Allergy Asthma Immunol Res.
2020 May;12(3):430-442. 10.4168/aair.2020.12.3.430.
Evaluation of Drug-Induced Liver Injury Developed During Hospitalization Using Electronic Health Record (EHR)-Based Algorithm
- Affiliations
-
- 1Department of Allergy and Clinical Immunology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. tbkim@amc.seoul.kr
- 2Pharmacovigilance Center, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- 3Department of Internal Medicine, Pusan National University School of Medicine, Busan, Korea.
- 4Department of Internal Medicine, Seoul National University Bundang Hospital, Seongnam, Korea.
- 5Department of Allergy and Respiratory Medicine, Konkuk University Medical Center, Seoul, Korea.
- 6Department of Internal Medicine, VHS Medical Center, Seoul, Korea.
- 7Department of Gastroenterology, Asan Liver Center, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- 8Department of Internal Medicine, Seoul Metropolitan Government-Seoul National University Boramae Medical Center, Seoul, Korea. iatrus13@hanmail.net
- KMID: 2471227
- DOI: http://doi.org/10.4168/aair.2020.12.3.430
Abstract
- PURPOSE
The incidence of drug-induced liver injury (DILI) has been increasing; however, few algorithms are available to identify DILI in electronic health records (EHRs). We aimed to identify and evaluate DILI with an appropriate screening algorithm.
METHODS
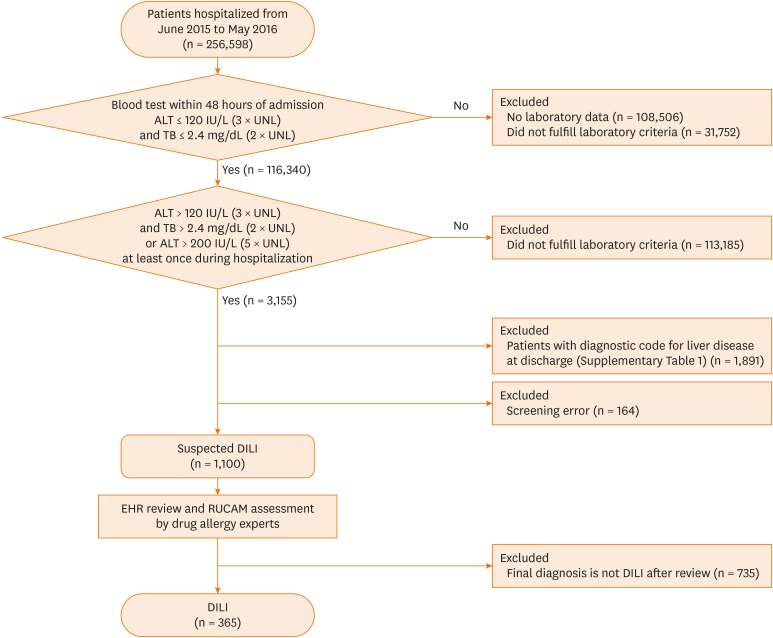
We collected data from 3 university hospitals between June 2015 and May 2016 using our newly developed algorithm for identifying DILI. Among patients with alanine transferase (ALT) ≤ 120 IU/L and total bilirubin (TB) ≤ 2.4 mg/dL in blood test results within 48 hours of admission, those who either had 1) ALT > 120 IU/L and TB > 2.4 mg/dL or 2) ALT > 200 IU/L at least once during hospitalization were identified. After excluding patients with liver disease-related diagnosis at discharge, medical records were retrospectively reviewed to evaluate epidemiological characteristics of DILI.
RESULTS
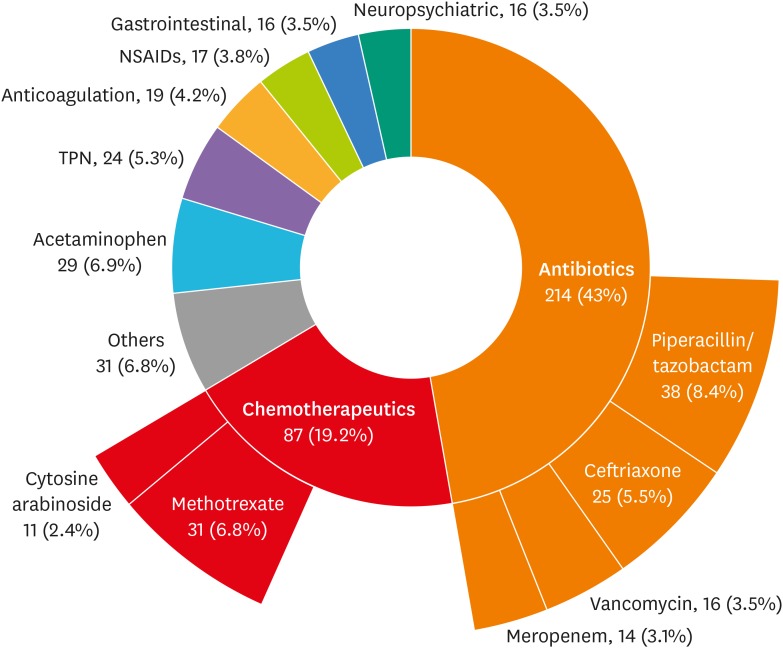
The total number of inpatients was 256,598, of whom 1,100 (0.43%) were selected by the algorithm as suspected DILI. Subsequently, 365 cases (0.14% of total inpatients, 95% confidence interval, 0.13-0.16) were identified as DILI, yielding a positive predictive value of 33.1%. Antibiotics (n = 214, 47.2%) were the major class of causative drug followed by chemotherapeutic agents (n = 87, 19.2%). The most common causative drug was piperacillin-tazobactam (n = 38, 8.4%); the incidence of DILI by individual agent was highest for methotrexate (19.4 cases/1,000 patients administered the drug). Common reasons for excluding suspected DILI cases were ischemic hepatitis and postoperative liver dysfunction.
CONCLUSIONS
Using our EHR-based algorithm, we identified that approximately 0.14% of patients developed DILI during hospitalization. Further studies are needed to modify criteria for more accurate identification of DILI.
Keyword
MeSH Terms
-
Alanine
Anti-Bacterial Agents
Bilirubin
Diagnosis
Drug-Induced Liver Injury*
Drug-Related Side Effects and Adverse Reactions
Electronic Health Records*
Hematologic Tests
Hepatitis
Hospitalization*
Hospitals, University
Humans
Incidence
Inpatients
Liver
Liver Diseases
Mass Screening
Medical Records
Methotrexate
Pharmacoepidemiology
Retrospective Studies
Transferases
Alanine
Anti-Bacterial Agents
Bilirubin
Methotrexate
Transferases
Figure
Cited by 2 articles
-
Active Pharmacovigilance of Drug-Induced Liver Injury Using Electronic Health Records
Sang-Heon Kim
Allergy Asthma Immunol Res. 2020;12(3):378-380. doi: 10.4168/aair.2020.12.3.378.Incidence and Economic Burden of Adverse Drug Reactions in Hospitalization: A Prospective Study in Korea
Bomi Seo, Min-Suk Yang, So-Young Park, Bo Young Park, Jung-Hyun Kim, Woo-Jung Song, Hyouk-Soo Kwon, Yoon-Seok Chang, You Sook Cho, Sae-Hoon Kim, Tae-Bum Kim
J Korean Med Sci. 2023;38(8):e56. doi: 10.3346/jkms.2023.38.e56.
Reference
-
1. Björnsson ES. Epidemiology and risk factors for idiosyncratic drug-induced liver injury. Semin Liver Dis. 2014; 34:115–122. PMID: 24879977.
Article2. Andrade RJ, Lucena MI, Fernández MC, Pelaez G, Pachkoria K, García-Ruiz E, et al. Drug-induced liver injury: an analysis of 461 incidences submitted to the Spanish registry over a 10-year period. Gastroenterology. 2005; 129:512–521. PMID: 16083708.
Article3. Hoofnagle JH. Drug-induced liver injury network (DILIN). Hepatology. 2004; 40:773. PMID: 15382161.
Article4. Bakke OM, Manocchia M, de Abajo F, Kaitin KI, Lasagna L. Drug safety discontinuations in the United Kingdom, the United States, and Spain from 1974 through 1993: a regulatory perspective. Clin Pharmacol Ther. 1995; 58:108–117. PMID: 7628177.
Article5. Navarro VJ, Senior JR. Drug-related hepatotoxicity. N Engl J Med. 2006; 354:731–739. PMID: 16481640.
Article6. Chalasani N, Fontana RJ, Bonkovsky HL, Watkins PB, Davern T, Serrano J, et al. Causes, clinical features, and outcomes from a prospective study of drug-induced liver injury in the United States. Gastroenterology. 2008; 135:1924–1934. 1934.e1–1924. PMID: 18955056.
Article7. Robles-Diaz M, Lucena MI, Kaplowitz N, Stephens C, Medina-Cáliz I, González-Jimenez A, et al. Use of Hy's law and a new composite algorithm to predict acute liver failure in patients with drug-induced liver injury. Gastroenterology. 2014; 147:109–118.e5. PMID: 24704526.
Article8. Chalasani N, Bonkovsky HL, Fontana R, Lee W, Stolz A, Talwalkar J, et al. Features and outcomes of 899 patients with drug-induced liver injury: the DILIN prospective study. Gastroenterology. 2015; 148:1340–1352.e7. PMID: 25754159.9. Chalasani NP, Hayashi PH, Bonkovsky HL, Navarro VJ, Lee WM, Fontana RJ, et al. ACG clinical guideline: the diagnosis and management of idiosyncratic drug-induced liver injury. Am J Gastroenterol. 2014; 109:950–966. PMID: 24935270.
Article10. Teschke R, Frenzel C, Wolff A, Eickhoff A, Schulze J. Drug induced liver injury: accuracy of diagnosis in published reports. Ann Hepatol. 2014; 13:248–255. PMID: 24552867.
Article11. Chalasani N, Regev A. Drug-induced liver injury in patients with preexisting chronic liver disease in drug development: how to identify and manage? Gastroenterology. 2016; 151:1046–1051. PMID: 27765688.
Article12. Jinjuvadia K, Kwan W, Fontana RJ. Searching for a needle in a haystack: use of ICD-9-CM codes in drug-induced liver injury. Am J Gastroenterol. 2007; 102:2437–2443. PMID: 17662100.
Article13. Tan EH, Low EX, Dan YY, Tai BC. Systematic review and meta-analysis of algorithms used to identify drug-induced liver injury (DILI) in health record databases. Liver Int. 2018; 38:742–753. PMID: 29193566.
Article14. Aithal GP, Watkins PB, Andrade RJ, Larrey D, Molokhia M, Takikawa H, et al. Case definition and phenotype standardization in drug-induced liver injury. Clin Pharmacol Ther. 2011; 89:806–815. PMID: 21544079.
Article15. Danan G, Benichou C. Causality assessment of adverse reactions to drugs--I. A novel method based on the conclusions of international consensus meetings: application to drug-induced liver injuries. J Clin Epidemiol. 1993; 46:1323–1330. PMID: 8229110.
Article16. Bénichou C. Criteria of drug-induced liver disorders. Report of an international consensus meeting. J Hepatol. 1990; 11:272–276. PMID: 2254635.17. de Abajo FJ, Montero D, Madurga M, García Rodríguez LA. Acute and clinically relevant drug-induced liver injury: a population based case-control study. Br J Clin Pharmacol. 2004; 58:71–80. PMID: 15206996.
Article18. De Valle MB, Av Klinteberg V, Alem N, Olsson R, Björnsson E. Drug-induced liver injury in a Swedish university hospital out-patient hepatology clinic. Aliment Pharmacol Ther. 2006; 24:1187–1195. PMID: 17014577.
Article19. Bjornsson ES, Bergmann OM, Bjornsson HK, Kvaran RB, Olafsson S. Incidence, presentation, and outcomes in patients with drug-induced liver injury in the general population of Iceland. Gastroenterology. 2013; 144:1419–1425. 1425.e1–1413. PMID: 23419359.
Article20. Sgro C, Clinard F, Ouazir K, Chanay H, Allard C, Guilleminet C, et al. Incidence of drug-induced hepatic injuries: a French population-based study. Hepatology. 2002; 36:451–455. PMID: 12143055.
Article21. Suk KT, Kim DJ, Kim CH, Park SH, Yoon JH, Kim YS, et al. A prospective nationwide study of drug-induced liver injury in Korea. Am J Gastroenterol. 2012; 107:1380–1387. PMID: 22733303.
Article22. Meier Y, Cavallaro M, Roos M, Pauli-Magnus C, Folkers G, Meier PJ, et al. Incidence of drug-induced liver injury in medical inpatients. Eur J Clin Pharmacol. 2005; 61:135–143. PMID: 15726344.
Article23. Temple R. Hy’s law: predicting serious hepatotoxicity. Pharmacoepidemiol Drug Saf. 2006; 15:241–243. PMID: 16552790.
Article24. Björnsson E, Olsson R. Outcome and prognostic markers in severe drug-induced liver disease. Hepatology. 2005; 42:481–489. PMID: 16025496.
Article25. U.S. Department of Health and Human Services Food and Drug Administration, Center for Drug Evaluation and Research (CDER), Center for Biologics Evaluation and Research (CBER). Drug-induced liver injury: premarketing clinical evaluation: July 2009. Silver Spring: U.S. Food and Drug Administration;2019. cited 2019 Apr 19. Available from: https://www.fda.gov/downloads/guidances/UCM174090.pdf.26. Hunt CM. Expanding our toolkit to better identify drug-induced liver injury in electronic medical records. Liver Int. 2018; 38:585–587. PMID: 29575769.
Article27. Classen DC, Pestotnik SL, Evans RS, Burke JP. Computerized surveillance of adverse drug events in hospital patients. JAMA. 1991; 266:2847–2851. PMID: 1942452.
Article28. Tegeder I, Levy M, Muth-Selbach U, Oelkers R, Neumann F, Dormann H, et al. Retrospective analysis of the frequency and recognition of adverse drug reactions by means of automatically recorded laboratory signals. Br J Clin Pharmacol. 1999; 47:557–564. PMID: 10336581.
Article29. Sistanizad M, Peterson GM. Drug-induced liver injury in the Australian setting. J Clin Pharm Ther. 2013; 38:115–120. PMID: 23350857.
Article30. Cheetham TC, Lee J, Hunt CM, Niu F, Reisinger S, Murray R, et al. An automated causality assessment algorithm to detect drug-induced liver injury in electronic medical record data. Pharmacoepidemiol Drug Saf. 2014; 23:601–608. PMID: 24920207.
Article31. Bagheri H, Michel F, Lapeyre-Mestre M, Lagier E, Cambus JP, Valdiguié P, et al. Detection and incidence of drug-induced liver injuries in hospital: a prospective analysis from laboratory signals. Br J Clin Pharmacol. 2000; 50:479–484. PMID: 11069443.
Article32. Devarbhavi H, Dierkhising R, Kremers WK, Sandeep MS, Karanth D, Adarsh CK. Single-center experience with drug-induced liver injury from India: causes, outcome, prognosis, and predictors of mortality. Am J Gastroenterol. 2010; 105:2396–2404. PMID: 20648003.
Article33. Björnsson ES. Drug-induced liver injury due to antibiotics. Scand J Gastroenterol. 2017; 52:617–623. PMID: 28276834.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Comparison of Open-Source Electronic Health Record Systems Based on Functional and User Performance Criteria
- Narrative Review for Exploring Barriers to Readiness of Electronic Health Record Implementation in Primary Health Care
- Electronic Health Record: Definition, Categories and Standards
- Pilot Algorithm Designed to Help Early Detection of HMG-CoA Reductase Inhibitor-Induced Hepatotoxicity
- Active Pharmacovigilance of Drug-Induced Liver Injury Using Electronic Health Records