Transl Clin Pharmacol.
2016 Dec;24(4):183-188. 10.12793/tcp.2016.24.4.183.
Pharmacokinetic characteristics of cilostazol 200 mg controlled-release tablet compared with two cilostazol 100 mg immediate-release tablets (Pletal) after single oral dose in healthy Korean male volunteers
- Affiliations
-
- 1Department of Medical Science, College of Medicine, Chungnam National University, Daejeon 35015, Republic of Korea. boniii@cnu.ac.kr
- 2Korea United Pharm.Inc., Seoul 06116, Republic of Korea.
- 3Caleb Multilab Inc., Seoul 06745, Republic of Korea.
- 4Clinical Trials Center, Chungnam National University Hospital, Daejeon 35015, South Korea.
- 5Department of Pharmacology, College of Medicine, Chungnam National University, Daejeon 35015, Republic of Korea.
- KMID: 2413840
- DOI: http://doi.org/10.12793/tcp.2016.24.4.183
Abstract
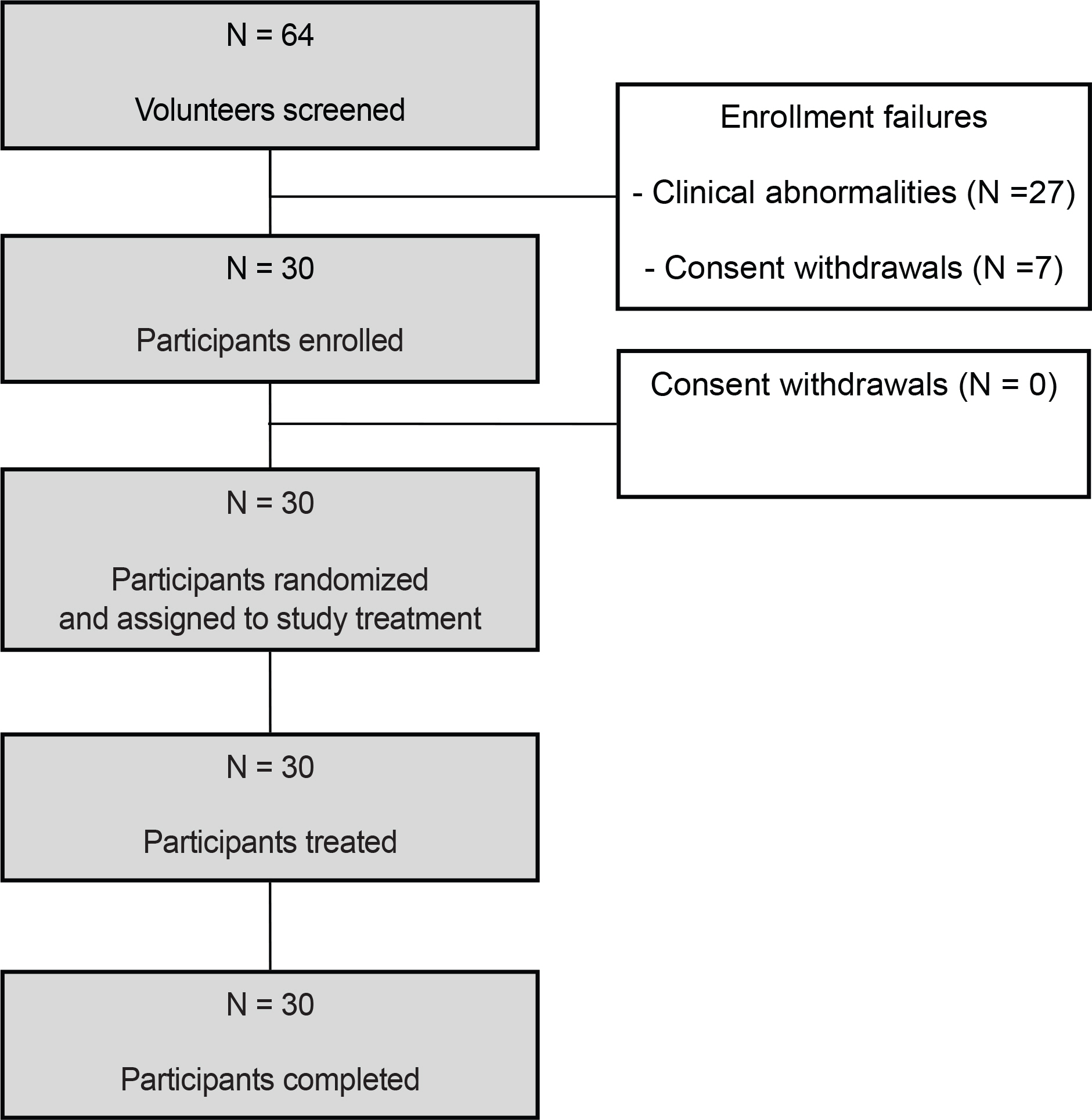
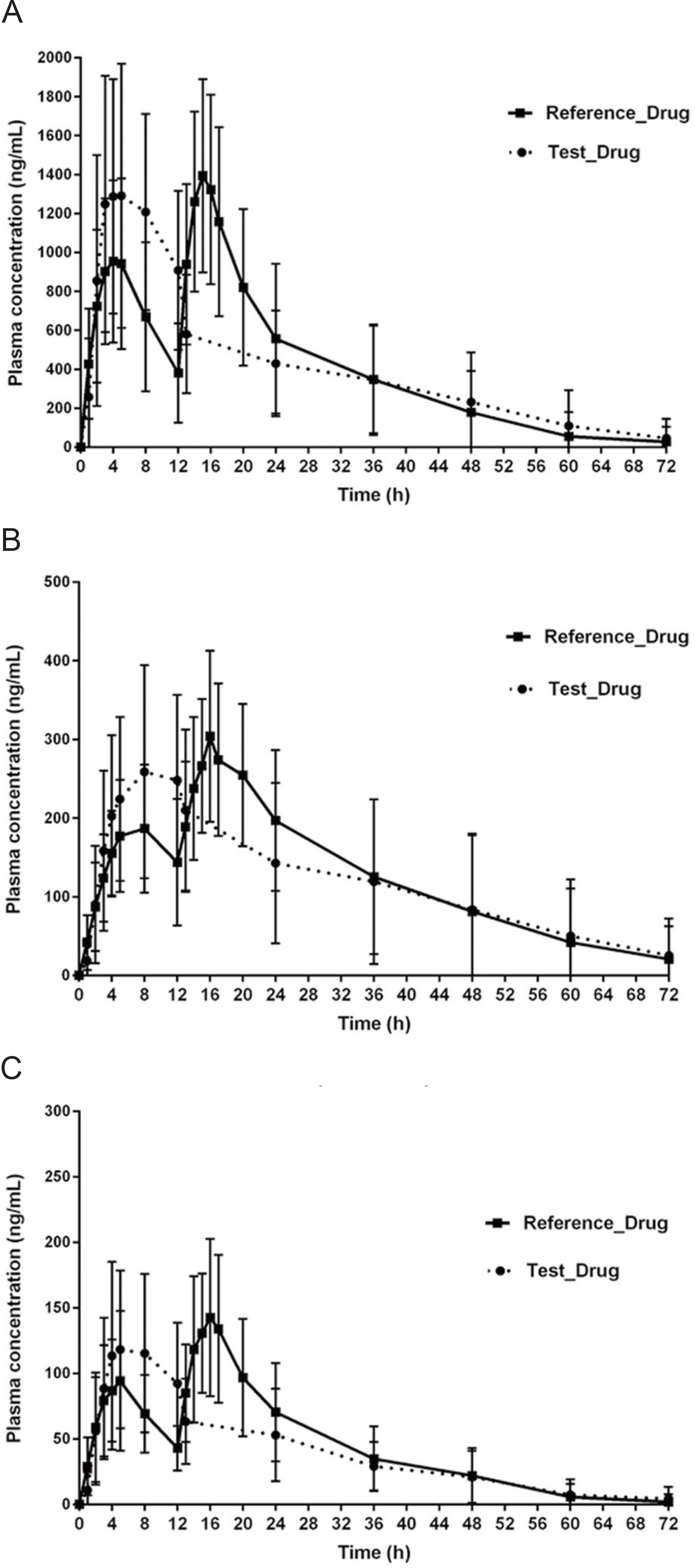
- Cilostazol controlled-release (CR) tablets have recently been developed by Korea United Pharm (Seoul, Korea). The tablets use a patented double CR system, which improves drug compliance by allowing "once daily" administration and reduces adverse events by sustaining a more even plasma concentration for 24 h. We conducted an open, randomized, two-period, two-treatment, crossover study to compare the pharmacokinetic (PK) characteristics and tolerability of cilostazol when administered to healthy Korean male volunteers as CR or immediate release (IR) tablets (Pletal, Korea Otsuka Pharmaceutical Co., Gyeonggi-do, Korea). Each volunteer was randomly allocated to receive a single tablet of cilostazol CR (200 mg) or two tablets of cilostazol IR (100 mg) with a 7-day washout period between treatments. Plasma cilostazol, OPC-13015 (3,4-dehydrocilostazol), and OPC-13213 (4'-trans-hydroxycilostazol) were assayed using liquid chromatography-tandem mass spectrometry for PK analysis. Thirty participants completed the study with no clinically relevant safety issues. The peak concentrations (C(max), mean ± SD) of cilostazol CR and cilostazol IR were 1414.6 ± 49.3 and 1413.1 ± 35.2 ng/mL, respectively, and the areas under the plasma concentration-time curve from 0 to the last concentration (AUC(last)) were 23928.7 ± 65.9 and 25312.0 ± 62.6 ng·h/mL, respectively. The geometric mean ratios (cilostazol CR/cilostazol IR, GMR) of the C(max) and AUC(last) values were 1.001 (90% CI: 0.822, 1.220) and 0.945 (90% CI: 0.814, 1.098), respectively. The frequencies of adverse events were similar. The present study showed that cilostazol PK and tolerability were comparable when administered to healthy Korean men, regardless of whether administered as cilostazol CR or IR.
Keyword
MeSH Terms
Figure
Reference
-
1.Sorkin EM., Markham A. Cilostazol. Drugs Aging. 1999. 14:63–71.
Article2.Horie N., Kaminogo M., Izumo T., Hayashi K., Tsujino A., Nagata I. Cilostazol may prevent cardioembolic stroke in patients undergoing antiplatelet therapy. Neurol Res. 2015. 37:619–623. DOI: doi: 10.1179/1743132815Y.0000000021.
Article3.Liu JS., Chuang TJ., Chen JH., Lee CH., Hsieh CH., Lin TK, et al. Cilostazol attenuates the severity of peripheral arterial occlusive disease in patients with type 2 diabetes: the role of plasma soluble receptor for advanced glycation end-products. Endocrine. 2015. 49:703–710. DOI: doi: 10.1007/s12020-015-0545-6.
Article4.Tanaka T., Ishikawa T., Hagiwara M., Onoda K., Itoh H., Hidaka H. Effects of cilostazol, a selective cAMP phosphodiesterase inhibitor on the contraction of vascular smooth muscle. Pharmacology. 1988. 36:313–320.
Article5.Dawson DL., Cutler BS., Meissner MH., Strandness DE Jr. Cilostazol has beneficial effects in treatment of intermittent claudication. Results from a multi-center, randomized, prospective, double-blind trial. Circulation. 1998. 98:678–686.
Article6.Dawson DL., Cutler BS., Hiatt WR., Hobson RW 2nd., Martin JD., Bortey EB, et al. A comparison of cilostazol and pentoxifylline for treating intermittent claudication. Am J med Med. 2000. 109:523–530.
Article7.Shin KH., Yoon G., Yoon IS., Park JW. Preparation and evaluation of oral controlled-release cilostazol formulation: pharmacokinetics and antithrombotic efficacy in dogs and healthy male Korean participants. J Pharm Pharmacol. 2014. 66:961–974. DOI: doi: 10.1111/jphp.12227.
Article8.Karalis V., Macheras P. Examining the role of metabolites in bioequivalence assessment. J Pharm Pharm Sci. 2010. 13:198–217.
Article9.Srinivas NR. Considerations for metabolite pharmacokinetic data in bioavail-ability/bioequivalence assessments. Overview of the recent trends. Arzneimit-telforschung. 2009. 59:155–165. DOI: doi: 10.1055/s-0031-1296380.10.Jackson AJ., Robbie G., Marroum P. Metabolites and bioequivalence: past and present. Clin Pharmacokinet. 2004. 43:655–672.11.Kim HS., Kim GY., Yeo CW., Oh MK., Ghim JL., Shon JH, et al. The effect of Ginkgo biloba extracts on the pharmacokinetics and pharmacodynamics of cilostazol and its active metabolites in healthy Korean subjects. Br J Clin Pharmacol. 2014. 77:821–830. DOI: doi: 10.1111/bcp.12236.12.Kim YH., Ghim JL., Jung JA., Cho SH., Choe S., Choi HY, et al. Pharmacokinetic comparison of sustained- and immediate-release formulations of cilostazol after multiple oral doses in fed healthy male Korean volunteers. Drug Des Devel Ther. 2015. 9:3571–3577. DOI: doi: 10.2147/DDDT.S86845.13.Woo SK., Kang WK., Kwon KI. Pharmacokinetic and pharmacodynamic modeling of the antiplatelet and cardiov ascular effects of cilostazol in healthy humans. Clin Pharmacol Ther. 2002. 71:246–252.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Pharmacokinetic comparison of two valproic acid formulations: a plain and a controlled release enteric-coated tablets
- Pharmacokinetic Equivalence of the High Dose Strength Fixed-Dose Combination Tablet of Gemigliptin/Metformin Sustained Release (SR) and Individual Component Gemigliptin and Metformin XR Tablets in Healthy Subjects
- Pharmacokinetics and Safety of Levodropropizine Controlled Release Tablet after Repeated Dosing in Healthy Male Volunteers
- Comparative pharmacokinetic and tolerability evaluation of two simvastatin 20 mg formulations in healthy Korean male volunteers
- Pharmacokinetics and pharmacodynamics of a fixed-dose combination of gemigliptin/metformin sustained release 25/500 mg compared to the loose combination in healthy male subjects