Korean J Gastroenterol.
2020 Sep;76(3):167-170. 10.4166/kjg.2020.76.3.16.
Severe Persistent Jaundice after the Abuse of an Anabolic Androgenic Steroid Analogue
- Affiliations
-
- 1Departments of Internal Medicine, Korea University College of Medicine, Seoul, Korea
- 2Departments of Pathology, Korea University College of Medicine, Seoul, Korea
- KMID: 2506674
- DOI: http://doi.org/10.4166/kjg.2020.76.3.16
Abstract
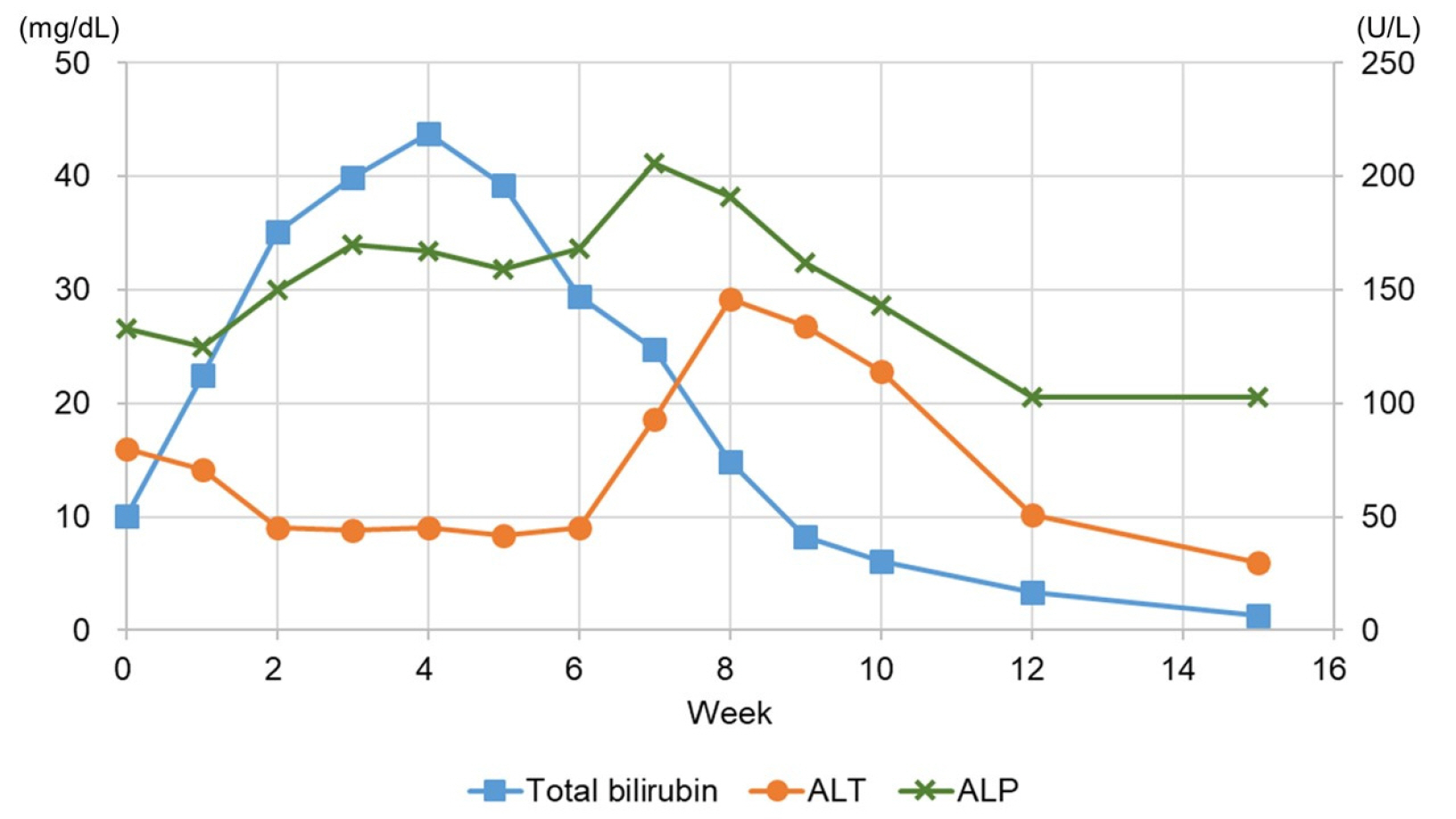
- Hepatic disorders with prominent cholestasis can be caused by a range of conditions, and anabolic androgenic steroids have been considered a cause of protracted cholestasis. A 29-year-old man who had taken an anabolic androgenic steroid analogue for 2 months visited the hospital complaining of jaundice and indigestion. After stopping the medication, the hyperbilirubinemia tended to decrease, but a transiently elevated aminotransferase level was observed. The endogenous testosterone level also decreased initially but recovered soon after. The liver function profiles were normalized after 2 months of conservative management. This case emphasizes that close drug history taking, including anabolic steroids, is important for identifying the cause of unexplained persistent jaundice.
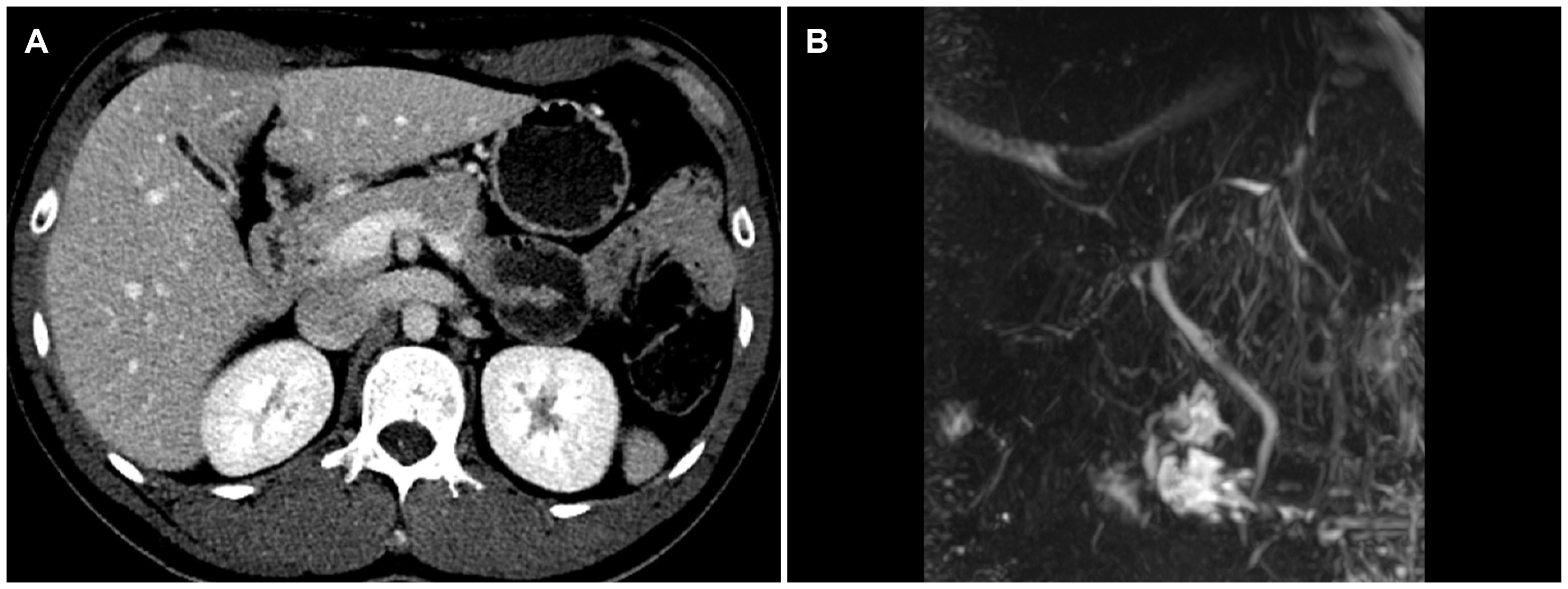
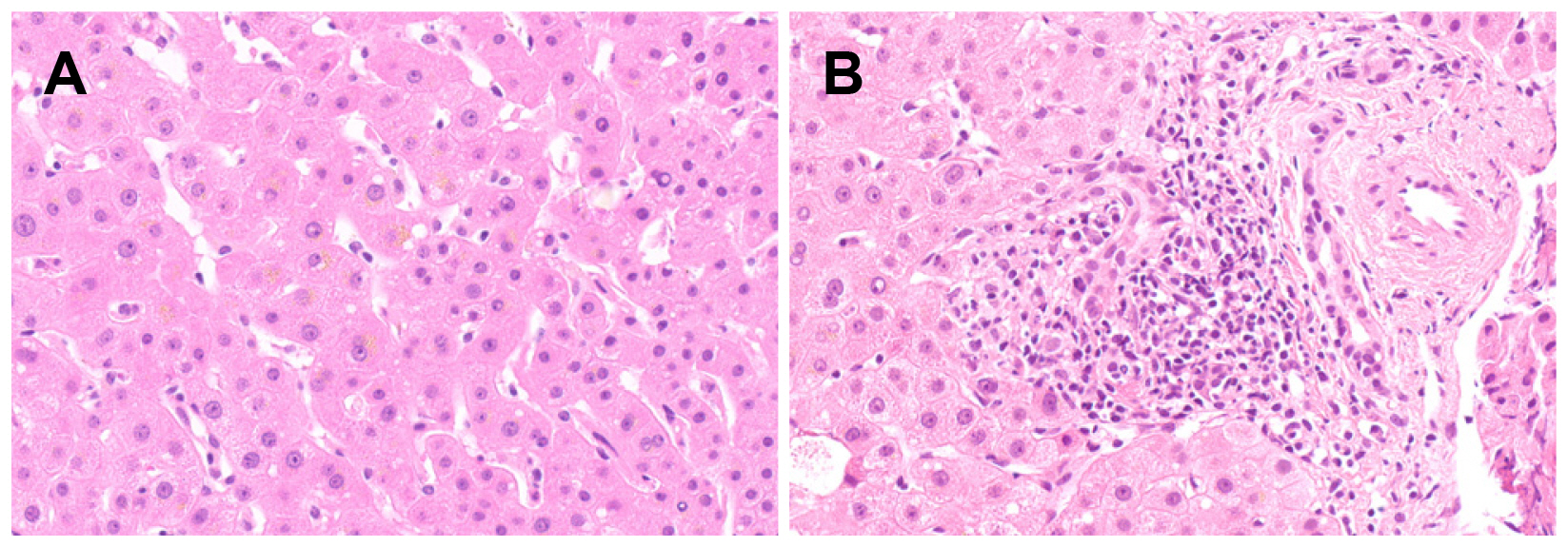
Figure
Reference
-
1. Kwo PY, Cohen SM, Lim JK. 2017; ACG clinical guideline: evaluation of abnormal liver chemistries. Am J Gastroenterol. 112:18–35. DOI: 10.1038/ajg.2016.517. PMID: 27995906.
Article2. Fargo MV, Grogan SP, Saguil A. 2017; Evaluation of jaundice in adults. Am Fam Physician. 95:164–168. PMID: 28145671.3. Strassburg CP. 2010; Hyperbilirubinemia syndromes (Gilbert-Meulengracht, Crigler-Najjar, Dubin-Johnson, and Rotor syndrome). Best Pract Res Clin Gastroenterol. 24:555–571. DOI: 10.1016/j.bpg.2010.07.007. PMID: 20955959.
Article4. Robles-Diaz M, Gonzalez-Jimenez A, Medina-Caliz I, et al. 2015; Distinct phenotype of hepatotoxicity associated with illicit use of anabolic androgenic steroids. Aliment Pharmacol Ther. 41:116–125. DOI: 10.1111/apt.13023. PMID: 25394890.
Article5. Elsharkawy AM, McPherson S, Masson S, Burt AD, Dawson RT, Hudson M. 2012; Cholestasis secondary to anabolic steroid use in young men. BMJ. 344:e468. DOI: 10.1136/bmj.e468. PMID: 22302781.
Article6. European Association for the Study of the Liver. 2019; EASL clinical practice guidelines: drug-induced liver injury. J Hepatol. 70:1222–1261. DOI: 10.1016/j.jhep.2019.02.014. PMID: 30926241.7. Small M, Beastall GH, Semple CG, Cowan RA, Forbes CD. 1984; Alteration of hormone levels in normal males given the anabolic steroid stanozolol. Clin Endocrinol (Oxf). 21:49–55. DOI: 10.1111/j.1365-2265.1984.tb00135.x. PMID: 6430603. PMCID: PMC5605175.
Article8. Wehr E, Pilz S, Boehm BO, März W, Obermayer-Pietsch B. 2010; Association of vitamin D status with serum androgen levels in men. Clin Endocrinol (Oxf). 73:243–248. DOI: 10.1111/j.1365-2265.2009.03777.x. PMID: 20050857. PMCID: PMC5992301.
Article9. Lee DM, Tajar A, Pye SR, et al. 2012; Association of hypogonadism with vitamin D status: the European Male Ageing Study. Eur J Endocrinol. 166:77–85. DOI: 10.1530/EJE-11-0743. PMID: 22048968.
Article10. Kim TH, Yun SG, Choi J, et al. 2020; Differential impact of serum 25-hydroxyvitamin D3 levels on the prognosis of patients with liver cirrhosis according to MELD and Child-Pugh scores. J Korean Med Sci. 35:e129. DOI: 10.3346/jkms.2020.35.e129. PMID: 32419396. PMCID: PMC7234861.
Article11. Díaz FC, Sáez-González E, Benlloch S, et al. 2016; Albumin dialysis with MARS for the treatment of anabolic steroid-induced cholestasis. Ann Hepatol. 15:939–943. DOI: 10.5604/16652681.1222114. PMID: 27740530.12. Krishnan PV, Feng ZZ, Gordon SC. 2009; Prolonged intrahepatic cholestasis and renal failure secondary to anabolic androgenic steroid-enriched dietary supplements. J Clin Gastroenterol. 43:672–675. DOI: 10.1097/MCG.0b013e318188be6d. PMID: 19238093.
Article13. Rosenfeld GA, Chang A, Poulin M, Kwan P, Yoshida E. 2011; Cholestatic jaundice, acute kidney injury and acute pancreatitis secondary to the recreational use of methandrostenolone: a case report. J Med Case Rep. 5:138. DOI: 10.1186/1752-1947-5-138. PMID: 21470406. PMCID: PMC3079674.
Article14. Niedfeldt MW. 2018; Anabolic steroid effect on the liver. Curr Sports Med Rep. 17:97–102. DOI: 10.1249/JSR.0000000000000467. PMID: 29521706.
Article15. Solimini R, Rotolo MC, Mastrobattista L, et al. 2017; Hepatotoxicity associated with illicit use of anabolic androgenic steroids in doping. Eur Rev Med Pharmacol Sci. 21(Suppl 1):7–16. PMID: 28379599.16. El Khoury C, Sabbouh T, Farhat H, Ferzli A. 2017; Severe cholestasis and bile cast nephropathy induced by anabolic steroids successfully treated with plasma exchange. Case Rep Med. 2017:4296474. DOI: 10.1155/2017/4296474. PMID: 29391869. PMCID: PMC5748144.
Article17. Morgan RE, Trauner M, van Staden CJ, et al. 2010; Interference with bile salt export pump function is a susceptibility factor for human liver injury in drug development. Toxicol Sci. 118:485–500. DOI: 10.1093/toxsci/kfq269. PMID: 20829430.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Spontaneous Corpus Cavernosum Abscess in a Healthy Man Using Long-Term Androgenic Anabolic Steroids
- Stevens-Johnson syndrome and abuse of anabolic steroids
- Acne Conglobata Induced by Anabolic Androgenic Steroids
- Hepatocellular carcinoma in body builders; an emerging rare but serious complication of androgenic anabolic steroid use
- Management of Anabolic Steroid-Induced Infertility: Novel Strategies for Fertility Maintenance and Recovery