Arch Hand Microsurg.
2020 Mar;25(1):24-32. 10.12790/ahm.19.0067.
Intraoperative Arthroscopic Findings of Ulnar Impaction Syndrome
- Affiliations
-
- 1Department of Orthopedic Surgery, Yeouido St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea
- KMID: 2501107
- DOI: http://doi.org/10.12790/ahm.19.0067
Abstract
- Purpose
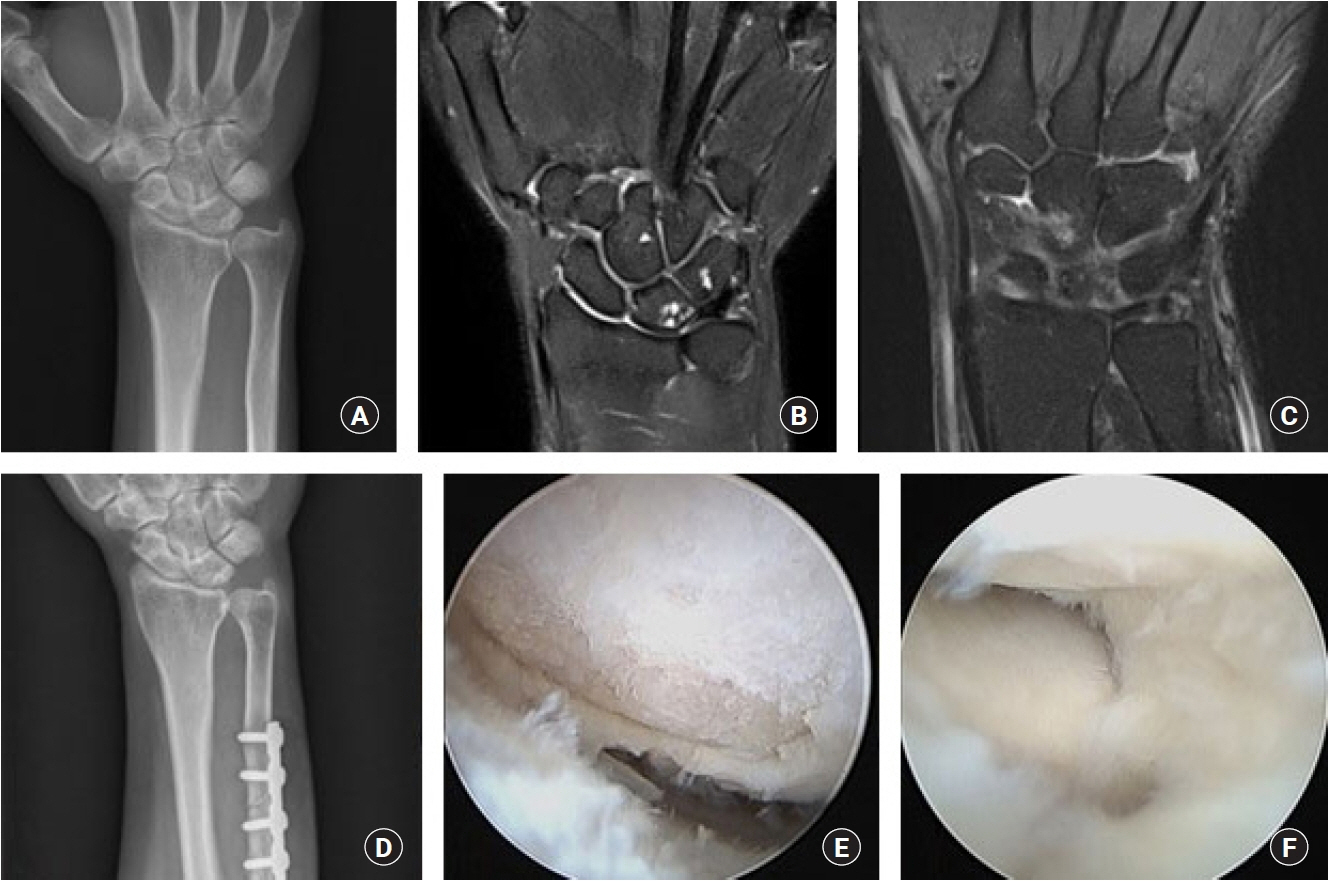
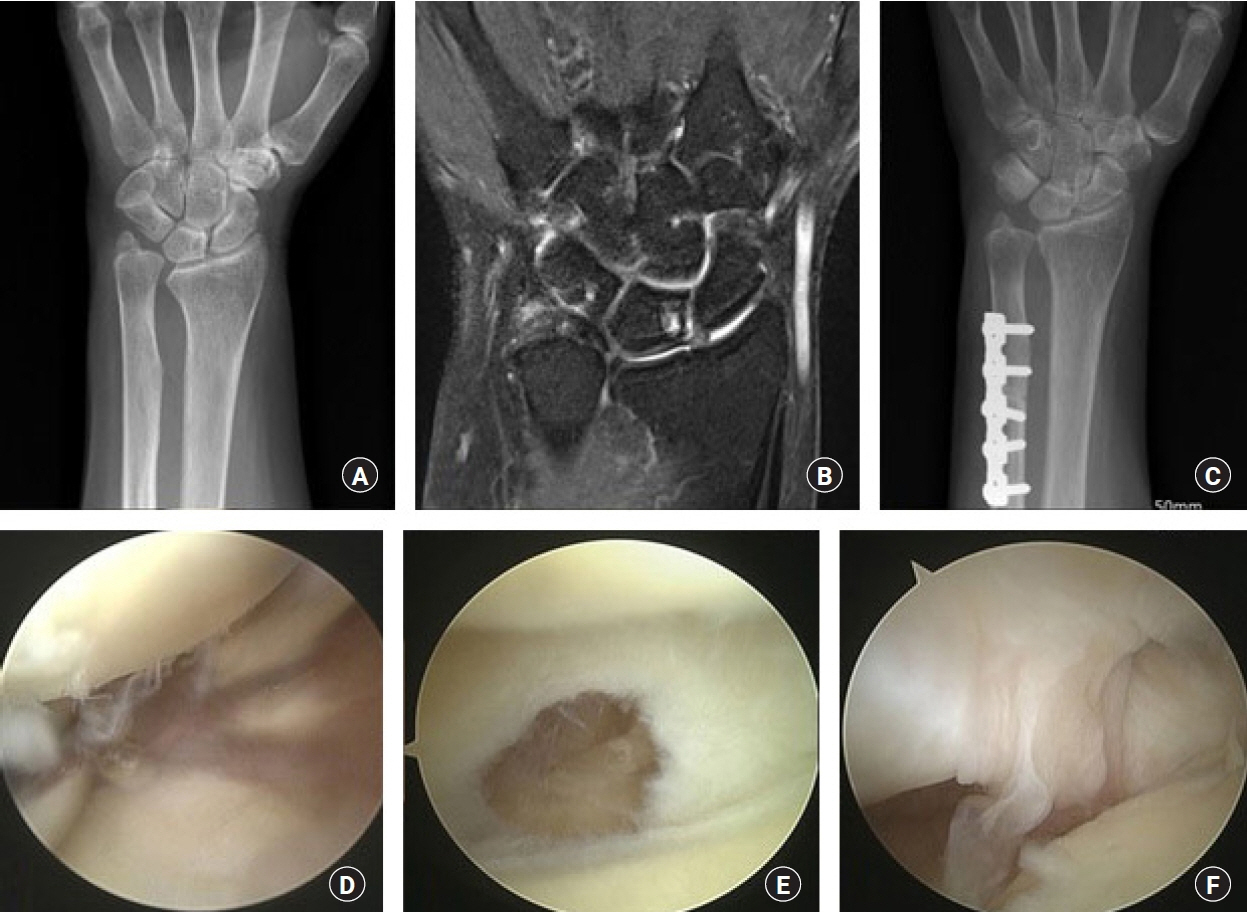
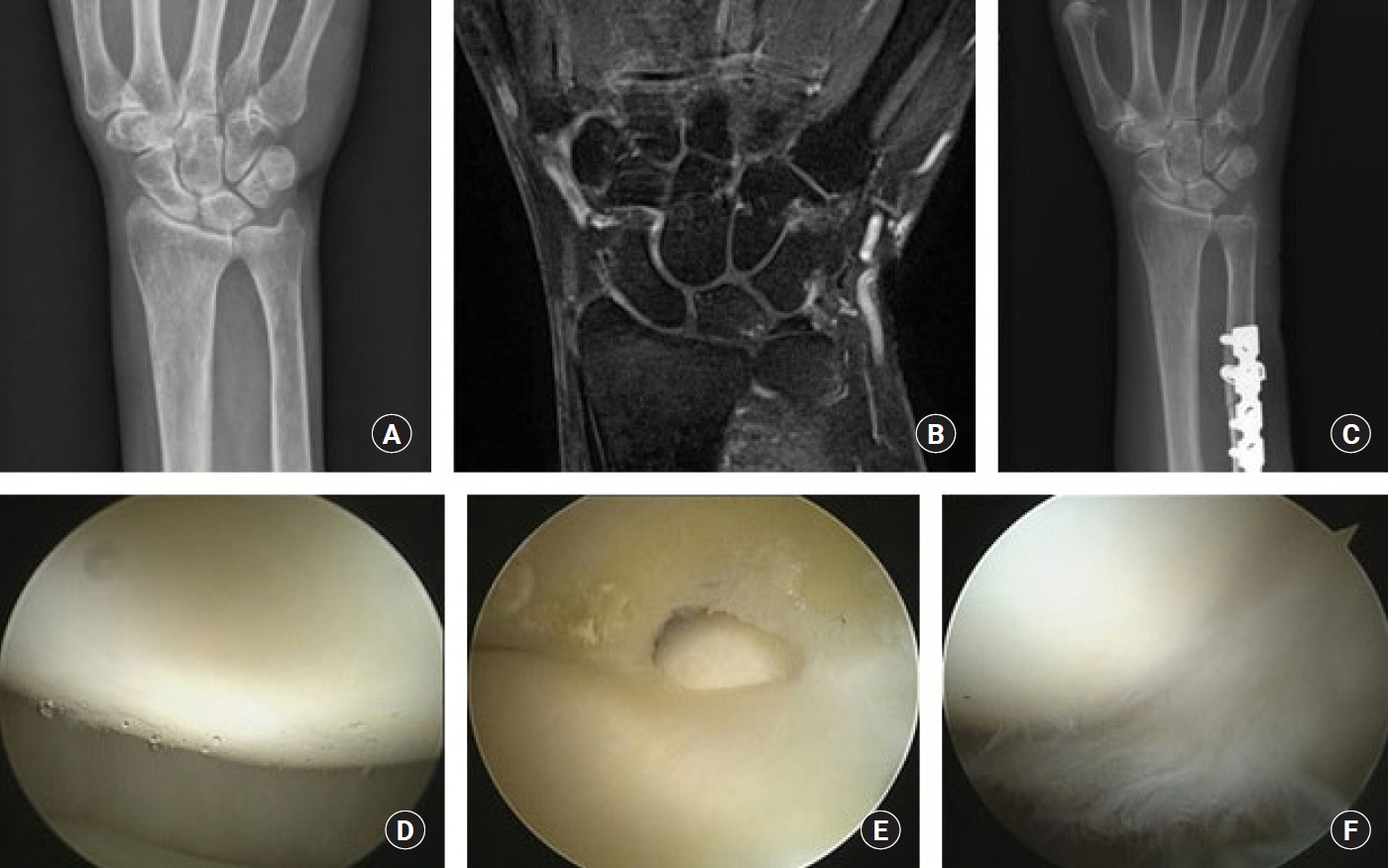
The purpose of this study was to compare the preoperative magnetic resonance imaging (MRI) findings and intraoperative arthroscopic findings in ulnar impaction syndrome, and to assess the necessity of the arthroscopic evaluation in ulnar shortening osteotomy.
Methods
We retrospectively reviewed 46 patients of idiopathic ulnar impaction syndrome, treated by ulnar shortening osteotomy and arthroscopic evaluation, from February 2012 to June 2018. In preoperative MRI and intraoperative arthroscopy, lunate, triquetrum, triangular fibrocartilage complex (TFCC) and lunotriquetral ligament were evaluated.
Results
The chance to see the findings of chondromalacia of the lunate and triquetral cartilage in arthroscopy (58% and 2%) was less than the findings of abnormal signal intensity of the lunate and triquetrum in MRI (65% and 22%). The incidence of TFCC lesion in arthroscopy (89%) was almost equal with that of abnormal signal intensity of TFCC in MRI findings (87%). But there were 24 cases (52%) where the prediction of triangular fibrocartilage lesions in MRI was incorrect. In 19 cases (41%), arthroscopic findings were not appropriate with the disease stage of Palmer’s original classification.
Conclusion
Although some recent reports say that arthroscopic surgery is not necessary in the treatment of ulnar impaction syndrome, we could confirm the value of arthroscopic evaluation for the identification of ulnocarpal pathologies. We would like to recommend the arthroscopic evaluation of ulnocarpal joint at the same time of the ulnar shortening procedure.
Figure
Reference
-
References
1. Friedman SL, Palmer AK. The ulnar impaction syndrome. Hand Clin. 1991; 7:295–310.
Article2. Bernstein MA, Nagle DJ, Martinez A, Stogin JM Jr, Wiedrich TA. A comparison of combined arthroscopic triangular fibrocartilage complex debridement and arthroscopic wafer distal ulna resection versus arthroscopic triangular fibrocartilage complex debridement and ulnar shortening osteotomy for ulnocarpal abutment syndrome. Arthroscopy. 2004; 20:392–401.
Article3. Cheon SJ, Kang JH, Cho JY, Lim JM. Treatment of ulnar impaction syndrome using arthroscopy and ulnar shortening osteotomy. J Korean Orthop Assoc. 2007; 42:310–7.
Article4. Park JW, Kim SK, Park JH, Wang JH, Lee SJ, Jeon WJ. Results of Ulnar Shortening for Ulnar Impaction Syndrome in the Wrist. J Korean Soc Surg Hand. 2006; 11:14–9.5. Kim BS, Song HS. A comparison of ulnar shortening osteotomy alone versus combined arthroscopic triangular fibrocartilage complex debridement and ulnar shortening osteotomy for ulnar impaction syndrome. Clin Orthop Surg. 2011; 3:184–90.
Article6. Baek GH, Chung MS, Lee YH, Jeong GI, Lee CH. Arthroscopy of the wrist and ulnar shortening osteotomy for the treatment of the ulnar impaction syndrome. J Korean Orthop Assoc. 2001; 36:207–14.
Article7. Sachar K. Ulnar-sided wrist pain: evaluation and treatment of triangular fibrocartilage complex tears, ulnocarpal impaction syndrome, and lunotriquetral ligament tears. J Hand Surg Am. 2008; 33:1669–79.
Article8. Imaeda T, Nakamura R, Shionoya K, Makino N. Ulnar impaction syndrome: MR imaging findings. Radiology. 1996; 201:495–500.
Article9. Escobedo EM, Bergman AG, Hunter JC. MR imaging of ulnar impaction. Skeletal Radiol. 1995; 24:85–90.
Article10. Minami A, Ishikawa J, Suenaga N, Kasashima T. Clinical results of treatment of triangular fibrocartilage complex tears by arthroscopic debridement. J Hand Surg Am. 1996; 21:406–11.
Article11. Kreder HJ, Hanel DP, McKee M, Jupiter J, McGillivary G, Swiontkowski MF. X-ray film measurements for healed distal radius fractures. J Hand Surg Am. 1996; 21:31–9.
Article12. Palmer AK. Triangular fibrocartilage complex lesions: a classification. J Hand Surg Am. 1989; 14:594–606.
Article13. Chun S, Palmer AK. The ulnar impaction syndrome: follow-up of ulnar shortening osteotomy. J Hand Surg Am. 1993; 18:46–53.
Article14. Cerezal L, del Pinal F, Abascal F, Garcia-Valtuille R, Pereda T, Canga A. Imaging findings in ulnar-sided wrist impaction syndromes. Radiographics. 2002; 22:105–21.
Article15. Loh YC, Van Den Abbeele K, Stanley JK, Trail IA. The results of ulnar shortening for ulnar impaction syndrome. J Hand Surg Br. 1999; 24:316–20.
Article16. Hobby JL, Tom BD, Bearcroft PW, Dixon AK. Magnetic resonance imaging of the wrist: diagnostic performance statistics. Clin Radiol. 2001; 56:50–7.
Article17. Magee T. Comparison of 3-T MRI and arthroscopy of intrinsic wrist ligament and TFCC tears. AJR Am J Roentgenol. 2009; 192:80–5.
Article18. Koh KH, Lee HL, Chang YS, Park MJ. Arthroscopy during ulnar shortening for idiopathic ulnar impaction syndrome. Orthopedics. 2013; 36:e1495–500.
Article19. Park JC, Sur YJ, Rhee SK, Song SW, Lee SM, Han SH. Ulnar shortening osteotomy for the treatment of ulnar impaction syndrome. J Korean Soc Surg Hand. 2009; 14:172–8.20. Roh YH, Song JH, Gong HS, Baek GH. Comparison of clinical outcomes after ulnar shortening osteotomy for ulnar impaction syndrome with or without arthroscopic debridement. J Hand Surg Eur Vol. 2019; 44:589–93.21. Tatebe M, Shinohara T, Okui N, Yamamoto M, Hirata H, Imaeda T. Clinical, radiographic, and arthroscopic outcomes after ulnar shortening osteotomy: a long-term follow-up study. J Hand Surg Am. 2012; 37:2468–74.
Article22. Low S, Herold A, Unglaub F, Megerle K, Erne H. Treatment of ulnar impaction syndrome with and without central TFC lesion. J Wrist Surg. 2018; 7:133–40.23. Hulsizer D, Weiss AP, Akelman E. Ulna-shortening osteotomy after failed arthroscopic debridement of the triangular fibrocartilage complex. J Hand Surg Am. 1997; 22:694–8.
Article24. Ruch DS, Poehling GG. Arthroscopic management of partial scapholunate and lunotriquetral injuries of the wrist. J Hand Surg Am. 1996; 21:412–7.
Article25. Westkaemper JG, Mitsionis G, Giannakopoulos PN, Sotereanos DG. Wrist arthroscopy for the treatment of ligament and triangular fibrocartilage complex injuries. Arthroscopy. 1998; 14:479–83.
Article26. Weiss AP, Sachar K, Glowacki KA. Arthroscopic debridement alone for intercarpal ligament tears. J Hand Surg Am. 1997; 22:344–9.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Arthroscopy of the Wrist and Ulnar Shortening Osteotomy for the Treatment of the Ulnar Impaction Syndrome
- Treatment of Ulnar Impaction Syndrome using Arthroscopy and Ulnar Shortening Osteotomy
- Ulnar Shortening Osteotomy for the Treatment of Ulnar Impaction Syndrome
- A Comparison of Ulnar Shortening Osteotomy Alone Versus Combined Arthroscopic Triangular Fibrocartilage Complex Debridement and Ulnar Shortening Osteotomy for Ulnar Impaction Syndrome
- Updates on Ulnar Impaction Syndrome