J Korean Acad Conserv Dent.
2004 Nov;29(6):520-531. 10.5395/JKACD.2004.29.6.520.
Rheological properties of resin composites according to the change of monomer and filler compositions
- Affiliations
-
- 1Department of Conservative Dentistry, College of Dentistry, Seoul National University, Korea. inboglee@snu.ac.kr
- KMID: 2175653
- DOI: http://doi.org/10.5395/JKACD.2004.29.6.520
Abstract
OBJECTIVES
The aim of this study was to investigate the effect of monomer and filler compositions on the rheological properties related to the handling characteristics of resin composites.
METHODS
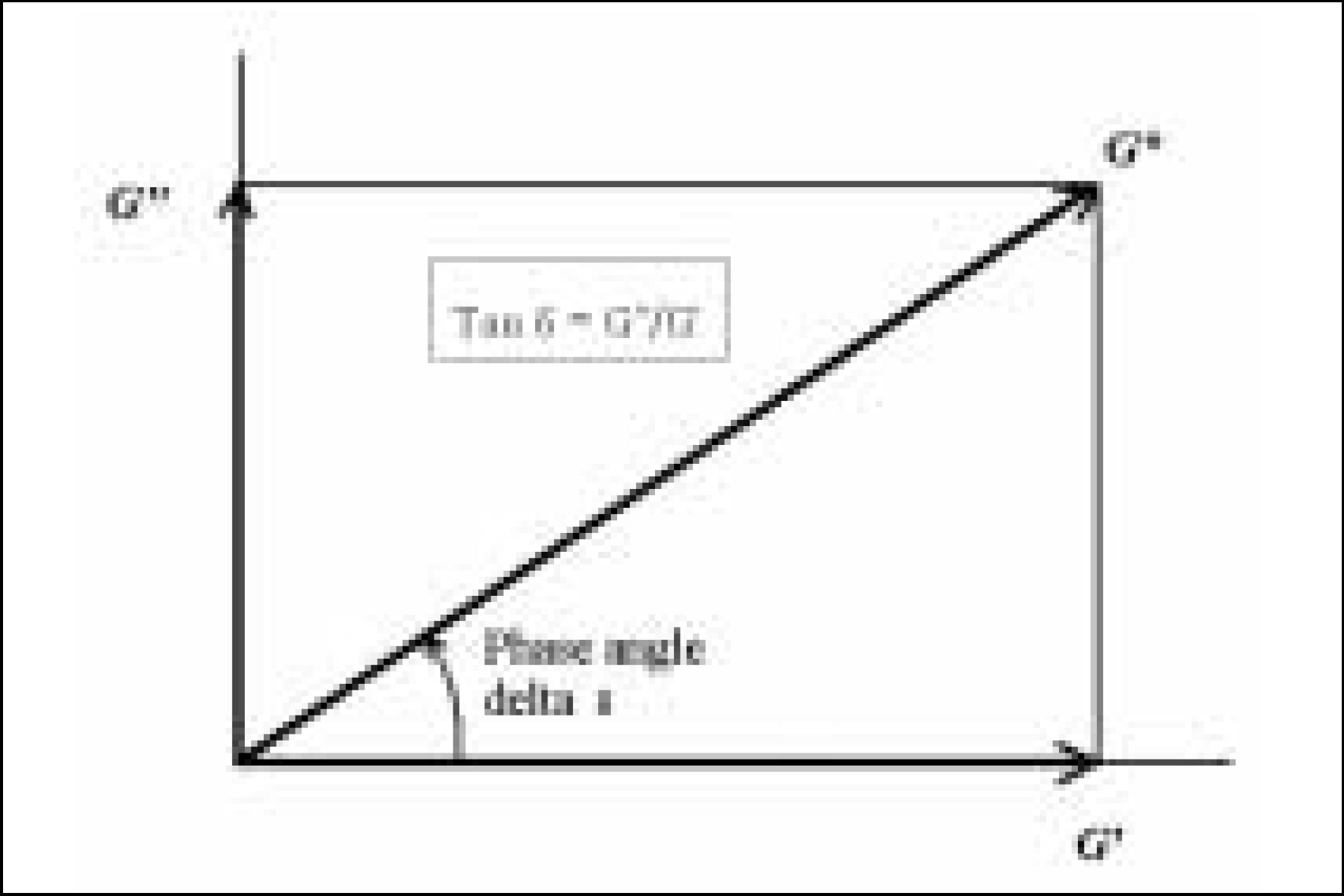
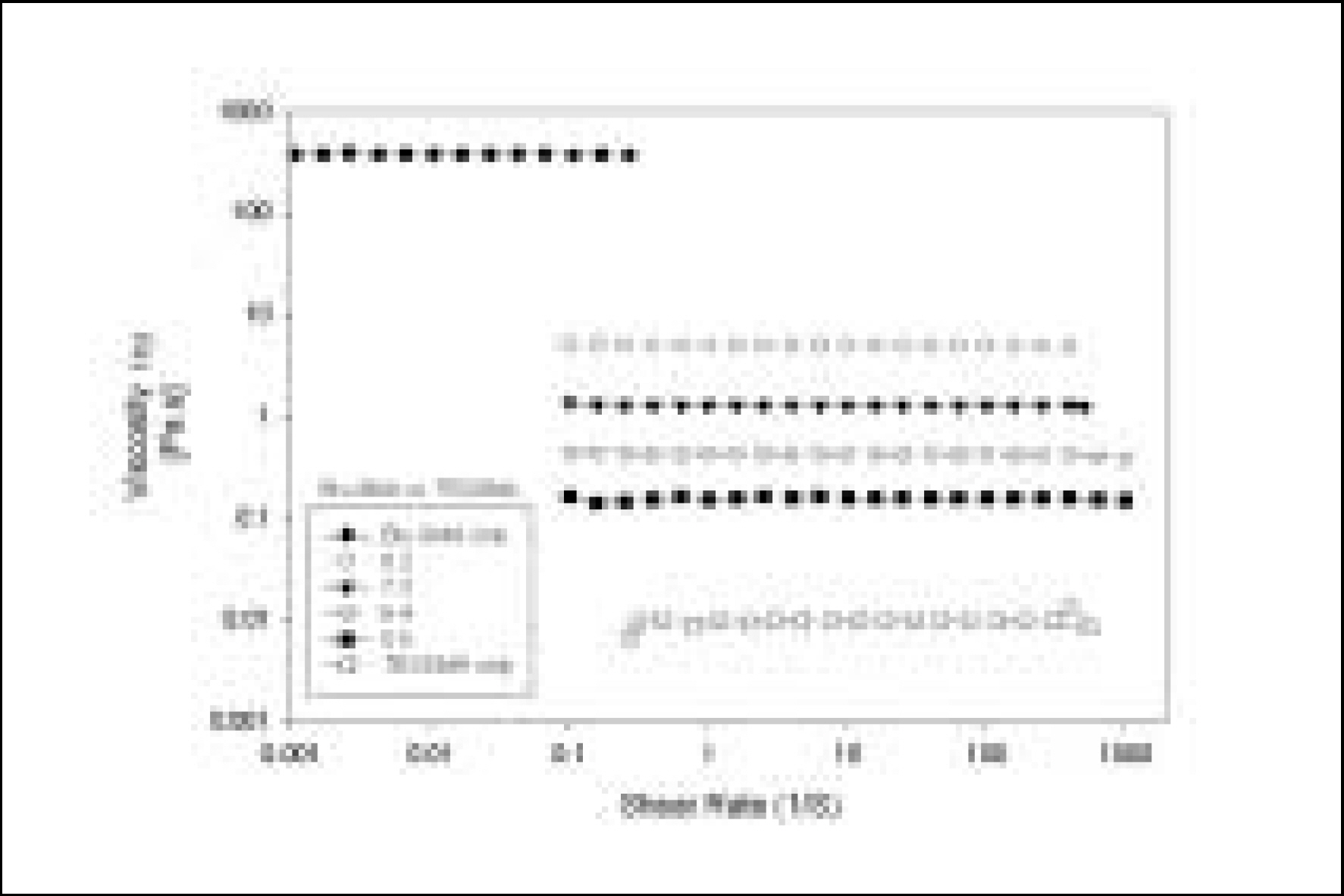
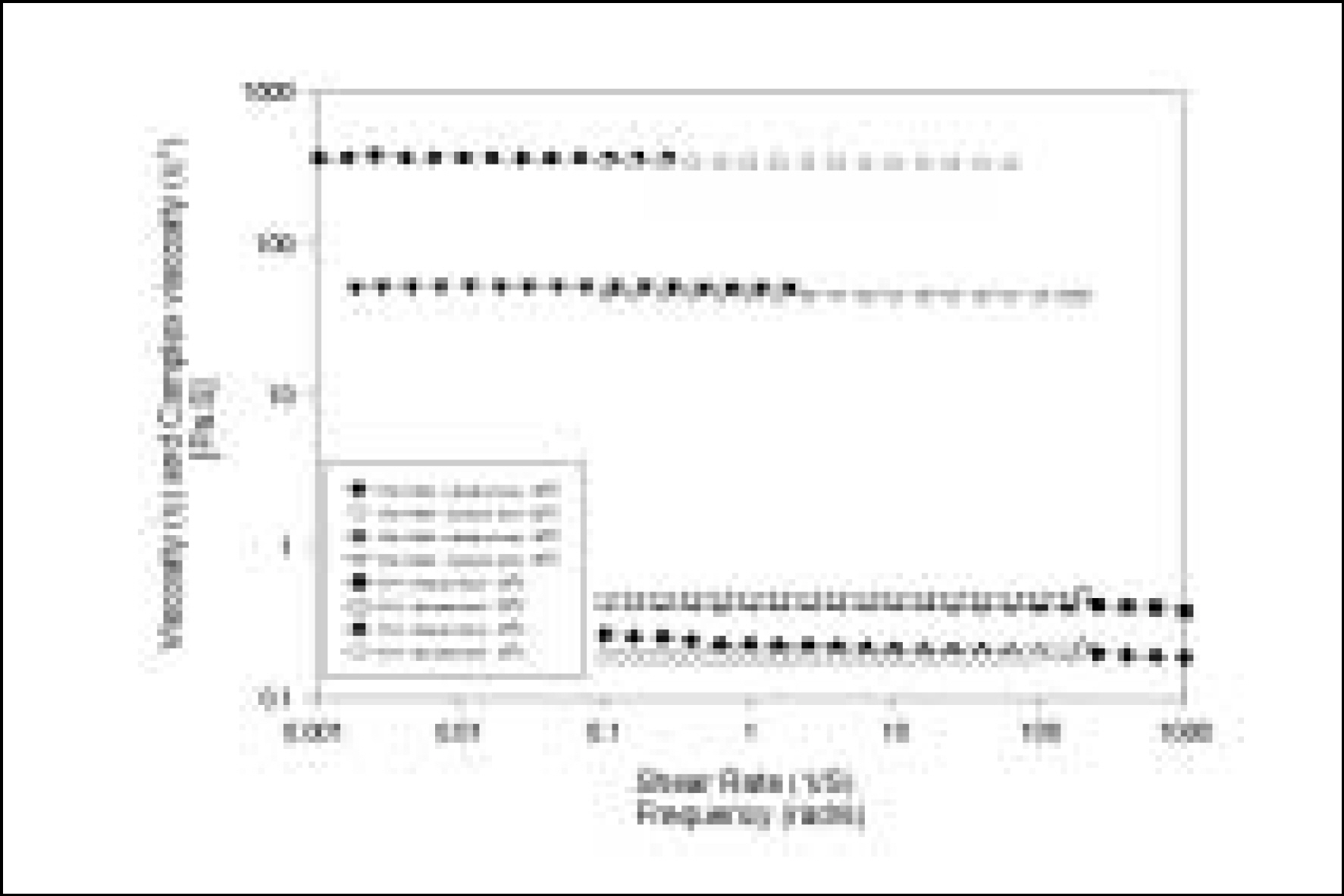
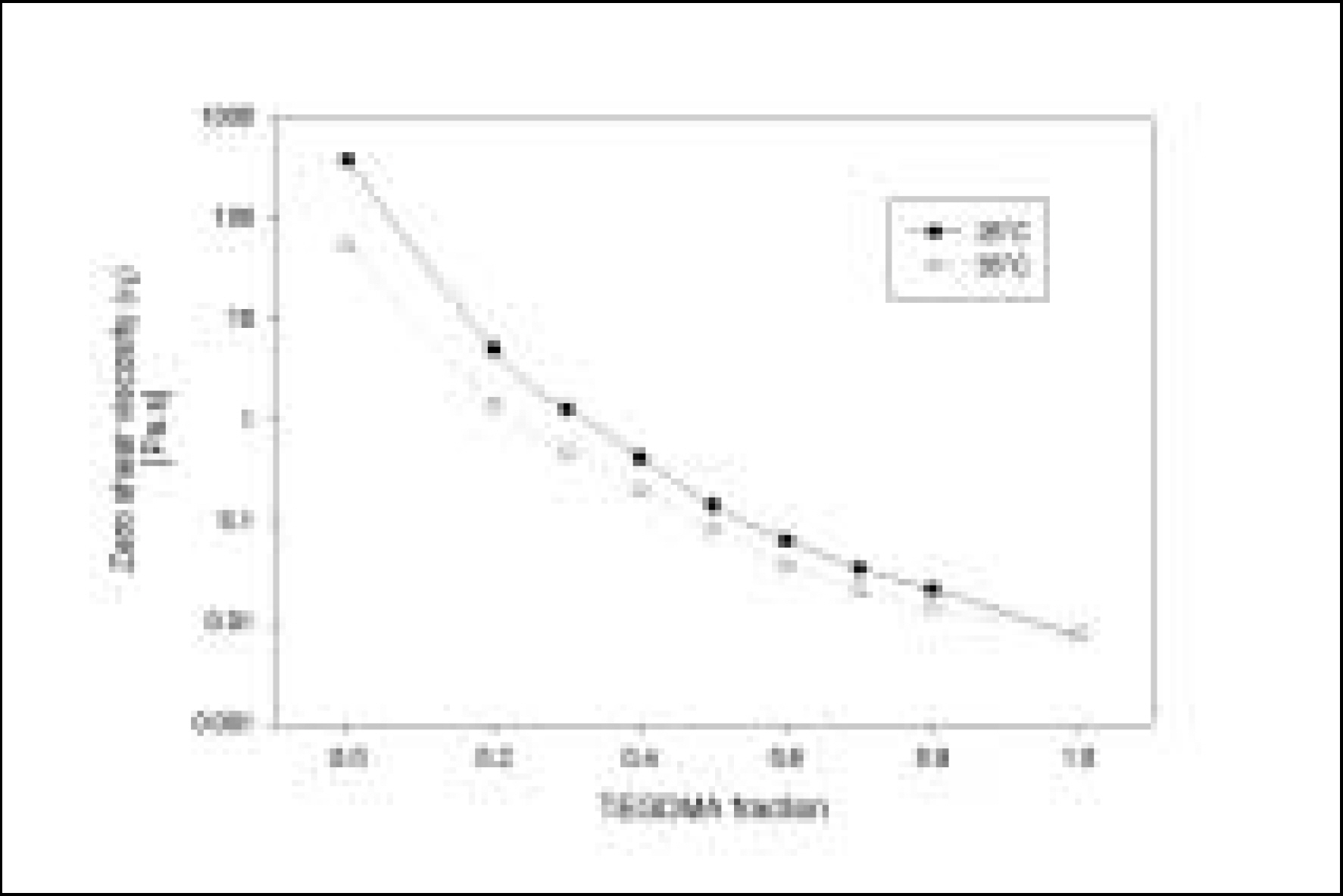
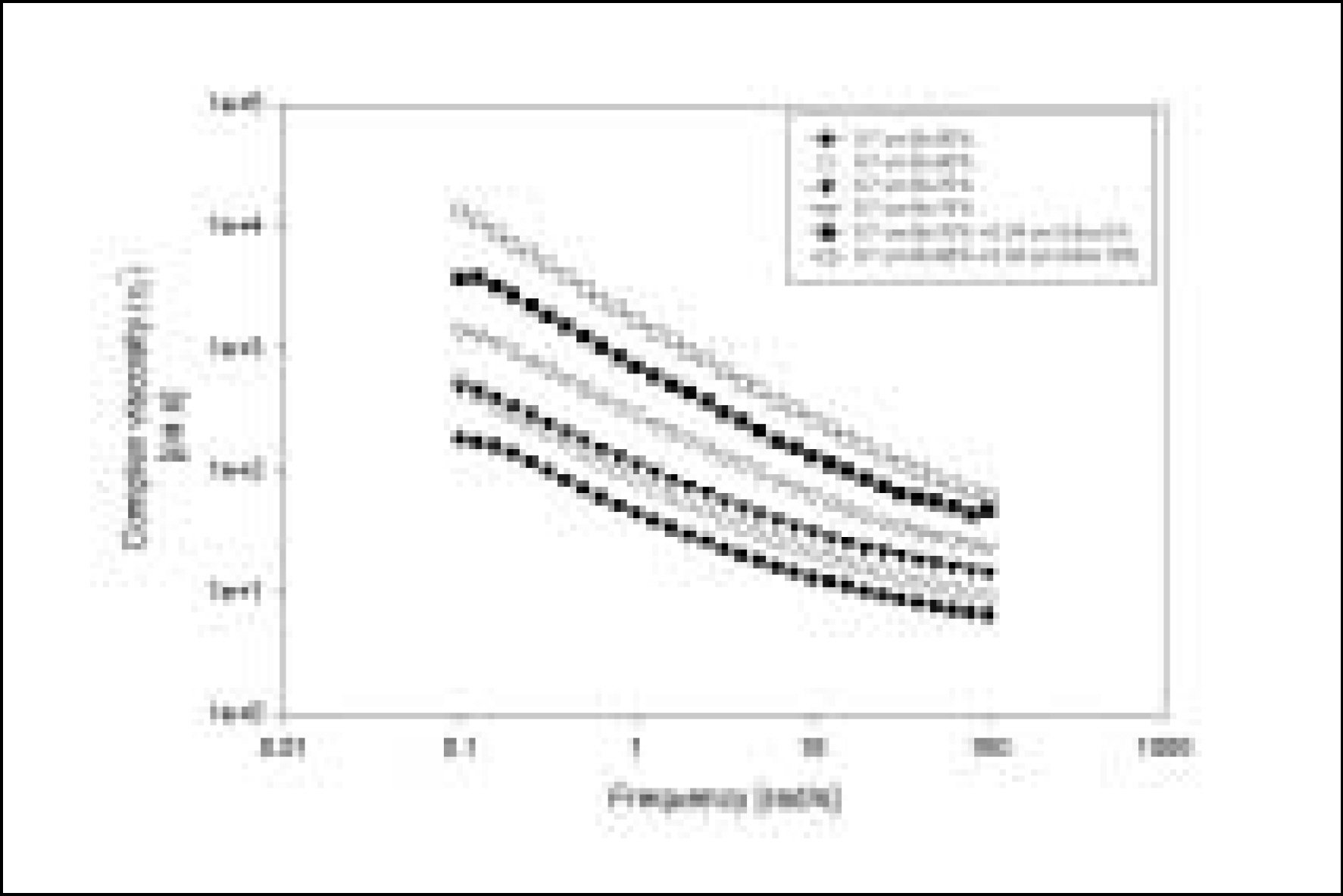
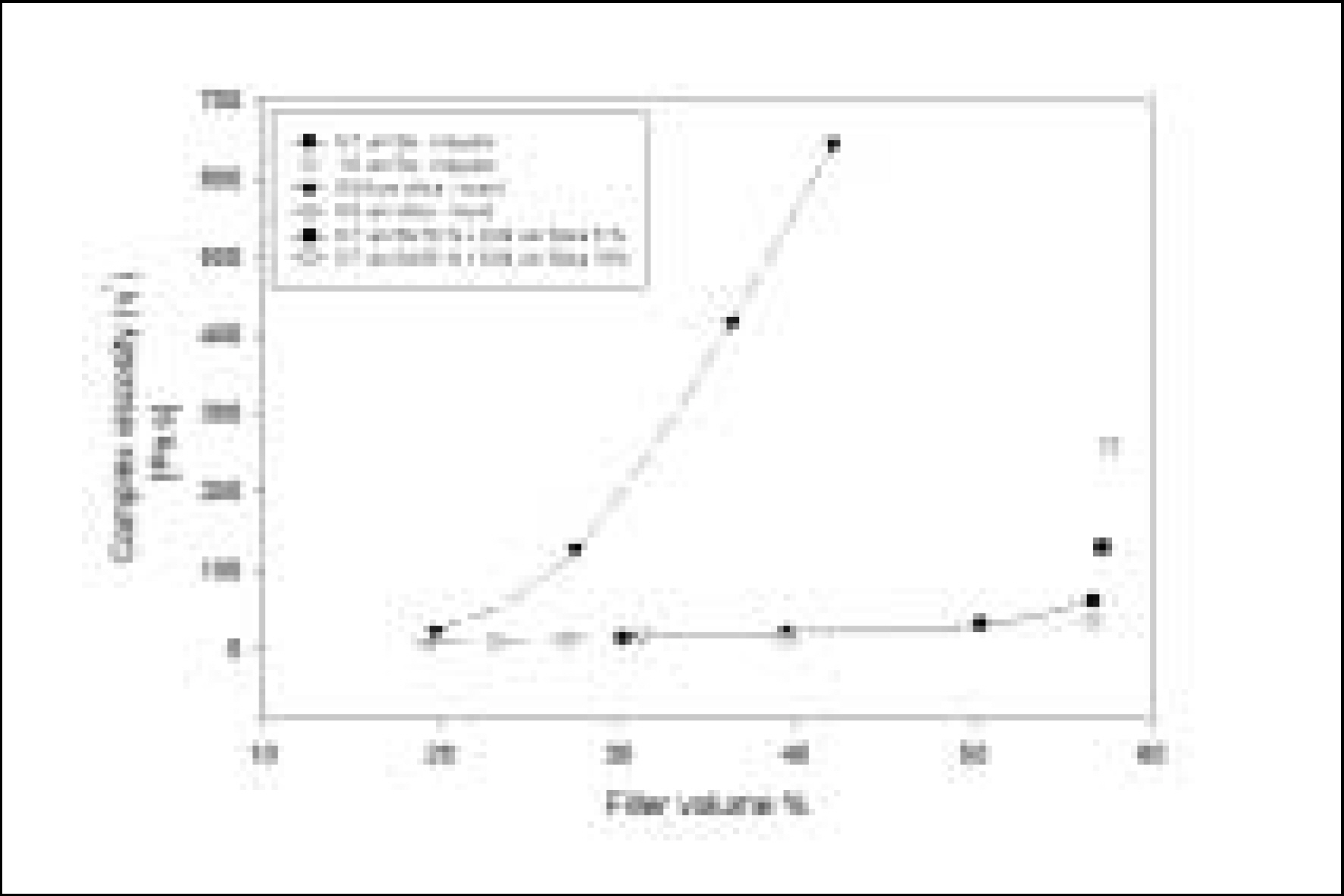
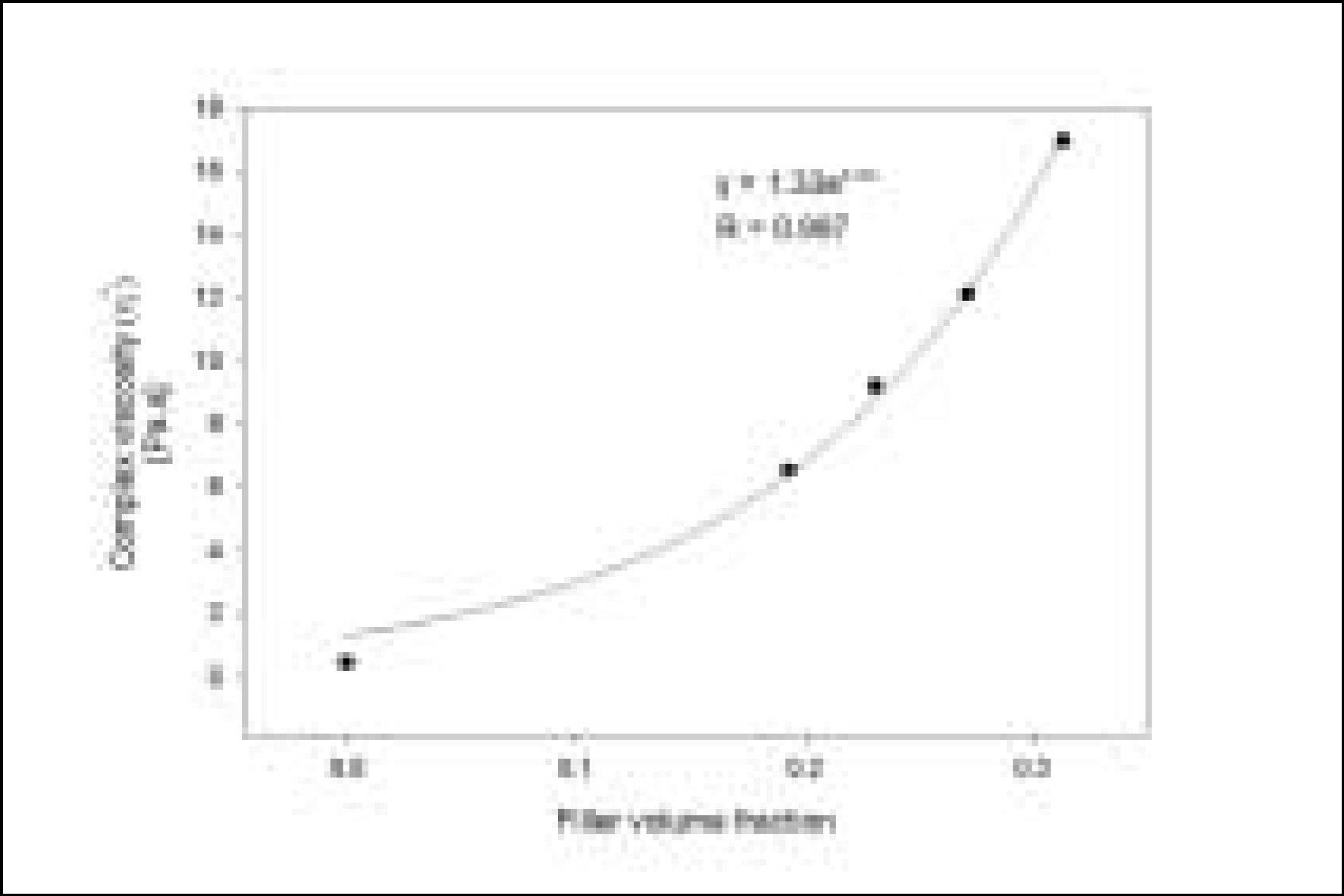
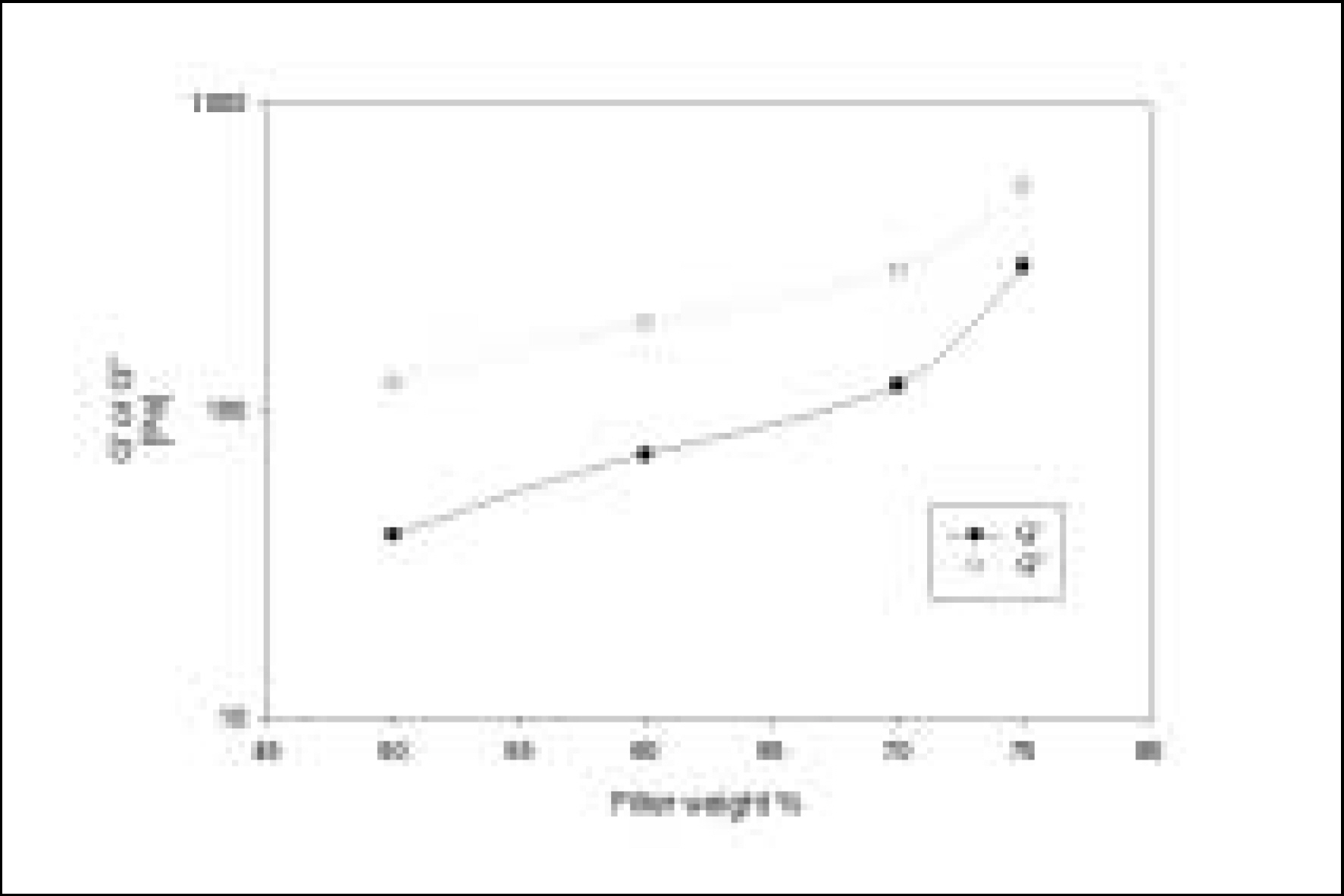
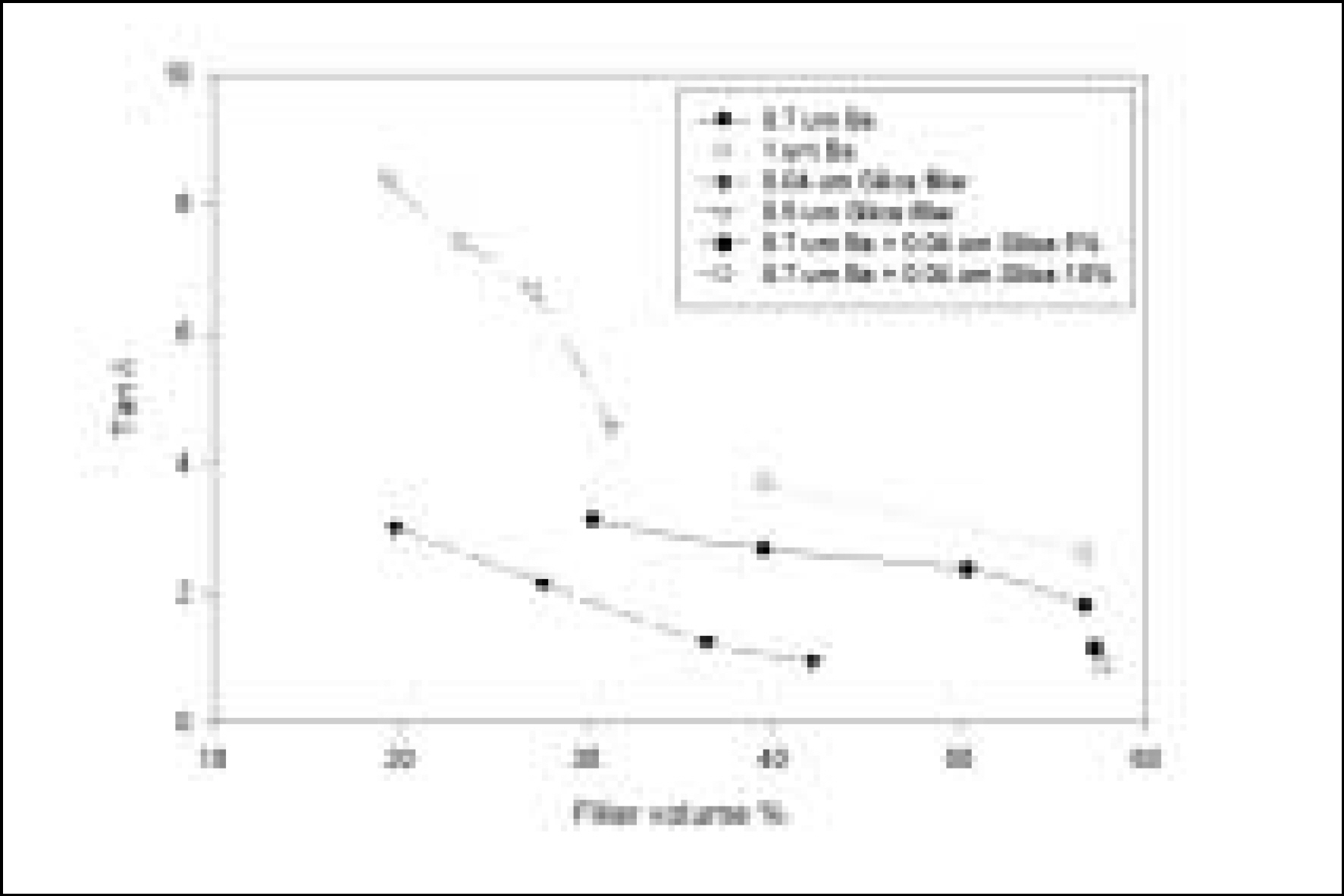
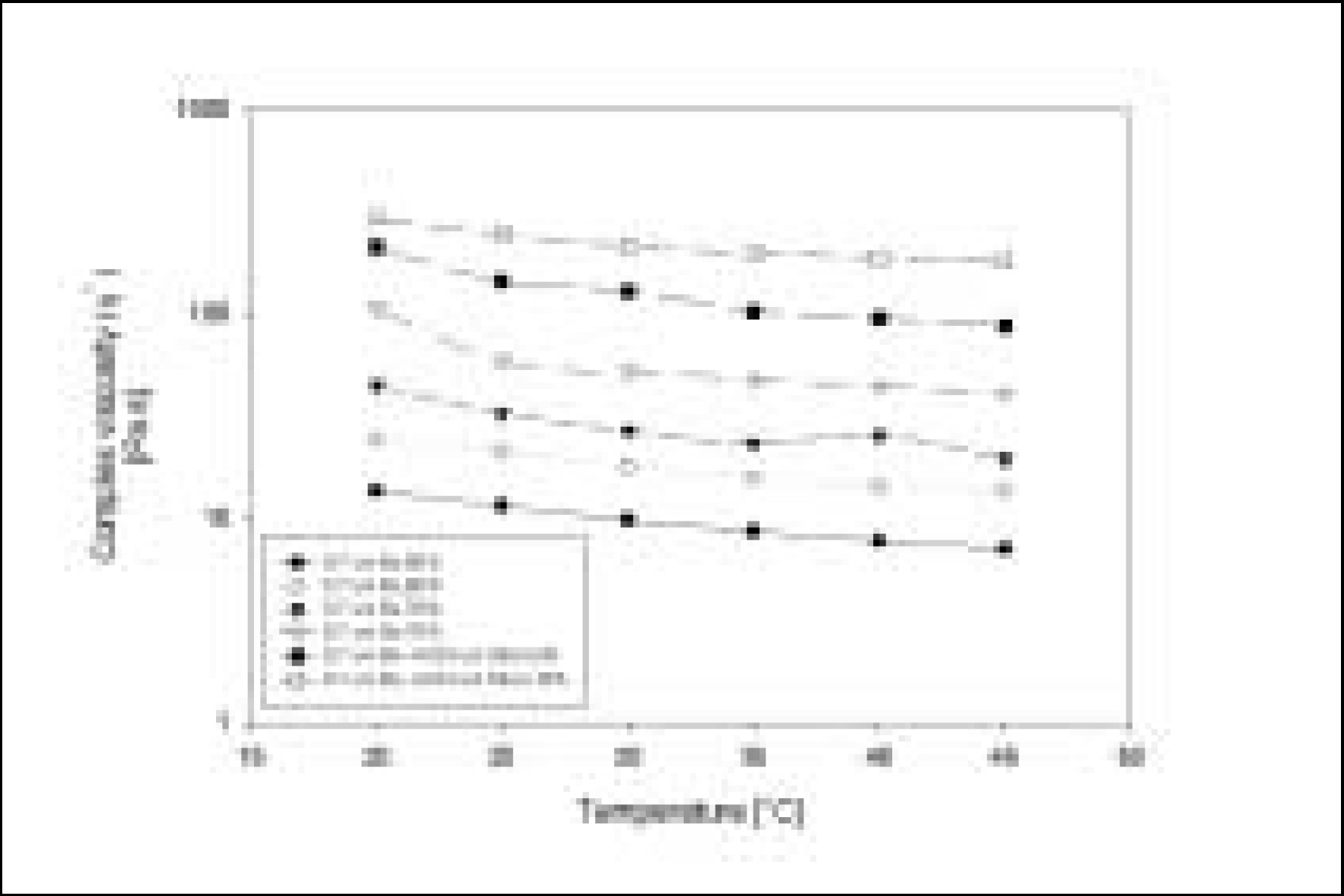
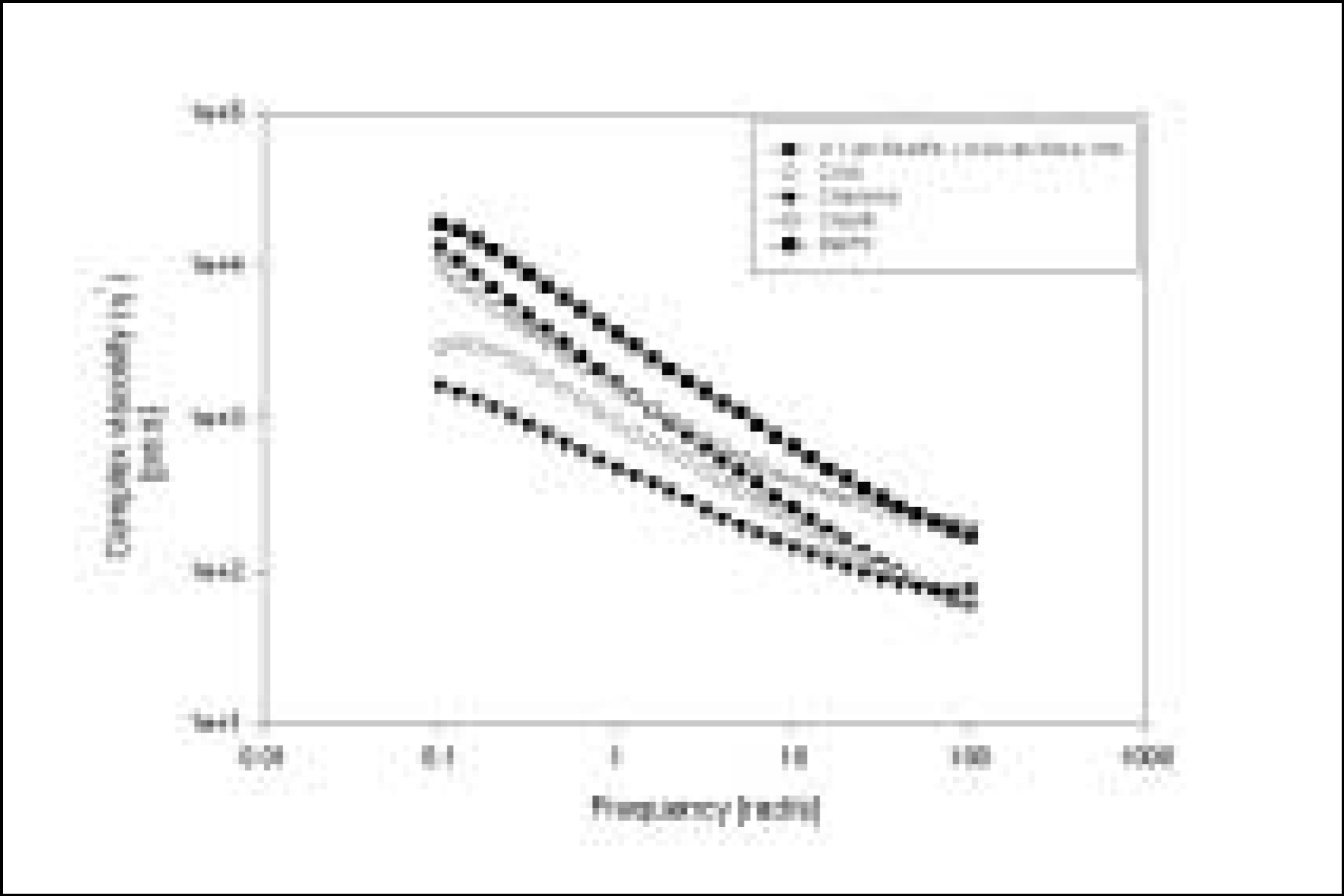
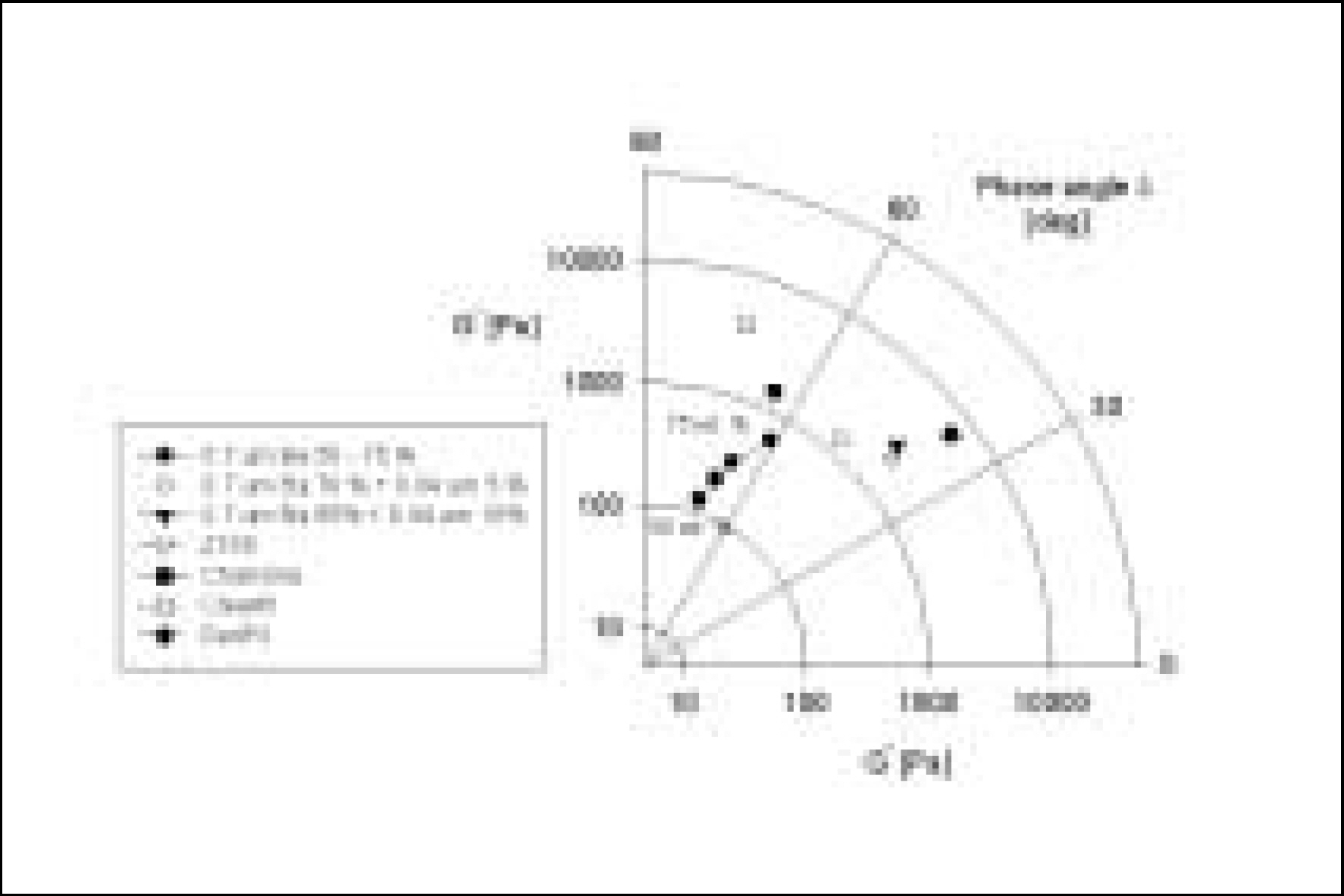
Resin matrices that Bis-GMA as base monomer was blended with TEGDMA as diluent at various ratio were mixed with the Barium glass (0.7 um and 1.0 um), 0.04 um fumed silica and 0.5 um round silica. All used fillers were silane treated. In order to vary the viscosity of experimental composites, the type and content of incorporated fillers were changed. Using a rheometer, a steady shear test and a dynamic oscillatory shear test were used to evaluate the viscosity (eta) of resin matrix, and the storage shear modulus (G'), the loss shear modulus (G"), the loss tangent (tandelta) and the complex viscosity (eta*) of the composites as a function of frequency omega = 0.1-100 rad/s. To investigate the effect of temperature on the viscosity of composites, a temperature sweep test was also undertaken.
RESULTS
Resin matrices were Newtonian fluid regardless of diluent concentration and all experimental composites exhibited pseudoplastic behavior with increasing shear rate. The viscosity of composites was exponentially increased with increasing filler volume%. In the same filler volume, the smaller the fillers were used, the higher the viscosities were. The effect of filler size on the viscosity was increased with increasing filler content. Increasing filler content reduced tandelta by increasing the G' further than the G". The viscosity of composites was decreased exponentially with increasing temperature.
MeSH Terms
Figure
Cited by 2 articles
-
Effect of a new resin monomer on the microleakage of composite resin restorations
JH Bae, YK Kim, PY Yoon, MA Lee, BH Cho
J Korean Acad Conserv Dent. 2007;32(5):469-475. doi: 10.5395/JKACD.2007.32.5.469.Surface roughness of experimental composite resins using confocal laser scanning microscope
JH Bae, MA Lee, BH Cho
J Korean Acad Conserv Dent. 2008;33(1):1-8. doi: 10.5395/JKACD.2008.33.1.001.
Reference
-
References
1. Bayne SC, Thompson JY, Swift EJ. . A characterization of firstgeneration flowable composites. JADA. 129:567–577. 1998.
Article2. Leinfelder KF, Prasad A. A new condensable composite for the restoration of posterior teeth. Dent Today. 17(2):112–6. 1998.3. Leinfelder KF, Radz GM, Nash RW. A report on a new condensable composite resin. Compendium. 19(3):230–237. 1998.4. Opdam NJM, Roeters JJM, Peters TCRB. . Consistency of resin composites for posterior use. Dent Mater. 12:350–354. 1996.
Article5. Jacobsen PH, Whiting R, Richardson PCA. Viscosity of setting anterior restorative materials. Brit dent J. 143:393. 1977.
Article6. Ferracane JL, Moser JB, Greener EH. Rheology of composite restoratives. J Dent Res. 60(9):1678–85. 1981.
Article7. Lee IB, Son HH, Um CM. Rheological Properties of flowable, conventional hybrid, and condensable composite resins. Dent Mater. 19:298–307. 2003.8. Tyas MJ, Jones DW, Rizkalla AS. The evaluation of resin composite consistency. Dent Mater. 14:424–428. 1998.
Article9. Al-sharaa KA, Watts DC. Stickiness prior to setting of some light cured resin composites. Dent Mater. 19:182–187. 2003.10. Willems G, Lambrechts P, Braaem M. . A classification of dental composites according to their morphological and mechanical characteristics. Dent Mater. 8:310–319. 1992.
Article11. Craig RG. Restorative dental materials. 1993. Saunders co.;10th edi. p. 77–83.12. Duran RL, Powers JM, Craig RG. Viscoelastic and dynamic properties of soft liners and tissue conditioners. J Dent Res. 58(8):1801–1807. 1979.
Article13. Goldberg AJ. Viscoelastic properties of silicon, polysul-fide, and polyether impression materials. J Dent Res. 53(5):1033–39. 1974.14. Watt DC. Elastic moduli and visco-elastic relaxation. J Dent. 22:154–158. 1994.15. McCabe JF, Bowman AJ. The rheological properties of dental impression materials. Brit dent J. 151:179. 1981.
Article16. Braden M. Viscosity and consistency of impression rubbers. J Dent Res. 46(2):429–433. 1967.
Article17. Barnes HA, Hutton JF, Walters K. An introduction to rheology. 1989. Elsevier Science Publishing co.18. 문탁 진. 유변학. 1988. 민음사. 1st edi. p. 7–80. p. 190–195.19. 김상 용. 고분자물리학. 1994. 서울대학교출판부. 2nd edi. p. 7–26.20. 한국유변학회. 유변학의 이론과 응용. 2001. 1st edi. p. 3–103. p. 175–191.21. Taylor DF, Kalachandra SK, Sankarapandian M, McGrath JE. Relationship between filler and matrix resin characteristics and the properties of uncured composite pastes. Biomaterials. 19:197–204. 1998.
Article22. Silikas N, Watts DC. Rheology of urethane dimethacrylate and diluent formulations. Dent Mater. 15:257–261. 1999.
Article23. Tavas MA, Watts DC. The use of a cone and plate viscometer for determination of flow properties of unfilled resins and etching gels. J of Oral Rehabil. 16:185–192. 1989.
Article24. Kim SW, Kim OY, Jang JS. Rheological properties of fumed silica filled Bis-GMA dispersions. Polymer Eng and Sci. 38:1142–1148. 1998.
Article25. Schulze KA, Zaman AA, Soderholm KJ. Effect of filler fraction on strength, viscosity and porosity of experimental compomer materials. J of Dent. 31:373–382. 2003.
Article26. Musanje L, Ferracane JL. Effects of resin formulation and nanofiller surface treatment on the properties of experimental hybrid resin composite. Biomaterials. 25:4065–4071. 2004.
Article27. Ikejima I, Nomoto R. McCabe. Shear punch strength and flexural strength of model composies with varying filler volume fraction, particle size and silanation. Dent Mater. 19:206–211. 2003.28. Dulik D, Bernier R, Brauer GM. Effect of diluent monomer on the physical properties of Bis-GMA based composites. J Dent Res. 60:983–989. 1981.29. Asmussen E, Peutzfeldt A. Influence of UEDMA, BisGMA and TEGDMA on selected mechanical properties of experimental resin composites. Dent Mater. 14:51–56. 1998.
Article30. Braem M, Finger W, Doren VE, Lambrechts P, Vanherle G. Mechanical properties and filler fraction of dental composites. Dent Mater. 5:346–349. 1989.
Article31. Soderholm KJM. Influence of silane treatment and filler fraction on thermal expansion of composite resins. Dent Res. 63:1321–1326. 1984.
Article32. Lim BS, Ferracane JL, Condon JR, Adey JD. Effect of filler fraction and filler surface treatment on wear of microfilled composites. Dent Mater. 18:1–11. 2002.
Article33. Mohsen NM, Craig RG. Effect of silanation of fillers on their dispersability by monomer systems. J of Oral Rehab. 22:183–189. 1995.
Article34. Kwon TM, Jhon MS, Choi HJ. Viscosity of magnetic particle suspension. J mole liquid. 75:115–126. 1998.
Article35. Halvorson RH, Erickson RL, Davidson CL. The effect of filler and silane content on conversion of resin based composite. Dent Mater. 19:327–333. 2003.36. Beatty MW, Swartz ML, Moore BK, Phillips RW, Roberts TA. Effect of microfiller fraction and silane treatment on resin composite properties. J Biomed Mater Res. 40:12–23. 1998.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Overview of Filler: Compositions, Effects, Rheological Consideration
- Effect of hema and tegdma on the properties of experimental composite resins
- Effect of resin matrix on degree of conversion and fracture toughness of dental composites
- Self-adhesion of low-viscosity composites to dentin surface
- Slumping tendency and rheological property of flowable composites