Korean J Physiol Pharmacol.
2022 May;26(3):207-218. 10.4196/kjpp.2022.26.3.207.
The effect of fibroblast growth factor receptor inhibition on resistance exercise training-induced adaptation of bone and muscle quality in mice
- Affiliations
-
- 1Department of Physiology, Seoul National University College of Medicine, Seoul 03080, Korea
- 2Department of Sport and Exercise Science, Seoul Women's University, Seoul 01797, Korea
- 3Department of Nuclear Medicine, Seoul National University Bundang Hospital, Seongnam 13620, Korea
- 4Ischemic/Hypoxic Disease Institute, Seoul National University College of Medicine, Seoul 03080, Korea
- 5Health and Exercise Science Laboratory, Institute of Sport Science, Seoul National University, Seoul 08826, Korea
- 6Department of Physical Education, Seoul National University, Seoul 08826, Korea
- 7Institute on Aging, Seoul National University, Seoul 08826, Korea
- KMID: 2529403
- DOI: http://doi.org/10.4196/kjpp.2022.26.3.207
Abstract
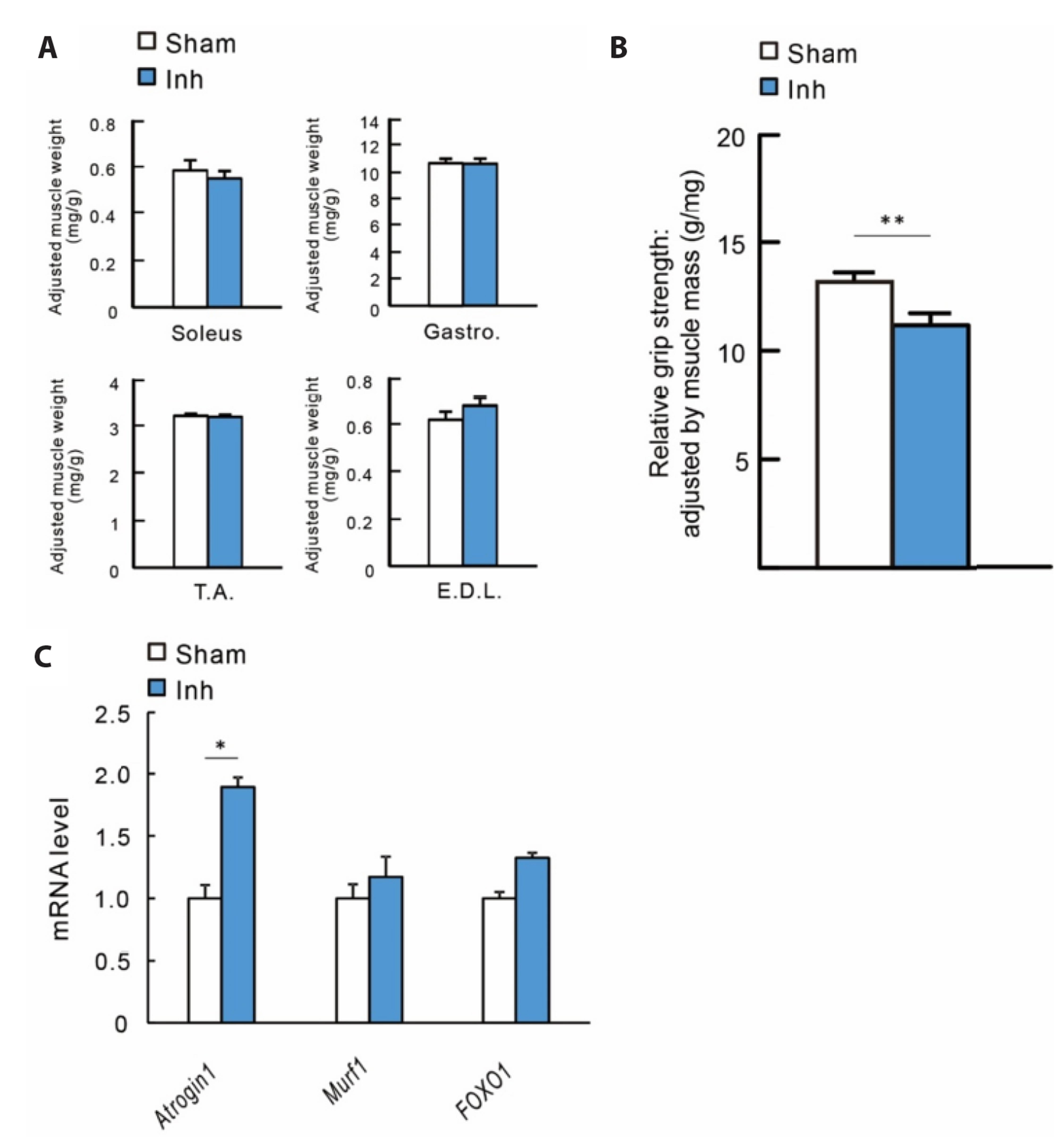
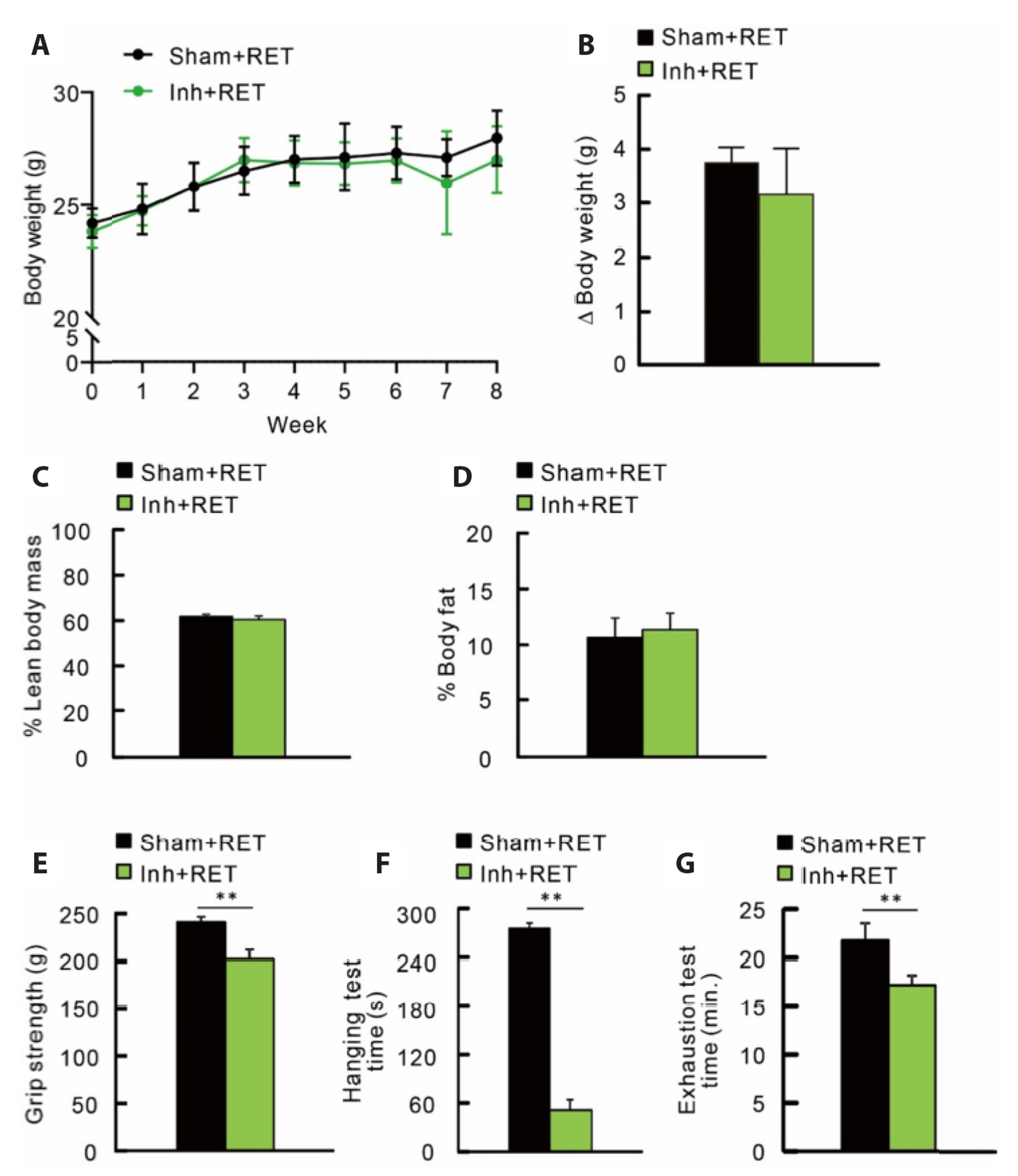
- Aging in mammals, including humans, is accompanied by loss of bone and muscular function and mass, characterized by osteoporosis and sarcopenia. Although resistance exercise training (RET) is considered an effective intervention, its effect is blunted in some elderly individuals. Fibroblast growth factor (FGF) and its receptor, FGFR, can modulate bone and muscle quality during aging and physical performance. To elucidate this possibility, the FGFR inhibitor NVP-BGJ398 was administrated to C57BL/6n mice for 8 weeks with or without RET. Treatment with NVPBGJ398 decreased grip strength, muscular endurance, running capacity and bone quality in the mice. FGFR inhibition elevated bone resorption and relevant gene expression, indicating altered bone formation and resorption. RET attenuated tibial bone resorption, accompanied by changes in the expression of relevant genes. However, RET did not overcome the detrimental effect of NVP-BGJ398 on muscular function. Taken together, these findings provide evidence that FGFR signaling may have a potential role in the maintenance of physical performance and quality of bone and muscles.
Figure
Reference
-
1. Albrand G, Munoz F, Sornay-Rendu E, DuBoeuf F, Delmas PD. 2003; Independent predictors of all osteoporosis-related fractures in healthy postmenopausal women: the OFELY study. Bone. 32:78–85. DOI: 10.1016/S8756-3282(02)00919-5. PMID: 12584039.
Article2. Stenholm S, Harris TB, Rantanen T, Visser M, Kritchevsky SB, Ferrucci L. 2008; Sarcopenic obesity: definition, cause and consequences. Curr Opin Clin Nutr Metab Care. 11:693–700. DOI: 10.1097/MCO.0b013e328312c37d. PMID: 18827572. PMCID: PMC2633408.
Article3. Hairi NN, Cumming RG, Naganathan V, Handelsman DJ, Le Couteur DG, Creasey H, Waite LM, Seibel MJ, Sambrook PN. 2010; Loss of muscle strength, mass (sarcopenia), and quality (specific force) and its relationship with functional limitation and physical disability: the Concord Health and Ageing in Men Project. J Am Geriatr Soc. 58:2055–2062. DOI: 10.1111/j.1532-5415.2010.03145.x. PMID: 21054284.
Article4. van den Berg LE, Zandbergen AA, van Capelle CI, de Vries JM, Hop WC, van den Hout JM, Reuser AJ, Zillikens MC, van der Ploeg AT. 2010; Low bone mass in Pompe disease: muscular strength as a predictor of bone mineral density. Bone. 47:643–649. DOI: 10.1016/j.bone.2010.06.021. PMID: 20601298.5. Hirschfeld HP, Kinsella R, Duque G. 2017; Osteosarcopenia: where bone, muscle, and fat collide. Osteoporos Int. 28:2781–2790. DOI: 10.1007/s00198-017-4151-8. PMID: 28733716.
Article6. Kanis J. On behalf of the World Health Organization Scientific Group. 2018. Assessment of osteoporosis at the primary health-care level. Technical report. World Health Organization Collaborating Centre for Metabolic Bone Diseases, University of Sheffield;Sheffield: DOI: 10.1007/s00198-017-4151-8.7. Janssen I, Heymsfield SB, Ross R. 2002; Low relative skeletal muscle mass (sarcopenia) in older persons is associated with functional impairment and physical disability. J Am Geriatr Soc. 50:889–896. DOI: 10.1046/j.1532-5415.2002.50216.x. PMID: 12028177.
Article8. Chung HY, Cesari M, Anton S, Marzetti E, Giovannini S, Seo AY, Carter C, Yu BP, Leeuwenburgh C. 2009; Molecular inflammation: underpinnings of aging and age-related diseases. Ageing Res Rev. 8:18–30. DOI: 10.1016/j.arr.2008.07.002. PMID: 18692159. PMCID: PMC3782993.
Article9. Burr DB. 1997; Muscle strength, bone mass, and age-related bone loss. J Bone Miner Res. 12:1547–1551. DOI: 10.1359/jbmr.1997.12.10.1547. PMID: 9333114.
Article10. Isaacson J, Brotto M. 2014; Physiology of mechanotransduction: how do muscle and bone "talk" to one another? Clin Rev Bone Miner Metab. 12:77–85. DOI: 10.1007/s12018-013-9152-3. PMID: 25838800. PMCID: PMC4380142.
Article11. Kawao N, Kaji H. 2015; Interactions between muscle tissues and bone metabolism. J Cell Biochem. 116:687–695. DOI: 10.1002/jcb.25040. PMID: 25521430.
Article12. Beck BR, Daly RM, Singh MA, Taaffe DR. 2017; Exercise and Sports Science Australia (ESSA) position statement on exercise prescription for the prevention and management of osteoporosis. J Sci Med Sport. 20:438–445. DOI: 10.1016/j.jsams.2016.10.001. PMID: 27840033.
Article13. Greig CA, Gray C, Rankin D, Young A, Mann V, Noble B, Atherton PJ. 2011; Blunting of adaptive responses to resistance exercise training in women over 75y. Exp Gerontol. 46:884–890. DOI: 10.1016/j.exger.2011.07.010. PMID: 21821111.
Article14. Karinkanta S, Heinonen A, Sievänen H, Uusi-Rasi K, Pasanen M, Ojala K, Fogelholm M, Kannus P. 2007; A multi-component exercise regimen to prevent functional decline and bone fragility in home-dwelling elderly women: randomized, controlled trial. Osteoporos Int. 18:453–462. DOI: 10.1007/s00198-006-0256-1. PMID: 17103296.
Article15. Javaheri B, Pitsillides AA. 2019; Aging and mechanoadaptive responsiveness of bone. Curr Osteoporos Rep. 17:560–569. Erratum in: Curr Osteoporos Rep. 2020. doi: 10.1007/s11914-019-00553-7. DOI: 10.1007/s11914-019-00553-7. PMID: 31760583. PMCID: PMC6944648.
Article16. Spencer EM, Liu CC, Si EC, Howard GA. 1991; In vivo actions of insulin-like growth factor-I (IGF-I) on bone formation and resorption in rats. Bone. 12:21–26. DOI: 10.1016/8756-3282(91)90050-S. PMID: 2054232.
Article17. Marie PJ. 2012; Fibroblast growth factor signaling controlling bone formation: an update. Gene. 498:1–4. DOI: 10.1016/j.gene.2012.01.086. PMID: 22342254.
Article18. Fukumoto S, Martin TJ. 2009; Bone as an endocrine organ. Trends Endocrinol Metab. 20:230–236. DOI: 10.1016/j.tem.2009.02.001. PMID: 19546009.
Article19. Mera P, Laue K, Ferron M, Confavreux C, Wei J, Galán-Díez M, Lacampagne A, Mitchell SJ, Mattison JA, Chen Y, Bacchetta J, Szulc P, Kitsis RN, de Cabo R, Friedman RA, Torsitano C, McGraw TE, Puchowicz M, Kurland I, Karsenty G. 2016; Osteocalcin signaling in myofibers is necessary and sufficient for optimum adaptation to exercise. Cell Metab. 23:1078–1092. Erratum in: Cell Metab. 2017;25:218. DOI: 10.1016/j.cmet.2016.05.004. PMID: 27304508. PMCID: PMC4910629.
Article20. Cowan CM, Quarto N, Warren SM, Salim A, Longaker MT. 2003; Age-related changes in the biomolecular mechanisms of calvarial osteoblast biology affect fibroblast growth factor-2 signaling and osteogenesis. J Biol Chem. 278:32005–32013. Erratum in: J Biol Chem. 2003;278:45040. DOI: 10.1016/S0021-9258(20)82534-4. PMID: 12788918.
Article21. Ou G, Charles L, Matton S, Rodner C, Hurley M, Kuhn L, Gronowicz G. 2010; Fibroblast growth factor-2 stimulates the proliferation of mesenchyme-derived progenitor cells from aging mouse and human bone. J Gerontol A Biol Sci Med Sci. 65:1051–1059. DOI: 10.1093/gerona/glq114. PMID: 20643704. PMCID: PMC2949335.
Article22. Komi-Kuramochi A, Kawano M, Oda Y, Asada M, Suzuki M, Oki J, Imamura T. 2005; Expression of fibroblast growth factors and their receptors during full-thickness skin wound healing in young and aged mice. J Endocrinol. 186:273–289. DOI: 10.1677/joe.1.06055. PMID: 16079254.
Article23. Shetty AK, Hattiangady B, Shetty GA. 2005; Stem/progenitor cell proliferation factors FGF-2, IGF-1, and VEGF exhibit early decline during the course of aging in the hippocampus: role of astrocytes. Glia. 51:173–186. DOI: 10.1002/glia.20187. PMID: 15800930.
Article24. Shimada T, Kakitani M, Yamazaki Y, Hasegawa H, Takeuchi Y, Fujita T, Fukumoto S, Tomizuka K, Yamashita T. 2004; Targeted ablation of Fgf23 demonstrates an essential physiological role of FGF23 in phosphate and vitamin D metabolism. J Clin Invest. 113:561–568. DOI: 10.1172/JCI200419081. PMCID: PMC338262. PMID: 14966565.
Article25. Zhang Y, Xie Y, Berglund ED, Coate KC, He TT, Katafuchi T, Xiao G, Potthoff MJ, Wei W, Wan Y, Yu RT, Evans RM, Kliewer SA, Mangelsdorf DJ. 2012; The starvation hormone, fibroblast growth factor-21, extends lifespan in mice. Elife. 1:e00065. DOI: 10.7554/eLife.00065. PMID: 23066506. PMCID: PMC3466591. PMID: 4b4ce4845cdd40c5b506669b2962b9ef.
Article26. Wöhrle S, Henninger C, Bonny O, Thuery A, Beluch N, Hynes NE, Guagnano V, Sellers WR, Hofmann F, Kneissel M, Graus Porta D. 2013; Pharmacological inhibition of fibroblast growth factor (FGF) receptor signaling ameliorates FGF23-mediated hypophosphatemic rickets. J Bone Miner Res. 28:899–911. DOI: 10.1002/jbmr.1810. PMID: 23129509.
Article27. Duncan ND, Williams DA, Lynch GS. 1998; Adaptations in rat skeletal muscle following long-term resistance exercise training. Eur J Appl Physiol Occup Physiol. 77:372–378. DOI: 10.1007/s004210050347. PMID: 9562367.
Article28. Meyer OA, Tilson HA, Byrd WC, Riley MT. 1979; A method for the routine assessment of fore- and hindlimb grip strength of rats and mice. Neurobehav Toxicol. 1:233–236. PMID: 551317.29. Halldorsdottir S, Carmody J, Boozer CN, Leduc CA, Leibel RL. 2009; Reproducibility and accuracy of body composition assessments in mice by dual energy x-ray absorptiometry and time domain nuclear magnetic resonance. Int J Body Compos Res. 7:147–154. PMID: 21909234. PMCID: PMC3169293.30. Bevier WC, Wiswell RA, Pyka G, Kozak KC, Newhall KM, Marcus R. 1989; Relationship of body composition, muscle strength, and aerobic capacity to bone mineral density in older men and women. J Bone Miner Res. 4:421–432. DOI: 10.1002/jbmr.5650040318. PMID: 2763878.
Article31. Li X, Mohan S, Gu W, Wergedal J, Baylink DJ. 2001; Quantitative assessment of forearm muscle size, forelimb grip strength, forearm bone mineral density, and forearm bone size in determining humerus breaking strength in 10 inbred strains of mice. Calcif Tissue Int. 68:365–369. DOI: 10.1007/s00223-001-0004-7. PMID: 11685425.
Article32. Baek K, Hwang HR, Park HJ, Kwon A, Qadir AS, Ko SH, Woo KM, Ryoo HM, Kim GS, Baek JH. 2014; TNF-α upregulates sclerostin expression in obese mice fed a high-fat diet. J Cell Physiol. 229:640–650. DOI: 10.1002/jcp.24487. PMID: 24446199.
Article33. Gomes MD, Lecker SH, Jagoe RT, Navon A, Goldberg AL. 2001; Atrogin-1, a muscle-specific F-box protein highly expressed during muscle atrophy. Proc Natl Acad Sci U S A. 98:14440–14445. DOI: 10.1073/pnas.251541198. PMID: 11717410. PMCID: PMC64700.
Article34. Sandri M, Sandri C, Gilbert A, Skurk C, Calabria E, Picard A, Walsh K, Schiaffino S, Lecker SH, Goldberg AL. 2004; Foxo transcription factors induce the atrophy-related ubiquitin ligase atrogin-1 and cause skeletal muscle atrophy. Cell. 117:399–412. DOI: 10.1016/S0092-8674(04)00400-3. PMID: 15109499. PMCID: PMC3619734.
Article35. Clavel S, Coldefy AS, Kurkdjian E, Salles J, Margaritis I, Derijard B. 2006; Atrophy-related ubiquitin ligases, atrogin-1 and MuRF1 are up-regulated in aged rat Tibialis Anterior muscle. Mech Ageing Dev. 127:794–801. DOI: 10.1016/j.mad.2006.07.005. PMID: 16949134.
Article36. Kirby TJ, Lee JD, England JH, Chaillou T, Esser KA, McCarthy JJ. 2015; Blunted hypertrophic response in aged skeletal muscle is associated with decreased ribosome biogenesis. J Appl Physiol (1985). 119:321–327. DOI: 10.1152/japplphysiol.00296.2015. PMID: 26048973. PMCID: PMC4538281.
Article37. Kosek DJ, Kim JS, Petrella JK, Cross JM, Bamman MM. 2006; Efficacy of 3 days/wk resistance training on myofiber hypertrophy and myogenic mechanisms in young vs. older adults. J Appl Physiol (1985). 101:531–544. DOI: 10.1152/japplphysiol.01474.2005. PMID: 16614355.
Article38. Slivka D, Raue U, Hollon C, Minchev K, Trappe S. 2008; Single muscle fiber adaptations to resistance training in old (>80 yr) men: evidence for limited skeletal muscle plasticity. Am J Physiol Regul Integr Comp Physiol. 295:R273–R280. DOI: 10.1152/ajpregu.00093.2008. PMID: 18448613. PMCID: PMC2494807.
Article39. Garn SM, Leonard WR, Hawthorne VM. 1986; Three limitations of the body mass index. Am J Clin Nutr. 44:996–997. DOI: 10.1093/ajcn/44.6.996. PMID: 3788846.
Article40. Romero-Corral A, Somers VK, Sierra-Johnson J, Thomas RJ, Collazo-Clavell ML, Korinek J, Allison TG, Batsis JA, Sert-Kuniyoshi FH, Lopez-Jimenez F. 2008; Accuracy of body mass index in diagnosing obesity in the adult general population. Int J Obes (Lond). 32:959–966. DOI: 10.1038/ijo.2008.11. PMID: 18283284. PMCID: PMC2877506.
Article41. Eswarakumar VP, Lax I, Schlessinger J. 2005; Cellular signaling by fibroblast growth factor receptors. Cytokine Growth Factor Rev. 16:139–149. DOI: 10.1016/j.cytogfr.2005.01.001. PMID: 15863030.
Article42. Yu K, Xu J, Liu Z, Sosic D, Shao J, Olson EN, Towler DA, Ornitz DM. 2003; Conditional inactivation of FGF receptor 2 reveals an essential role for FGF signaling in the regulation of osteoblast function and bone growth. Development. 130:3063–3074. DOI: 10.1242/dev.00491. PMID: 12756187.
Article43. White KE, Cabral JM, Davis SI, Fishburn T, Evans WE, Ichikawa S, Fields J, Yu X, Shaw NJ, McLellan NJ, McKeown C, Fitzpatrick D, Yu K, Ornitz DM, Econs MJ. 2005; Mutations that cause osteoglophonic dysplasia define novel roles for FGFR1 in bone elongation. Am J Hum Genet. 76:361–367. DOI: 10.1086/427956. PMID: 15625620. PMCID: PMC1196382.
Article44. Dono R, Texido G, Dussel R, Ehmke H, Zeller R. 1998; Impaired cerebral cortex development and blood pressure regulation in FGF-2-deficient mice. EMBO J. 17:4213–4225. DOI: 10.1093/emboj/17.15.4213. PMID: 9687490. PMCID: PMC1170755.
Article45. Choi SC, Kim SJ, Choi JH, Park CY, Shim WJ, Lim DS. 2008; Fibroblast growth factor-2 and -4 promote the proliferation of bone marrow mesenchymal stem cells by the activation of the PI3K-Akt and ERK1/2 signaling pathways. Stem Cells Dev. 17:725–736. DOI: 10.1089/scd.2007.0230. PMID: 18788932.
Article46. Patel VS, Judex S, Rubin J, Rubin CT. Bilezikian JP, Martin TJ, Clemens TL, Rosen CJ, editors. 2020. Mechanisms of exercise effects on bone quantity and quality. Principles of bone biology. 4th ed. Academic Press;Cambridge: p. 1759–1784. DOI: 10.1016/B978-0-12-814841-9.00076-2.
Article47. Frost HM. 2003; Bone's mechanostat: a 2003 update. Anat Rec A Discov Mol Cell Evol Biol. 275:1081–1101. DOI: 10.1002/ar.a.10119. PMID: 14613308.
Article48. Klein-Nulend J, Bakker AD, Bacabac RG, Vatsa A, Weinbaum S. 2013; Mechanosensation and transduction in osteocytes. Bone. 54:182–190. DOI: 10.1016/j.bone.2012.10.013. PMID: 23085083.
Article49. Galea GL, Lanyon LE, Price JS. 2017; Sclerostin's role in bone's adaptive response to mechanical loading. Bone. 96:38–44. DOI: 10.1016/j.bone.2016.10.008. PMID: 27742499. PMCID: PMC5340132.
Article50. Kirk B, Feehan J, Lombardi G, Duque G. 2020; Muscle, bone, and fat crosstalk: the biological role of myokines, osteokines, and adipokines. Curr Osteoporos Rep. 18:388–400. DOI: 10.1007/s11914-020-00599-y. PMID: 32529456.
Article51. DiGirolamo DJ, Mukherjee A, Fulzele K, Gan Y, Cao X, Frank SJ, Clemens TL. 2007; Mode of growth hormone action in osteoblasts. J Biol Chem. 282:31666–31674. DOI: 10.1074/jbc.M705219200. PMID: 17698843. PMCID: PMC2667784.
Article52. Steringer JP, Müller HM, Nickel W. 2015; Unconventional secretion of fibroblast growth factor 2--a novel type of protein translocation across membranes? J Mol Biol. 427(6 Pt A):1202–1210. DOI: 10.1016/j.jmb.2014.07.012. PMID: 25051502.
Article53. Fujimura R, Ashizawa N, Watanabe M, Mukai N, Amagai H, Fukubayashi T, Hayashi K, Tokuyama K, Suzuki M. 1997; Effect of resistance exercise training on bone formation and resorption in young male subjects assessed by biomarkers of bone metabolism. J Bone Miner Res. 12:656–662. DOI: 10.1359/jbmr.1997.12.4.656. PMID: 9101378.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Resistance exercise training‑induced skeletal muscle strength provides protective effects on high‑fat‑diet‑induced metabolic stress in mice
- Zerumbone, Sesquiterpene Photochemical from Ginger, Inhibits Angiogenesis
- IGF-I Exerts an Anti-inflammatory Effect on Skeletal Muscle Cells through Down-regulation of TLR4 Signaling
- Myostatin as a Potential Therapeutic Target for Obesity and Insulin Resistance
- Pfeiffer Syndrome Type 2 with Sporadic Fibroblast Growth Factor Receptor 2 Mutation and Coccygeal Anomaly