Cancer Res Treat.
2022 Jan;54(1):140-149. 10.4143/crt.2021.385.
Acquired Resistance Mechanism of EGFR Kinase Domain Duplication to EGFR TKIs in Non–Small Cell Lung Cancer
- Affiliations
-
- 1Cancer Research Institute, Seoul National University, Seoul, Korea
- 2Department of Internal Medicine, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea
- 3Integrated Major in Innovative Medical Science, Seoul National University College of Medicine, Seoul, Korea
- 4Genomic Medicine Institute, Medical Research Center, Seoul National University, Seoul, Korea
- 5Department of Pathology, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea
- KMID: 2524595
- DOI: http://doi.org/10.4143/crt.2021.385
Abstract
- Purpose
Epidermal growth factor receptor kinase domain duplication (EGFR-KDD) is a rare and poorly understood oncogenic mutation in non–small cell lung cancer (NSCLC). We aimed to investigate the acquired resistance mechanism of EGFR-KDD against EGFR-TKIs.
Materials and Methods
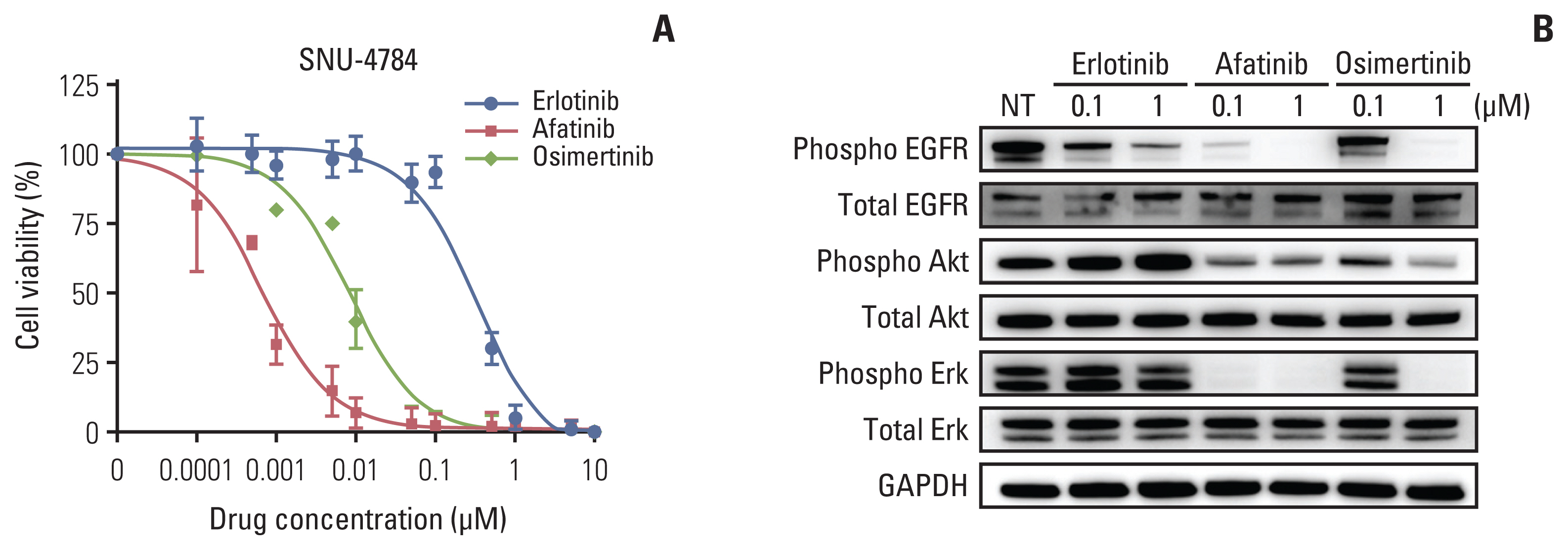
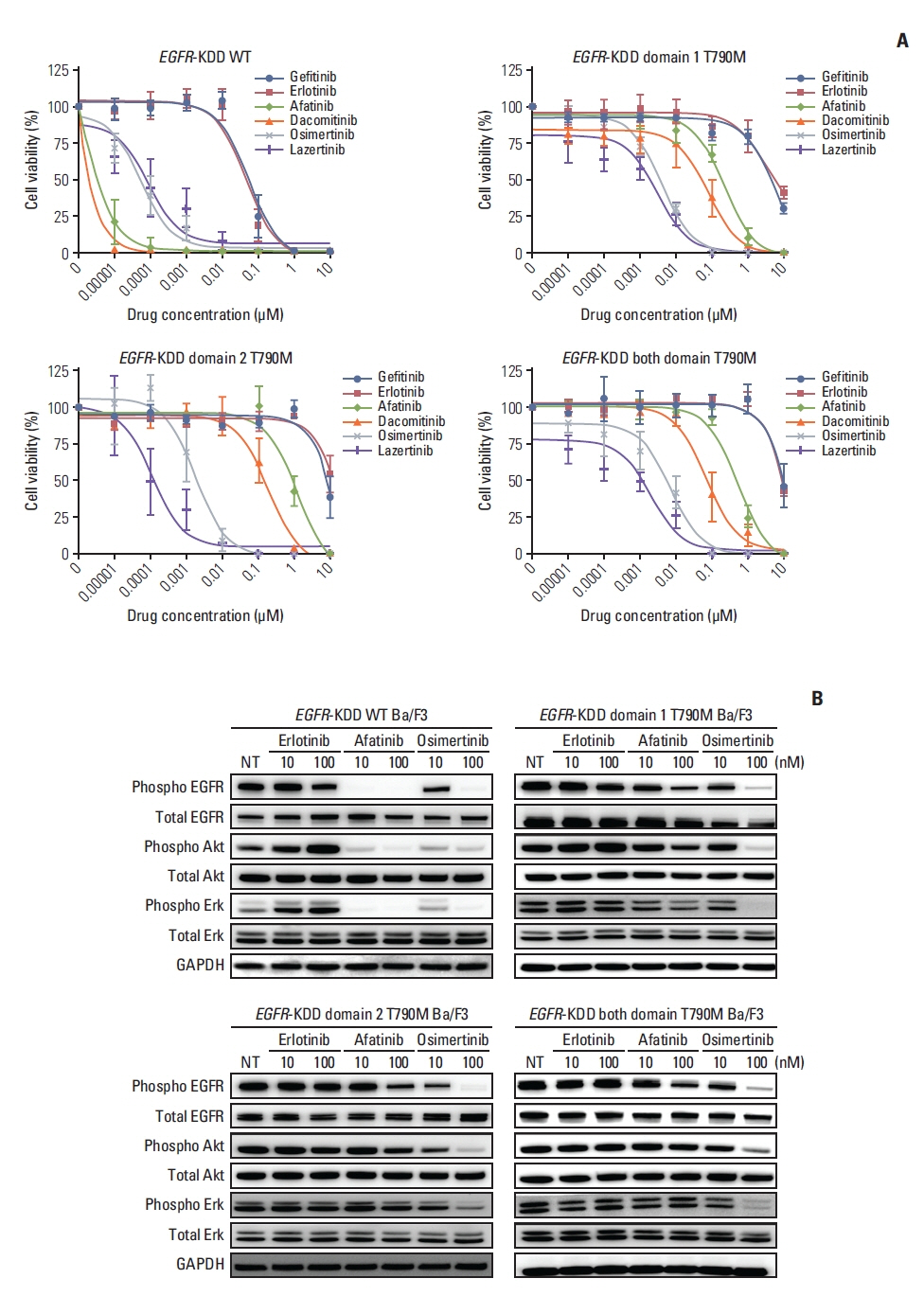
We identified EGFR-KDD in tumor tissue obtained from a patient with stage IV lung adenocarcinoma and established the patient-derived cell line SNU-4784. We also established several EGFR-KDD Ba/F3 cell lines: EGFR-KDD wild type (EGFR-KDDWT), EGFR-KDD domain 1 T790M (EGFR-KDDD1T), EGFR-KDD domain 2 T790M (EGFR-KDDD2T), and EGFR-KDD both domain T790M (EGFR-KDDBDT). We treated the cells with EGFR tyrosine kinase inhibitors (TKIs) and performed cell viability assays, immunoblot assays, and ENU (N-ethyl-N-nitrosourea) mutagenesis screening.
Results
In cell viability assays, SNU-4784 cells and EGFR-KDDWT Ba/F3 cells were sensitive to 2nd generation and 3rd generation EGFR TKIs. In contrast, the T790M-positive EGFR-KDD Ba/F3 cell lines (EGFR-KDDT790M) were only sensitive to 3rd generation EGFR TKIs. In ENU mutagenesis screening, we identified the C797S mutation in kinase domain 2 of EGFR-KDDBDT Ba/F3 cells. Based on this finding, we established an EGFR-KDD domain 1 T790M/domain 2 cis-T790M+C797S (EGFR-KDDT/T+C) Ba/F3 model, which was resistant to EGFR TKIs and anti-EGFR monoclonal antibody combined with EGFR TKIs.
Conclusion
Our study reveals that the T790M mutation in EGFR-KDD confers resistance to 1st and 2nd generation EGFR TKIs, but is sensitive to 3rd generation EGFR TKIs. In addition, we identified that the C797S mutation in kinase domain 2 of EGFR-KDDT790M mediates a resistance mechanism against 3rd generation EGFR TKIs.
Keyword
Figure
Reference
-
References
1. Herbst RS, Morgensztern D, Boshoff C. The biology and management of non-small cell lung cancer. Nature. 2018; 553:446–54.
Article2. Lynch TJ, Bell DW, Sordella R, Gurubhagavatula S, Okimoto RA, Brannigan BW, et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med. 2004; 350:2129–39.
Article3. Paez JG, Janne PA, Lee JC, Tracy S, Greulich H, Gabriel S, et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science. 2004; 304:1497–500.4. Seo JS, Ju YS, Lee WC, Shin JY, Lee JK, Bleazard T, et al. The transcriptional landscape and mutational profile of lung adenocarcinoma. Genome Res. 2012; 22:2109–19.
Article5. Gazdar AF. Activating and resistance mutations of EGFR in non-small-cell lung cancer: role in clinical response to EGFR tyrosine kinase inhibitors. Oncogene. 2009; 28(Suppl 1):S24–31.
Article6. Costa DB. Kinase inhibitor-responsive genotypes in EGFR mutated lung adenocarcinomas: moving past common point mutations or indels into uncommon kinase domain duplications and rearrangements. Transl Lung Cancer Res. 2016; 5:331–7.
Article7. Du Z, Lovly CM. Mechanisms of receptor tyrosine kinase activation in cancer. Mol Cancer. 2018; 17:58.
Article8. Gallant JN, Sheehan JH, Shaver TM, Bailey M, Lipson D, Chandramohan R, et al. EGFR kinase domain duplication (EGFR-KDD) is a novel oncogenic driver in lung cancer that is clinically responsive to afatinib. Cancer Discov. 2015; 5:1155–63.
Article9. Du Z, Brown BP, Kim S, Ferguson D, Pavlick DC, Jayakumaran G, et al. Structure-function analysis of oncogenic EGFR Kinase Domain Duplication reveals insights into activation and a potential approach for therapeutic targeting. Nat Commun. 2021; 12:1382.
Article10. Zhu YC, Wang WX, Xu CW, Tan QH, Li JY, Zhuang W, et al. Lung adenocarcinoma patient with an EGFR kinase domain duplication (KDD) and the response to icotinib. J Thorac Dis. 2018; 10:E359–63.
Article11. Oxnard GR, Arcila ME, Sima CS, Riely GJ, Chmielecki J, Kris MG, et al. Acquired resistance to EGFR tyrosine kinase inhibitors in EGFR-mutant lung cancer: distinct natural history of patients with tumors harboring the T790M mutation. Clin Cancer Res. 2011; 17:1616–22.
Article12. Yu HA, Arcila ME, Rekhtman N, Sima CS, Zakowski MF, Pao W, et al. Analysis of tumor specimens at the time of acquired resistance to EGFR-TKI therapy in 155 patients with EGFR-mutant lung cancers. Clin Cancer Res. 2013; 19:2240–7.
Article13. Sequist LV, Waltman BA, Dias-Santagata D, Digumarthy S, Turke AB, Fidias P, et al. Genotypic and histological evolution of lung cancers acquiring resistance to EGFR inhibitors. Sci Transl Med. 2011; 3:75ra26.
Article14. Im SW, Chae J, Jang SS, Choi J, Yun J, Cha S, et al. A newly developed capture-based sequencing panel for genomic assay of lung cancer. Genes Genomics. 2020; 42:751–9.
Article15. Kim TM, Song A, Kim DW, Kim S, Ahn YO, Keam B, et al. Mechanisms of acquired resistance to AZD9291: a mutation-selective, irreversible EGFR inhibitor. J Thorac Oncol. 2015; 10:1736–44.16. Lee Y, Kim TM, Kim DW, Kim S, Kim M, Keam B, et al. Preclinical modeling of osimertinib for NSCLC with EGFR exon 20 insertion mutations. J Thorac Oncol. 2019; 14:1556–66.
Article17. Normanno N, De Luca A, Bianco C, Strizzi L, Mancino M, Maiello MR, et al. Epidermal growth factor receptor (EGFR) signaling in cancer. Gene. 2006; 366:2–16.
Article18. Ahn MJ, Han JY, Lee KH, Kim SW, Kim DW, Lee YG, et al. Lazertinib in patients with EGFR mutation-positive advanced non-small-cell lung cancer: results from the dose escalation and dose expansion parts of a first-in-human, open-label, multicentre, phase 1–2 study. Lancet Oncol. 2019; 20:1681–90.
Article19. Jia Y, Yun CH, Park E, Ercan D, Manuia M, Juarez J, et al. Overcoming EGFR(T790M) and EGFR(C797S) resistance with mutant-selective allosteric inhibitors. Nature. 2016; 534:129–32.
Article20. Uchibori K, Inase N, Araki M, Kamada M, Sato S, Okuno Y, et al. Brigatinib combined with anti-EGFR antibody overcomes osimertinib resistance in EGFR-mutated non-small-cell lung cancer. Nat Commun. 2017; 8:14768.
Article21. Ozer BH, Wiepz GJ, Bertics PJ. Activity and cellular localization of an oncogenic glioblastoma multiforme-associated EGF receptor mutant possessing a duplicated kinase domain. Oncogene. 2010; 29:855–64.
Article22. Baik CS, Wu D, Smith C, Martins RG, Pritchard CC. Durable response to tyrosine kinase inhibitor therapy in a lung cancer patient harboring epidermal growth factor receptor tandem kinase domain duplication. J Thorac Oncol. 2015; 10:e97–9.
Article23. Garassino MC, Martelli O, Broggini M, Farina G, Veronese S, Rulli E, et al. Erlotinib versus docetaxel as second-line treatment of patients with advanced non-small-cell lung cancer and wild-type EGFR tumours (TAILOR): a randomised controlled trial. Lancet Oncol. 2013; 14:981–8.
Article24. Kawaguchi T, Ando M, Asami K, Okano Y, Fukuda M, Nakagawa H, et al. Randomized phase III trial of erlotinib versus docetaxel as second- or third-line therapy in patients with advanced non-small-cell lung cancer: Docetaxel and Erlotinib Lung Cancer Trial (DELTA). J Clin Oncol. 2014; 32:1902–8.
Article25. Wang J, Li X, Xue X, Ou Q, Wu X, Liang Y, et al. Clinical outcomes of EGFR kinase domain duplication to targeted therapies in NSCLC. Int J Cancer. 2019; 144:2677–82.
Article26. Kim JT, Zhang W, Lopategui J, Vail E, Balmanoukian A. Patient with stage IV NSCLC and CNS metastasis with EGFR exon 18–25 kinase domain duplication with response to osimertinib as a first-line therapy. JCO Precision Oncol. 2021; 5:88–92.27. Thress KS, Paweletz CP, Felip E, Cho BC, Stetson D, Dougherty B, et al. Acquired EGFR C797S mutation mediates resistance to AZD9291 in non-small cell lung cancer harboring EGFR T790M. Nat Med. 2015; 21:560–2.
Article28. Niederst MJ, Hu H, Mulvey HE, Lockerman EL, Garcia AR, Piotrowska Z, et al. The allelic context of the C797S mutation acquired upon treatment with third-generation EGFR inhibitors impacts sensitivity to subsequent treatment strategies. Clin Cancer Res. 2015; 21:3924–33.
Article29. Park S, Ku BM, Jung HA, Sun JM, Ahn JS, Lee SH, et al. EGFR C797S as a resistance mechanism of lazertinib in non-small cell lung cancer with EGFR T790M mutation. Cancer Res Treat. 2020; 52:1288–90.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- EGFR Tyrosine Kinase Inhibitors for NSCLC
- Chronicles of EGFR Tyrosine Kinase Inhibitors: Targeting EGFR C797S Containing Triple Mutations
- Mechanisms of Acquired Resistance to Epidermal Growth Factor Receptor Inhibitors and Overcoming Strategies in Lung Cancer
- Treatment of Non-small Cell Lung Carcinoma after Failure of Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitor
- EGFR C797S as a Resistance Mechanism of Lazertinib in Non-small Cell Lung Cancer with EGFR T790M Mutation