Clin Exp Otorhinolaryngol.
2019 May;12(2):206-211. 10.21053/ceo.2018.01116.
Characteristics of Smell Identification Test in Patients With Parkinson Disease
- Affiliations
-
- 1Department of Otolaryngology-Head and Neck Surgery, Kobe University Graduate School of Medicine, Kobe, Japan. nibu@med.kobe-u.ac.jp
- 2Department of Rehabilitation Science, Kobe University Graduate School of Health Sciences, Kobe, Japan.
- KMID: 2447430
- DOI: http://doi.org/10.21053/ceo.2018.01116
Abstract
OBJECTIVES
Parkinson disease (PD) is frequently associated with olfactory disorder at early stage, which is caused by deposition of Lewy bodies emerging from the olfactory bulb to higher olfactory centers. Early detection of olfactory disorder in the patients with PD may lead to the early diagnosis and treatment for this refractory disease.
METHODS
Visual analog scale (VAS), Jet Stream Olfactometry, and Japanese smell identification test, Open Essence (OE), were carried out on 39 patients with PD. Thirty-one patients with postviral olfactory disorder (PVOD), which was caused by the olfactory mucosal dysfunction, were also enrolled in this study as control.
RESULTS
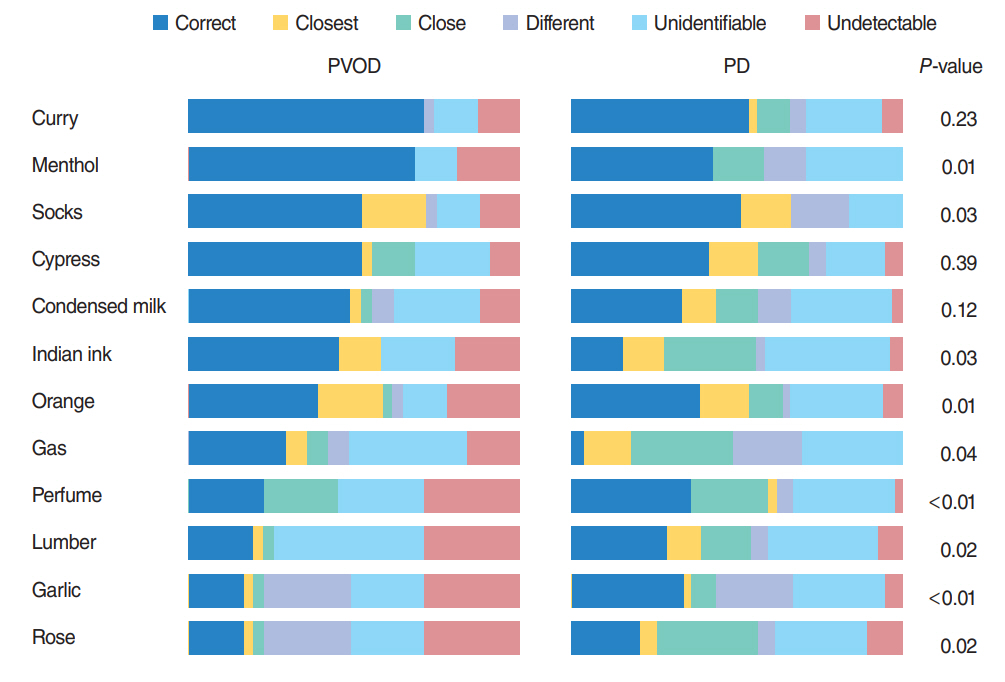
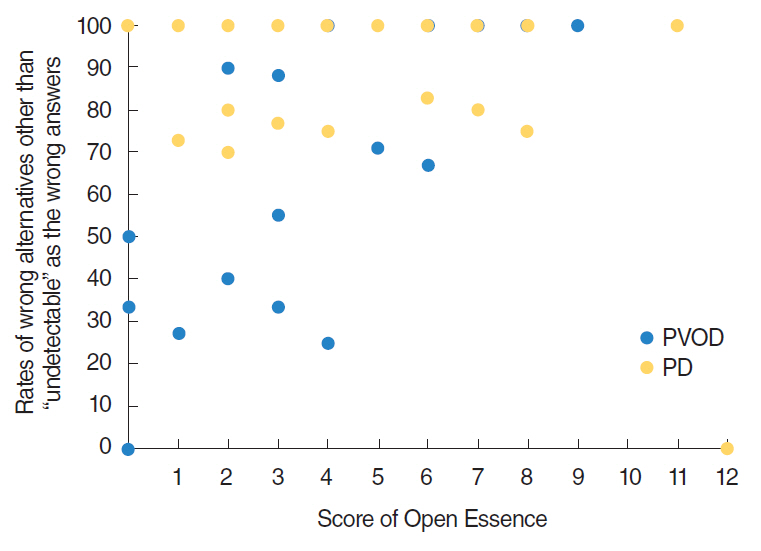
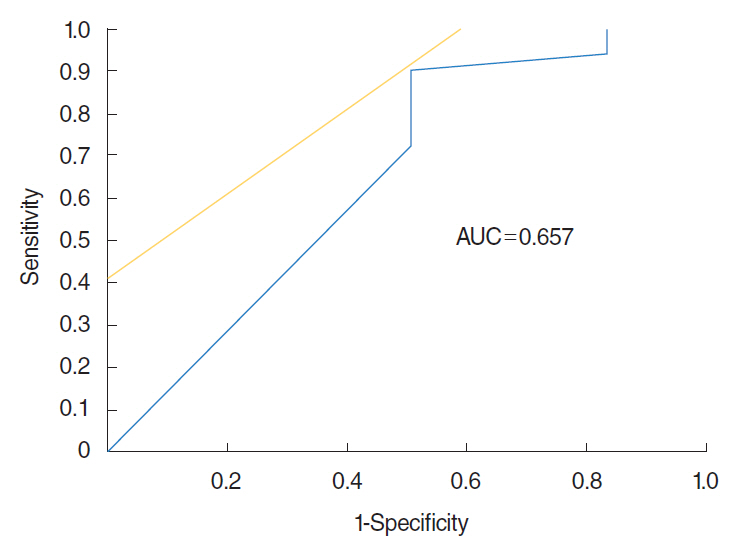
There were no significant differences in detection thresholds (2.2 vs. 1.4, P=0.13), recognition thresholds (3.9 vs. 3.5, P=0.39) and OE (4.8 vs. 4.2, P=0.47) between PVOD and PD, while VAS scores of PVOD and PD were significantly different (2.0 and 6.2, P<0.01). In OE, significant differences were observed in the accuracy rates of menthol (68% vs. 44%, P=0.04) and Indian ink (42% vs. 15%, P=0.01) between PVOD and PD. Of particular interest, patients with PVOD tended to select "no detectable," while patients with PD tended to select wrong alternative other than "no smell detected."
CONCLUSION
Discrepancy between VAS and OE, and high selected rates of wrong alternative other than "undetectable" in OE might be significant signs of olfactory dysfunction associated with PD.
MeSH Terms
Figure
Cited by 1 articles
-
Can Olfactory Tests Help to Diagnose Parkinson Disease?
Jin Kook Kim
Clin Exp Otorhinolaryngol. 2019;12(2):105-106. doi: 10.21053/ceo.2019.00199.
Reference
-
1. Braak H, Del Tredici K, Rub U, de Vos RA, Jansen Steur EN, Braak E. Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging. 2003; Mar-Apr. 24(2):197–211.
Article2. Ansari KA, Johnson A. Olfactory function in patients with Parkinson’s disease. J Chronic Dis. 1975; Oct. 28(9):493–7.
Article3. Hawkes CH, Shephard BC, Daniel SE. Olfactory dysfunction in Parkinson’s disease. J Neurol Neurosurg Psychiatry. 1997; May. 62(5):436–46.
Article4. Doty RL. Olfaction in Parkinson’s disease. Parkinsonism Relat Disord. 2007; 13 Suppl 3:S225–8.
Article5. Ross GW, Petrovitch H, Abbott RD, Tanner CM, Popper J, Masaki K, et al. Association of olfactory dysfunction with risk for future Parkinson’s disease. Ann Neurol. 2008; Feb. 63(2):167–73.
Article6. Baba T, Kikuchi A, Hirayama K, Nishio Y, Hosokai Y, Kanno S, et al. Severe olfactory dysfunction is a prodromal symptom of dementia associated with Parkinson’s disease: a 3 year longitudinal study. Brain. 2012; Jan. 135(Pt 1):161–9.
Article7. Doty RL, Shaman P, Applebaum SL, Giberson R, Siksorski L, Rosenberg L. Smell identification ability: changes with age. Science. 1984; Dec. 226(4681):1441–3.
Article8. Doty RL, Shaman P, Kimmelman CP, Dann MS. University of Pennsylvania smell identification test: a rapid quantitative olfactory function test for the clinic. Laryngoscope. 1984; Feb. 94(2 Pt 1):176–8.
Article9. Doty RL, Marcus A, Lee WW. Development of the 12-item Cross-Cultural Smell Identification Test (CC-SIT). Laryngoscope. 1996; Mar. 106(3 Pt 1):353–6.10. Kobal G, Hummel T, Sekinger B, Barz S, Roscher S, Wolf S. “Sniffin’ sticks”: screening of olfactory performance. Rhinology. 1996; Dec. 34(4):222–6.11. Fujio H, Doi K, Hasegawa S, Kobayakawa T, Nibu K. Evaluation of card-type odor identification test for Japanese patients with olfactory disturbance. Ann Otol Rhinol Laryngol. 2012; Jun. 121(6):413–8.
Article12. McCormack HM, Horne DJ, Sheather S. Clinical applications of visual analogue scales: a critical review. Psychol Med. 1988; Nov. 18(4):1007–19.
Article13. Ikeda K, Tabata K, Oshima T, Nishikawa H, Hidaka H, Takasaka T. Unilateral examination of olfactory threshold using the Jet Stream Olfactometer. Auris Nasus Larynx. 1999; Oct. 26(4):435–9.
Article14. Mesholam RI, Moberg PJ, Mahr RN, Doty RL. Olfaction in neurodegenerative disease: a meta-analysis of olfactory functioning in Alzheimer’s and Parkinson’s diseases. Arch Neurol. 1998; Jan. 55(1):84–90.15. Henkin RI, Larson AL, Powell RD. Hypogeusia, dysgeusia, hyposmia, and dysosmia following influenza-like infection. Ann Otol Rhinol Laryngol. 1975; Sep-Oct. 84(5 Pt 1):672–82.
Article16. Welge-Lussen A, Wolfensberger M. Olfactory disorders following upper respiratory tract infections. Adv Otorhinolaryngol. 2006; 63:125–32.17. Takeda A, Baba T, Kikuchi A, Hasegawa T, Sugeno N, Konno M, et al. Olfactory dysfunction and dementia in Parkinson’s disease. J Parkinsons Dis. 2014; 4(2):181–7.
Article18. Fujio H, Inokuchi G, Fukuda Y, Kuroki S, Kokan N, Nibu K, et al. A new screening tool, an odor identification test card to detect age-related changes in olfaction. Nihon Jibiinkoka Gakkai Kaiho. 2018; 121:38–43.
Article19. Watanabe Y, Suzuki K, Miyamoto T, Miyamoto M, Numao A, Fujita H, et al. A card-type odor identification test for Japanese patients with Parkinson’s disease and related disorders. Intern Med. 2017; Nov. 56(21):2871–8.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Alteration of Olfactory Function in Chronic Viral Hepatitis Patients
- Evaluation of Clinical Validity of Cross-Cultural Smell Identification Test (CC-SIT) in Korean
- The Usefulness of CC-SIT(Cross-Cultural Smell Identification Test) in Korea
- Selection of Olfactory Identification Items for Koreans
- Odorant Confusion Matrix Odor Identification Test Using Synthetic Odorants