J Korean Med Sci.
2012 Jan;27(1):96-100. 10.3346/jkms.2012.27.1.96.
Early-onset Childhood Sarcoidosis with Incidental Multiple Enchondromatosis
- Affiliations
-
- 1Department of Pediatrics, Wonkwang University Sanbon Medical Center, Gunpo, Korea.
- 2Department of Pediatrics, Chungnam University Hospital, Daejeon, Korea.
- 3Department of Radiology, Hanyang University Medical Center, Seoul, Korea.
- 4Department of Nuclear Medicine, Hanyang University Medical Center, Seoul, Korea.
- 5Department of Pathology, Hanyang University Medical Center, Seoul, Korea.
- 6Department of Pediatrics, Hanyang University Medical Center, Seoul, Korea. cord@hanyang.ac.kr
- KMID: 1792987
- DOI: http://doi.org/10.3346/jkms.2012.27.1.96
Abstract
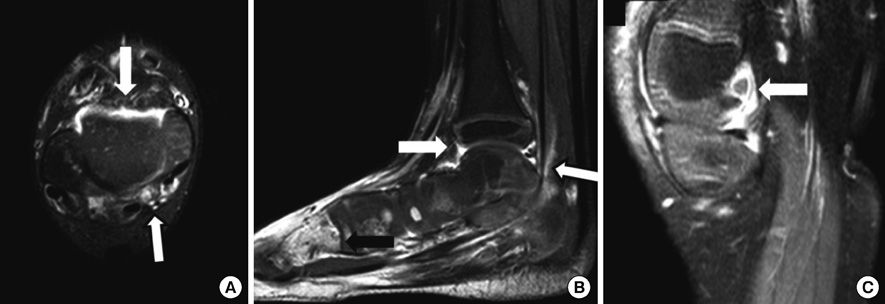
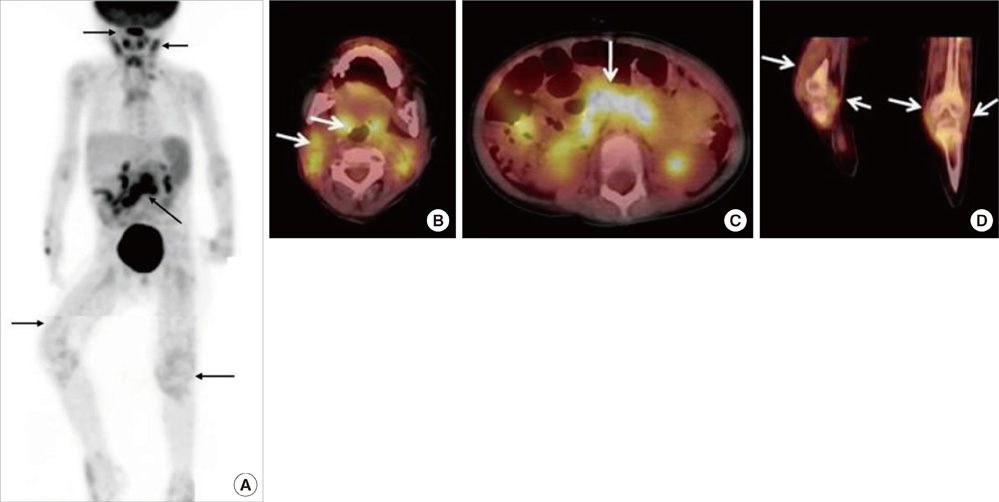
- The triad of rash, arthritis, and uveitis seems to be characteristic for early-onset childhood sarcoidosis. We describe an interesting case of early-onset childhood sarcoidosis coexisting enchondromatosis, which clinically masquerade as Langerhans cell histiocytosis. A 33 months old girl presented with skin rash, subcutaneous nodules with polyarthritis, and revealed the involvement of lymph nodes as well as spleen during work-up. She also presented with multiple osteolytic lesions which pathologically proven enchondromatosis. Oral prednisone was prescribed at 2 mg/kg/day for 2 months until when subcutaneous nodules and joint swellings almost disappeared, and then slowly tapered over a period of 5 months. We report an unusual case of early-onset childhood sarcoidosis presented with osteolytic bone lesions which were irrelevant to sarcoidosis.
Keyword
MeSH Terms
-
Administration, Oral
Anti-Inflammatory Agents/therapeutic use
Arthritis/complications
Child, Preschool
Diagnosis, Differential
Enchondromatosis/*complications/*diagnosis/drug therapy/radiography
Exanthema/etiology
Female
Humans
Positron-Emission Tomography and Computed Tomography
Prednisone/therapeutic use
Sarcoidosis/*complications/*diagnosis/drug therapy/radiography
Whole Body Imaging
Figure
Cited by 1 articles
-
Pediatric Sarcoidosis Misdiagnosed as Hepatosplenic Abscesses: A Case Report and Review
Su Min Lee, Hyungwook Choi, Sungmin Lim, Jehee Shin, Ji-Man Kang, Jong Gyun Ahn
J Rheum Dis. 2022;29(3):181-186. doi: 10.4078/jrd.2022.29.3.181.
Reference
-
1. Hunninghake GW, Costabel U, Ando M, Baughman R, Cordier JF, du Bois R, Eklund A, Kitaichi M, Lynch J, Rizzato G, Rose C, Selroos O, Semenzato G, Sharma OP. ATS/ERS/WASOG statement of sarcoidosis. American Thoracic Society/European Respiratory Society/World Association of Sarcoidosis and other Granulomatous Disorders. Sarcoidosis Vasc Diffuse Lung Dis. 1999. 16:149–173.2. Hetherington S. Sarcoidosis in young children. Am J Dis Child. 1982. 136:13–15.3. Häfner R, Vogel P. Sarcoidosis of early onset. A challenge for the pediatric rheumatologist. Clin Exp Rheumatol. 1993. 11:685–691.4. Fink CW, Cimaz R. Early onset sarcoidosis: not a benign disease. J Rheumatol. 1997. 24:174–177.5. Pattishall EN, Kendig EL Jr. Sarcoidosis in children. Pediatr Pulmonol. 1996. 22:195–203.6. Mallory SB, Paller AS, Ginsburg BC, McCrossin ID, Abernathy R. Sarcoidosis in children: differentiation from juvenile rheumatoid arthritis. Pediatr Dermatol. 1987. 4:313–319.7. Rosenberg AM, Yee EH, MacKenzie JW. Arthritis in childhood sarcoidosis. J Rheumatol. 1983. 10:987–990.8. Hoffmann AL, Milman N, Byg KE. Childhood sarcoidosis in Denmark 1979-1994: incidence, clinical features and laboratory results at presentation in 48 children. Acta Paediatr. 2004. 93:30–36.9. Baculard A, Blanc N, Boulé M, Fauroux B, Chadelat K, Boccon-Gibod L, Tournier G, Clement A. Pulmonary sarcoidosis in children: a follow-up study. Eur Respir J. 2001. 17:628–635.10. Fauroux B, Clèment A. Paediatric sarcoidosis. Paediatr Respir Rev. 2005. 6:128–133.11. Kim DS. Sarcoidosis in Korea: report of the Second Nationwide Survey. Sarcoidosis Vasc Diffuse Lung Dis. 2001. 18:176–180.12. Aouba A, Larousserie F, Le Guern V, Martin A, Guillevin L. Spumous histiocytic oligoarthritis coexisting with systemic Langerhans' cell histiocytosis: case report and literature review. Joint Bone Spine. 2009. 76:701–704.13. Wilcox A, Bharadwaj P, Sharma OP. Bone sarcoidosis. Curr Opin Rheumatol. 2000. 12:321–330.14. Nishiyama Y, Yamamoto Y, Fukunaga K, Takinami H, Iwado Y, Satoh K, Ohkawa M. Comparative evaluation of 18F-FDG PET and 67Ga scintigraphy in patients with sarcoidosis. J Nucl Med. 2006. 47:1571–1576.15. Teirstein AS, Machac J, Almeida O, Lu P, Padilla ML, Iannuzzi MC. Results of 188 whole-body fluorodeoxyglucose positron emission tomography scans in 137 patients with sarcoidosis. Chest. 2007. 132:1949–1953.16. Zhuang H, Alavi A. 18-fluorodeoxyglucose positron emission tomographic imaging in the detection and monitoring of infection and inflammation. Semin Nucl Med. 2002. 32:47–59.