Ann Dermatol.
2018 Oct;30(5):536-543. 10.5021/ad.2018.30.5.536.
Gene Expression Patterns of Cutaneous Squamous Cell Carcinoma and Actinic Keratosis: Biomarkers Screening for Skin Disease Diagnosis
- Affiliations
-
- 1Department of Dermatology, Affiliated Hospital of Hebei University of Engineering, Handan, China. lizhiufeng369@163.com LBG66@163.com
- KMID: 2419742
- DOI: http://doi.org/10.5021/ad.2018.30.5.536
Abstract
- BACKGROUND
Actinic keratosis (AK) was an intraepidermal tumor which caused by ultraviolet irradiation-induced skin damage.
OBJECTIVE
The aim was to screen biomarkers for development of skin disease by comparing the gene expression profiles between cutaneous squamous cell carcinoma (CSCC) and AK.
METHODS
GSE45216 with 30 cutaneous squamous cell carcinoma patients and 10 actinic keratosis patients were downloaded and significance analysis of microarrays was processed to screen differently expressed genes (DEGs). Fisher's exact test was processed for DEGs enrichment. Pathway relationship network systematically reflected the signal conduction and synergism between enriched pathways based on Kyoto Encyclopedia of Genes and Genomes database. Gene co-expression network was constructed according to gene expression data. Quantitative real-time-PCR was used to verify screened biomarkers.
RESULTS
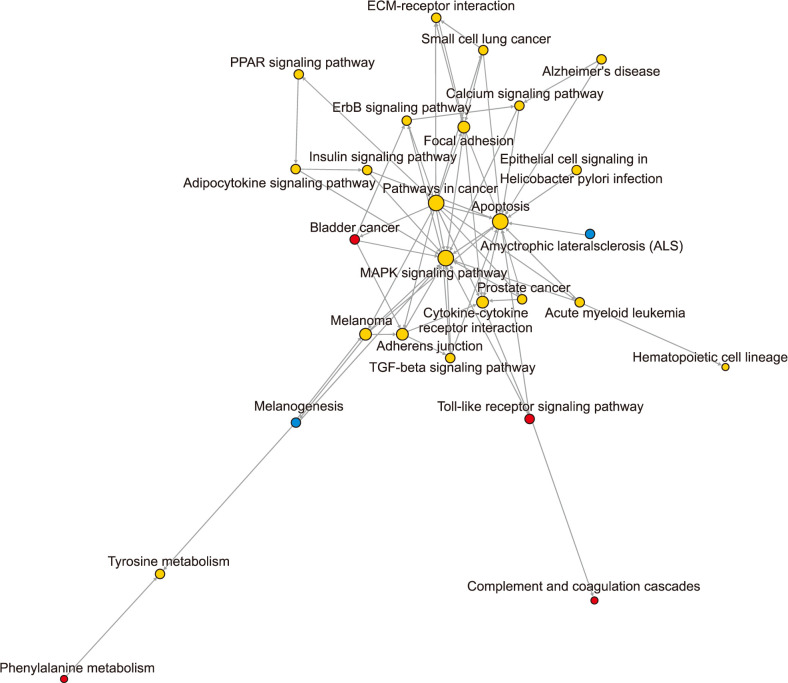
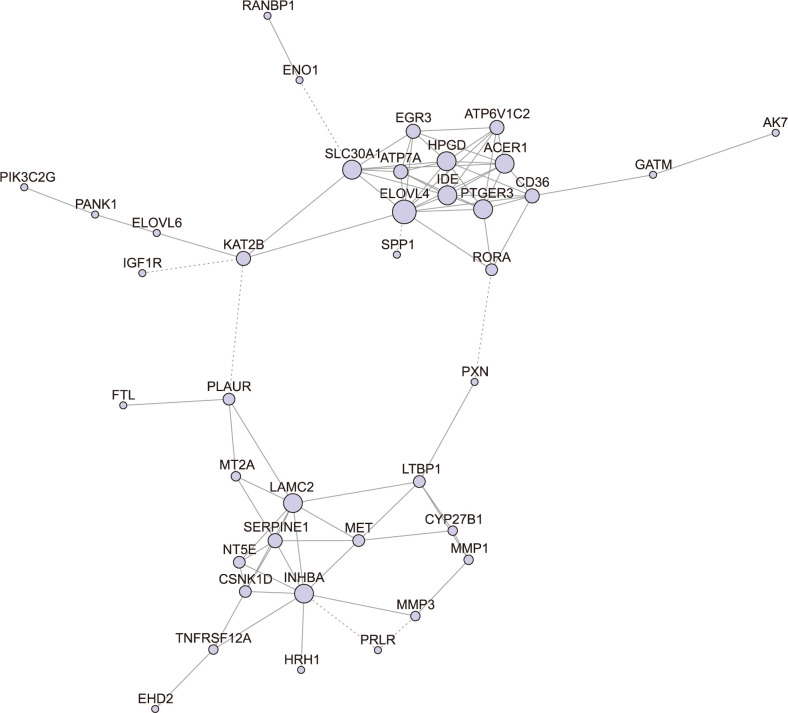
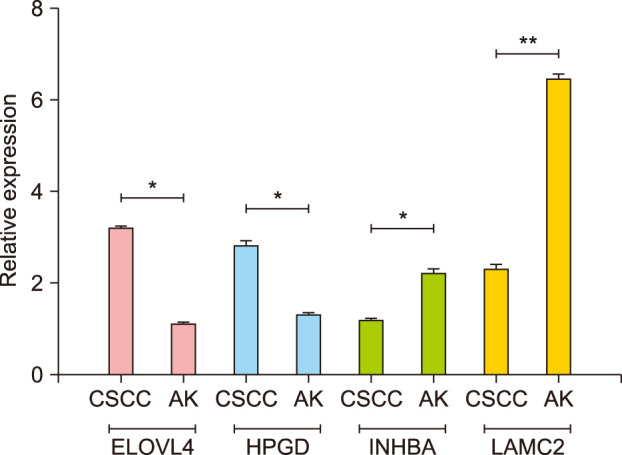
Total 410 DEGs were screened and enriched into various functions, such as signal transduction and negative regulation of apoptotic process. They also participated into cytokine-cytokine receptor interaction and focal adhesion. The pathway relationship network was constructed with 27 nodes. Hub nodes with higher degree of this network were mitogen-activated protein kinase signaling pathway and apoptosis. The gene co-expression network was constructed with 39 nodes. Thereinto, hub node was ELOVL fatty acid elongase. The expression levels of ELOVL4 and HPGD were significantly higher in CSCC samples than that in AK samples, while the expression levels of INHBA and LAMC2 in CSCC samples were significantly lower than that in AK samples.
CONCLUSION
These screened genes, including ELOVL4, HPGD, INHBA and LAMC2, played important roles in transformation from AK to CSCC.
Keyword
MeSH Terms
Figure
Reference
-
1. Xu D, Yuan R, Gu H, Liu T, Tu Y, Yang Z, et al. The effect of ultraviolet radiation on the transforming growth factor beta 1/Smads pathway and p53 in actinic keratosis and normal skin. Arch Dermatol Res. 2013; 305:777–786. PMID: 23632819.
Article2. Renzi C, Mastroeni S, Mannooranparampil TJ, Passarelli F, Caggiati A, Pasquini P. Skin cancer knowledge and preventive behaviors among patients with a recent history of cutaneous squamous cell carcinoma. Dermatology. 2008; 217:74–80. PMID: 18424897.
Article3. Ra SH, Li X, Binder S. Molecular discrimination of cutaneous squamous cell carcinoma from actinic keratosis and normal skin. Mod Pathol. 2011; 24:963–973. PMID: 21743436.
Article4. Anwar J, Wrone DA, Kimyai-Asadi A, Alam M. The development of actinic keratosis into invasive squamous cell carcinoma: evidence and evolving classification schemes. Clin Dermatol. 2004; 22:189–196. PMID: 15262304.
Article5. Berner A. [Actinic keratosis and development of cutaneous squamous cell carcinoma]. Tidsskr Nor Laegeforen. 2005; 125:1653–1654. Norwegian. PMID: 15976832.6. Mittelbronn MA, Mullins DL, Ramos-Caro FA, Flowers FP. Frequency of pre-existing actinic keratosis in cutaneous squamous cell carcinoma. Int J Dermatol. 1998; 37:677–681. PMID: 9762818.
Article7. Kitagishi Y, Matsuda S, Minami A, Ono Y, Nakanishi A, Ogura Y. Regulation in cell cycle via p53 and PTEN tumor suppressors. Cancer Stud Mol Med Open J. 2014; 1:1–7.8. Neto PD, Alchorne M, Michalany NS, Abreu M, Borra R. Reduced P53 staining in actinic keratosis is associated with squamous cell carcinoma: a preliminary study. Indian J Dermatol. 2013; 58:325.
Article9. Kanellou P, Zaravinos A, Zioga M, Stratigos A, Baritaki S, Soufla G, et al. Genomic instability, mutations and expression analysis of the tumour suppressor genes p14(ARF), p15(INK4b), p16(INK4a) and p53 in actinic keratosis. Cancer Lett. 2008; 264:145–161. PMID: 18331779.
Article10. Li-Weber M, Treiber MK, Giaisi M, Palfi K, Stephan N, Parg S, et al. Ultraviolet irradiation suppresses T cell activation via blocking TCR-mediated ERK and NF-kappa B signaling pathways. J Immunol. 2005; 175:2132–2143. PMID: 16081779.11. Lee YB, Kim EK, Park HJ, Cho BK, Park YM, Kim JW, et al. Expression of Fas and Fas ligand in primary cutaneous squamous cell carcinoma in association with grade of tumor differentiation. Int J Dermatol. 2013; 52:1092–1097. PMID: 23045958.
Article12. Hameetman L, Commandeur S, Bavinck JN, Wisgerhof HC, de Gruijl FR, Willemze R, et al. Molecular profiling of cutaneous squamous cell carcinomas and actinic keratoses from organ transplant recipients. BMC Cancer. 2013; 13:58. PMID: 23379751.
Article13. Padilla RS, Sebastian S, Jiang Z, Nindl I, Larson R. Gene expression patterns of normal human skin, actinic keratosis, and squamous cell carcinoma: a spectrum of disease progression. Arch Dermatol. 2010; 146:288–293. PMID: 20231500.
Article14. Nindl I, Dang C, Forschner T, Kuban RJ, Meyer T, Sterry W, et al. Identification of differentially expressed genes in cutaneous squamous cell carcinoma by microarray expression profiling. Mol Cancer. 2006; 5:30. PMID: 16893473.15. Lambert SR, Mladkova N, Gulati A, Hamoudi R, Purdie K, Cerio R, et al. Key differences identified between actinic keratosis and cutaneous squamous cell carcinoma by transcriptome profiling. Br J Cancer. 2014; 110:520–529. PMID: 24335922.
Article16. S Al-MurraniX GaoJ LeeS MalladiNZ Frantz. Hill's Pet Nutrition, Inc.Methods for the diagnosis, control and prophylaxis of inflammation and mitigation of inflammatory conditions in felines. United States patent. EP 25,688,253. 2016. 4. 27.17. Tusher VG, Tibshirani R, Chu G. SAM (significance analysis of microarrays): permits statistical distinction between microarray signals. In : Rédei GP, editor. Encyclopedia of genetics, genomics, proteomics, and informatics. 3rd ed. Dordrecht: Springer;2008. p. 1755.18. Nicol NH. Actinic keratosis: preventable and treatable like other precancerous and cancerous skin lesions. Plast Surg Nurs. 1989; 9:49–55. PMID: 2675146.19. Cheng SY, Luk NM, Chong LY. Special features of non-melanoma skin cancer in Hong Kong Chinese patients: 10-year retrospective study. Hong Kong Med J. 2001; 7:22–28. PMID: 11406672.20. Monroig O, Rotllant J, Cerdá-Reverter JM, Dick JR, Figueras A, Tocher DR. Expression and role of Elovl4 elongases in biosynthesis of very long-chain fatty acids during zebrafish Danio rerio early embryonic development. Biochim Biophys Acta. 2010; 1801:1145–1154. PMID: 20601113.
Article21. Harris RB, Foote JA, Hakim IA, Bronson DL, Alberts DS. Fatty acid composition of red blood cell membranes and risk of squamous cell carcinoma of the skin. Cancer Epidemiol Biomarkers Prev. 2005; 14:906–912. PMID: 15824162.
Article22. Watanabe R, Fujii H, Yamamoto A, Hashimoto T, Kameda K, Ito M, et al. Immunohistochemical distribution of cutaneous fatty acid-binding protein in human skin. J Dermatol Sci. 1997; 16:17–22. PMID: 9438903.
Article23. Jones SJ, Dicker AJ, Dahler AL, Saunders NA. E2F as a regulator of keratinocyte proliferation: implications for skin tumor development. J Invest Dermatol. 1997; 109:187–193. PMID: 9242506.
Article24. Entrez Gene. HPGD 15-hydroxyprostaglandin dehydrogenase [Homo sapiens (human)] [Internet]. Bethesda, MD: NCBI;cited 2012 Sep 21. Available from: https://www.ncbi.nlm.nih.gov/gene?Db=gene&Cmd=DetailsSearch&Term=3248.25. Seo H, Choi Y, Shim J, Yoo I, Ka H. Comprehensive analysis of prostaglandin metabolic enzyme expression during pregnancy and the characterization of AKR1B1 as a prostaglandin F synthase at the maternal-conceptus interface in pigs. Biol Reprod. 2001; 90:99.
Article26. Okano M, Yamamoto H, Ohkuma H, Kano Y, Kim H, Nishikawa S, et al. Significance of INHBA expression in human colorectal cancer. Oncol Rep. 2013; 30:2903–2908. PMID: 24085226.
Article27. XU D, Yuan RH, Chu Y, Tang Y, Gu H, Zhang L, et al. Regulation of p53 expression though transforming growth factor β1 (TGFβ1)/Smads pathway in actinic keratosis. J Dermatol. 2012; 45:638–640.28. Li J, Zhang Y, Romagosa R, Saghari S, Elgart G, Miner JH, et al. Laminin 10 in the angiogenesis and invasion of squamous cell carcinoma. J Invest Dermatol. 2006; 124(Suppl):A21.29. Mostafa WZ, WZ , Mahfouz SM, Bosseila M, Sobhi RM, Zaki NS. An Immunohistochemical study of laminin in cutaneous and mucosal squamous cell carcinomas. J Egypt Wom Dermatol Soc. 2007; 4:24–33.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Histopathological Study of Accompanying Actinic Keratosis and Bowen's Disease at the Periphery or within the Confines of Cutaneous Squamous Cell Carcinoma
- Cutaneous Horn in Premalignant and Malignant Conditions
- Pigmented Squamous Cell Carcinoma Arising from Pigmented Actinic Keratosis
- Alteration of G1/S Cell Cycle Regulatory Proteins in Carcinogenesis of Cutaneous Squamous Cell Carcinomas
- Various Skin Tumors Originating from Disseminated Superficial Actinic Porokeratosis