Korean J Ophthalmol.
2020 Feb;34(1):11-18. 10.3341/kjo.2019.0081.
Changes in the Ganglion Cell-inner Plexiform Layer after Consecutive Intravitreal Injections of Anti-vascular Endothelial Growth Factor in Age-related Macular Degeneration Patients
- Affiliations
-
- 1Department of Ophthalmology and Inha Vision Science Laboratory, Inha University Hospital, Inha University School of Medicine, Incheon, Korea. hschin@inha.ac.kr
- KMID: 2469282
- DOI: http://doi.org/10.3341/kjo.2019.0081
Abstract
- PURPOSE
To investigate the effect of intravitreal anti-vascular endothelial growth factor (VEGF) injections on ganglion cell-inner plexiform layer (GCIPL) thickness in patients with age-related macular degeneration (AMD).
METHODS
This retrospective study included patients with continuous anti-VEGF treatment who were administered at least three consecutive injections for unilateral neovascular AMD. The GCIPL thickness of the study eyes was compared before and after treatment and with healthy fellow eyes using spectral-domain optical coherence tomography. We also evaluated best-corrected visual acuity, age, and intraocular pressure.
RESULTS
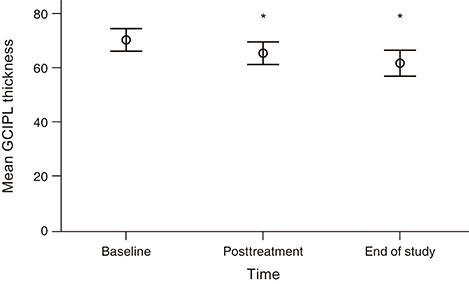
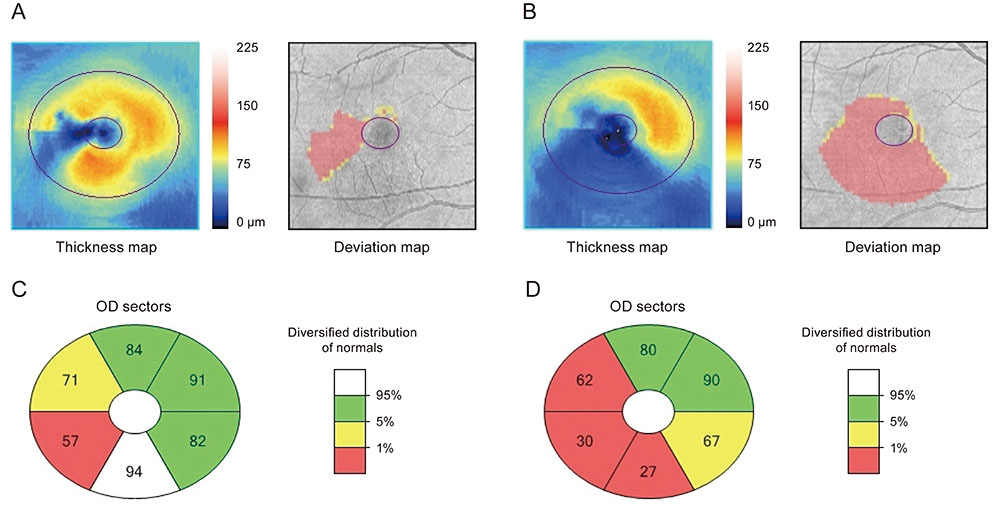
In total, 96 eyes of 48 patients (14 females and 34 males; mean ± standard deviation [SD] age, 70.10 ± 8.89 years) with mean number of 6.29 (SD ± 3.76) anti-VEGF injections and a mean follow-up period of 24.93 months (SD ± 19.86) were included in the study. After three consecutive intravitreal injections of anti-VEGF, the mean GCIPL thickness was significantly reduced from 70.50 (SD ± 14.06) to 65.97 (SD ± 13.91) µm. Borderline or nonsignificant decrease was also observed in GCIPL thickness for each sector. At the end of the study, the mean GCIPL thickness was further reduced to 62.56 (SD ± 16.30) µm, and significant decreases were also observed in all other sectors compared with baseline.
CONCLUSIONS
It has been observed that GCIPL thickness can decrease with only three consecutive anti-VEGF injections as well as with long-term treatment in AMD patients.
Keyword
MeSH Terms
Figure
Reference
-
1. Sobrin L, Seddon JM. Nature and nurture- genes and environment-predict onset and progression of macular degeneration. Prog Retin Eye Res. 2014; 40:1–15.2. Bird AC, Bressler NM, Bressler SB, et al. An international classification and grading system for age-related maculopathy and age-related macular degeneration. The International ARM Epidemiological Study Group. Surv Ophthalmol. 1995; 39:367–374.3. Ferris FL 3rd, Fine SL, Hyman L. Age-related macular degeneration and blindness due to neovascular maculopathy. Arch Ophthalmol. 1984; 102:1640–1642.
Article4. Bressler NM, Bressler SB, Fine SL. Age-related macular degeneration. Surv Ophthalmol. 1988; 32:375–413.
Article5. Ambati J, Ambati BK, Yoo SH, et al. Age-related macular degeneration: etiology, pathogenesis, and therapeutic strategies. Surv Ophthalmol. 2003; 48:257–293.
Article6. CATT Research Group. Martin DF, Maguire MG, et al. Ranibizumab and bevacizumab for neovascular age-related macular degeneration. N Engl J Med. 2011; 364:1897–1908.
Article7. Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group. Martin DF, Maguire MG, et al. Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: two-year results. Ophthalmology. 2012; 119:1388–1398.8. Blaauwgeers HG, Holtkamp GM, Rutten H, et al. Polarized vascular endothelial growth factor secretion by human retinal pigment epithelium and localization of vascular endothelial growth factor receptors on the inner choriocapillaris. Evidence for a trophic paracrine relation. Am J Pathol. 1999; 155:421–428.9. Marneros AG, Fan J, Yokoyama Y, et al. Vascular endothelial growth factor expression in the retinal pigment epithelium is essential for choriocapillaris development and visual function. Am J Pathol. 2005; 167:1451–1459.
Article10. Nishijima K, Ng YS, Zhong L, et al. Vascular endothelial growth factor-A is a survival factor for retinal neurons and a critical neuroprotectant during the adaptive response to ischemic injury. Am J Pathol. 2007; 171:53–67.
Article11. Cho HJ, Yoo SG, Kim HS, et al. Risk factors for geographic atrophy after intravitreal ranibizumab injections for retinal angiomatous proliferation. Am J Ophthalmol. 2015; 159:285–292.
Article12. Zinkernagel MS, Schorno P, Ebneter A, Wolf S. Scleral thinning after repeated intravitreal injections of antivascular endothelial growth factor agents in the same quadrant. Invest Ophthalmol Vis Sci. 2015; 56:1894–1900.
Article13. Foxton RH, Finkelstein A, Vijay S, et al. VEGF-A is necessary and sufficient for retinal neuroprotection in models of experimental glaucoma. Am J Pathol. 2013; 182:1379–1390.
Article14. Lee JM, Bae HW, Lee SY, et al. Effect of anti-vascular endothelial growth factor antibody on the survival of cultured retinal ganglion cells. Korean J Ophthalmol. 2017; 31:360–365.
Article15. Zucchiatti I, Parodi MB, Pierro L, et al. Macular ganglion cell complex and retinal nerve fiber layer comparison in different stages of age-related macular degeneration. Am J Ophthalmol. 2015; 160:602–607.
Article16. Brar VS, Sharma RK, Murthy RK, Chalam KV. Bevacizumab neutralizes the protective effect of vascular endothelial growth factor on retinal ganglion cells. Mol Vis. 2010; 16:1848–1853.17. Cheng CK, Peng PH, Tien LT, et al. Bevacizumab is not toxic to retinal ganglion cells after repeated intravitreal injection. Retina. 2009; 29:306–312.
Article18. Horsley MB, Mandava N, Maycotte MA, Kahook MY. Retinal nerve fiber layer thickness in patients receiving chronic anti-vascular endothelial growth factor therapy. Am J Ophthalmol. 2010; 150:558–561.
Article19. Entezari M, Ramezani A, Yaseri M. Changes in retinal nerve fiber layer thickness after two intravitreal bevacizumab injections for wet type age-related macular degeneration. J Ophthalmic Vis Res. 2014; 9:449–452.
Article20. Shin HJ, Shin KC, Chung H, Kim HC. Change of retinal nerve fiber layer thickness in various retinal diseases treated with multiple intravitreal antivascular endothelial growth factor. Invest Ophthalmol Vis Sci. 2014; 55:2403–2411.
Article21. Shin HY, Park HY, Jung KI, Park CK. Comparative study of macular ganglion cell-inner plexiform layer and peripapillary retinal nerve fiber layer measurement: structure-function analysis. Invest Ophthalmol Vis Sci. 2013; 54:7344–7353.
Article22. Ohkubo S, Higashide T, Udagawa S, et al. Focal relationship between structure and function within the central 10 degrees in glaucoma. Invest Ophthalmol Vis Sci. 2014; 55:5269–5277.
Article23. Beck M, Munk MR, Ebneter A, et al. Retinal ganglion cell layer change in patients treated with anti-vascular endothelial growth factor for neovascular age-related macular degeneration. Am J Ophthalmol. 2016; 167:10–17.
Article24. Lee HJ, Kim MS, Jo YJ, Kim JY. Ganglion cell-inner plexiform layer thickness in retinal diseases: repeatability study of spectral-domain optical coherence tomography. Am J Ophthalmol. 2015; 160:283–289.
Article25. Alshareef RA, Goud A, Mikhail M, et al. Segmentation errors in macular ganglion cell analysis as determined by optical coherence tomography in eyes with macular pathology. Int J Retina Vitreous. 2017; 3:25.
Article26. Kim HJ, Chung H, Kim HC. Artifacts associated with spectral-domain optical coherence tomography. J Korean Ophthalmol Soc. 2011; 52:943–951.
Article27. Ho J, Sull AC, Vuong LN, et al. Assessment of artifacts and reproducibility across spectral- and time-domain optical coherence tomography devices. Ophthalmology. 2009; 116:1960–1970.
Article28. Patel PJ, Chen FK, da Cruz L, Tufail A. Segmentation error in Stratus optical coherence tomography for neovascular age-related macular degeneration. Invest Ophthalmol Vis Sci. 2009; 50:399–404.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Treatment of Exudative Age-Related Macular Degeneration
- Ganglion Cell Layer Thickness after Anti-Vascular Endothelial Growth Factor Treatment in Retinal Vein Occlusion
- Comparison of Each Retinal Layer Thicknesses between Eyes with Central Retinal Vein Occlusion and Normal Contralateral Eyes
- The Effect of Intravitreal Bevacizumab Injection in Wet Age-Related Macular Degeneration Patients with Cataract Surgery
- Intravitreal injection of anti-vascular endothelial growth factor for patients with various retinal diseases