart Fail. 2021; 9:281–289. PMID: 33714743.
Article5. Jasseron C, Lebreton G, Cantrelle C, et al. Impact of heart transplantation on survival in patients on venoarterial extracorporeal membrane oxygenation at listing in France. Transplantation. 2016; 100:1979–1987. PMID: 27306536.
Article6. Montisci A, Donatelli F, Cirri S, Coscioni E, Maiello C, Napoli C. Veno-arterial extracorporeal membrane oxygenation as bridge to heart transplantation: the way forward. Transplant Direct. 2021; 7:e720. PMID: 34258387.
Article7. Lechiancole A, Sponga S, Isola M, Vendramin I, Maiani M, Livi U. Heart transplantation in patients supported by ECMO: is the APACHE IV score a predictor of survival? Artif Organs. 2018; 42:670–673. PMID: 29392738.
Article8. Kim D, Choi JO, Oh J, et al. The Korean Organ Transplant Registry (KOTRY): second official adult heart transplant report. Korean Circ J. 2019; 49:724–737. PMID: 31074219.
Article9. Kim IC, Youn JC, Kobashigawa JA. The past, present and future of heart transplantation. Korean Circ J. 2018; 48:565–590. PMID: 29968430.
Article10. Kim IC, Youn JC, Lee SE, Jung SH, Kim JJ. Donor heart utilization in Korea. Int J Heart Fail. 2020; 2:254–263. PMID: 36262172.
Article11. Fukuhara S, Takeda K, Kurlansky PA, Naka Y, Takayama H. Extracorporeal membrane oxygenation as a direct bridge to heart transplantation in adults. J Thorac Cardiovasc Surg. 2018; 155:1607–1618.e6. PMID: 29361299.
Article12. Barge-Caballero E, Almenar-Bonet L, Gonzalez-Vilchez F, et al. Clinical outcomes of temporary mechanical circulatory support as a direct bridge to heart transplantation: a nationwide Spanish registry. Eur J Heart Fail. 2018; 20:178–186. PMID: 28949079.
Article13. Poptsov V, Spirina E, Dogonasheva A, Zolotova E. Five years’ experience with a peripheral veno-arterial ECMO for mechanical bridge to heart transplantation. J Thorac Dis. 2019; 11:S889–S901. PMID: 31183168.
Article14. Lui C, Fraser CD 3rd, Suarez-Pierre A, et al. Evaluation of extracorporeal membrane oxygenation therapy as a bridging method. Ann Thorac Surg. 2021; 112:68–74. PMID: 33098881.
Article15. Raffa GM, Kowalewski M, Brodie D, et al. Meta-analysis of peripheral or central extracorporeal membrane oxygenation in postcardiotomy and non-postcardiotomy shock. Ann Thorac Surg. 2019; 107:311–321. PMID: 29959943.
Article16. Cheng R, Hachamovitch R, Kittleson M, et al. Complications of extracorporeal membrane oxygenation for treatment of cardiogenic shock and cardiac arrest: a meta-analysis of 1,866 adult patients. Ann Thorac Surg. 2014; 97:610–616. PMID: 24210621.
Article17. Mariscalco G, Salsano A, Fiore A, et al. Peripheral versus central extracorporeal membrane oxygenation for postcardiotomy shock: multicenter registry, systematic review, and meta-analysis. J Thorac Cardiovasc Surg. 2020; 160:1207–1216.e44. PMID: 31864699.
Article18. Moonsamy P, Axtell AL, Ibrahim NE, et al. Survival after heart transplantation in patients bridged with mechanical circulatory support. J Am Coll Cardiol. 2020; 75:2892–2905. PMID: 32527398.
Article19. Barge-Caballero E, Segovia-Cubero J, Almenar-Bonet L, et al. Preoperative INTERMACS profiles determine postoperative outcomes in critically ill patients undergoing emergency heart transplantation: analysis of the Spanish National Heart Transplant Registry. Circ Heart Fail. 2013; 6:763–772. PMID: 23674362.
Article20. Liu J, Yang BQ, Itoh A, Masood MF, Hartupee JC, Schilling JD. Impact of new UNOS allocation criteria on heart transplant practices and outcomes. Transplant Direct. 2021; 7:e642. PMID: 33335981.
Article21. Parker WF, Chung K, Anderson AS, Siegler M, Huang ES, Churpek MM. Practice changes at U.S. transplant centers after the new adult heart allocation policy. J Am Coll Cardiol. 2020; 75:2906–2916. PMID: 32527399.
Article22. Smith M, Vukomanovic A, Brodie D, Thiagarajan R, Rycus P, Buscher H. Duration of veno-arterial extracorporeal life support (VA ECMO) and outcome: an analysis of the Extracorporeal Life Support Organization (ELSO) registry. Crit Care. 2017; 21:45. PMID: 28264702.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Editorial on Paper Titled Comparison of Veno-Arterial Extracorporeal Membrane Oxygenation Type in Patients Listed for Heart Transplantation
- Right ventricular assist device with an oxygenator using extracorporeal membrane oxygenation as a bridge to lung transplantation in a patient with severe respiratory failure and right heart decompensation
- Extracorporeal Membrane Oxygenation for 67 Days as a Bridge to Heart Transplantation in a Postcardiotomy Patient with Failing Heart and Mediastinitis
- Extracorporeal Membrane Oxygenation for Complicated Scrub Typhus
- Veno-venous Extracorporeal Membrane Oxygenation with a Double Lumen Catheter for Pediatric Pulmonary Support