Blood Res.
2017 Jun;52(2):95-99. 10.5045/br.2017.52.2.95.
Dapsone therapy for refractory immune thrombocytopenia patients: a case series
- Affiliations
-
- 1Department of Internal Medicine, Seoul National University Bundang Hospital, Seongnam, Korea. smbang7@snu.ac.kr
- 2Department of Internal Medicine, Dongtan Sacred Heart Hospital, Hallym University College of Medicine, Hwaseong, Korea. monique@hallym.or.kr
- KMID: 2413325
- DOI: http://doi.org/10.5045/br.2017.52.2.95
Abstract
- BACKGROUND
Dapsone has been recommended as a second-line immunosuppressive agent for patients with immune thrombocytopenia (ITP).
METHODS
We retrospectively analyzed the efficacy and safety of dapsone therapy in patients with ITP.
RESULTS
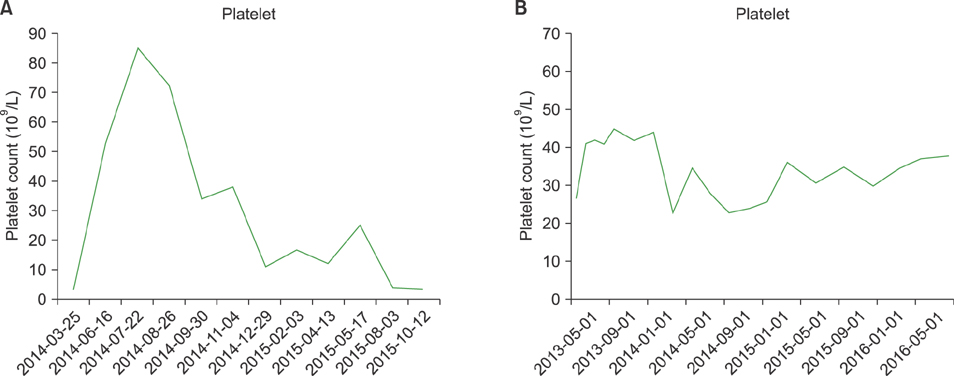
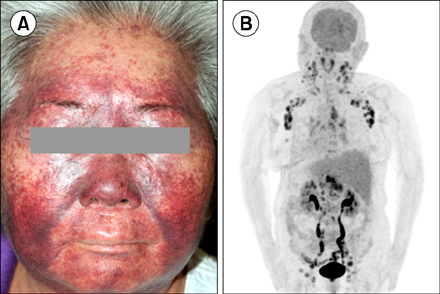
Nine ITP patients were treated with dapsone at a dose of 50-100 mg/day between May 2013 and March 2016. All patients were refractory to multiple previous treatments, with a median of 7 agents (range, 4-8), and 3 patients had undergone a previous splenectomy. The median pre-treatment platelet count was 4×10â¹/L (range, 3-27×10â¹/L). Only 1 patient (11.1%) responded to dapsone therapy. No severe adverse events were observed, except for 1 case of dapsone hypersensitivity syndrome.
CONCLUSION
Although dapsone is still useful for some patients, it may be ineffective in heavily pretreated patients with profound thrombocytopenia.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Dapsone therapy for immune thrombocytopenic purpura: old but still unfamiliar
Jaewoo Song
Blood Res. 2017;52(2):77-78. doi: 10.5045/br.2017.52.2.77.
Reference
-
1. Cines DB, Bussel JB, Liebman HA, Luning Prak ET. The ITP syndrome: pathogenic and clinical diversity. Blood. 2009; 113:6511–6521.
Article2. McMillan R, Wang L, Tomer A, Nichol J, Pistillo J. Suppression of in vitro megakaryocyte production by antiplatelet autoantibodies from adult patients with chronic ITP. Blood. 2004; 103:1364–1369.
Article3. Portielje JE, Westendorp RG, Kluin-Nelemans HC, Brand A. Morbidity and mortality in adults with idiopathic thrombocytopenic purpura. Blood. 2001; 97:2549–2554.
Article4. Godeau B, Porcher R, Fain O, et al. Rituximab efficacy and safety in adult splenectomy candidates with chronic immune thrombocytopenic purpura: results of a prospective multicenter phase 2 study. Blood. 2008; 112:999–1004.
Article5. Michel M, Suzan F, Adoue D, et al. Management of immune thrombocytopenia in adults: a population-based analysis of the French hospital discharge database from 2009 to 2012. Br J Haematol. 2015; 170:218–222.
Article6. Rodeghiero F, Ruggeri M. Is splenectomy still the gold standard for the treatment of chronic ITP? Am J Hematol. 2008; 83:91.
Article7. Neunert CE. Current management of immune thrombocytopenia. Hematology Am Soc Hematol Educ Program. 2013; 2013:276–282.
Article8. Ghanima W, Godeau B, Cines DB, Bussel JB. How I treat immune thrombocytopenia: the choice between splenectomy or a medical therapy as a second-line treatment. Blood. 2012; 120:960–969.
Article9. Cuker A, Cines DB, Neunert CE. Controversies in the treatment of immune thrombocytopenia. Curr Opin Hematol. 2016; 23:479–485.
Article10. Provan D, Stasi R, Newland AC, et al. International consensus report on the investigation and management of primary immune thrombocytopenia. Blood. 2010; 115:168–186.
Article11. Moss C, Hamilton PJ. Thrombocytopenia in systemic lupus erythematosus responsive to dapsone. BMJ. 1988; 297:266.
Article12. Damodar S, Viswabandya A, George B, Mathews V, Chandy M, Srivastava A. Dapsone for chronic idiopathic thrombocytopenic purpura in children and adults-a report on 90 patients. Eur J Haematol. 2005; 75:328–331.
Article13. Godeau B, Durand JM, Roudot-Thoraval F, et al. Dapsone for chronic autoimmune thrombocytopenic purpura: a report of 66 cases. Br J Haematol. 1997; 97:336–339.
Article14. Godeau B, Oksenhendler E, Bierling P. Dapsone for autoimmune thrombocytopenic purpura. Am J Hematol. 1993; 44:70–72.
Article15. Hernández F, Linares M, Colomina P, et al. Dapsone for refractory chronic idiopathic thrombocytopenic purpura. Br J Haematol. 1995; 90:473–475.
Article16. Rodeghiero F, Stasi R, Gernsheimer T, et al. Standardization of terminology, definitions and outcome criteria in immune thrombocytopenic purpura of adults and children: report from an international working group. Blood. 2009; 113:2386–2393.
Article17. Cuker A, Neunert CE. How I treat refractory immune thrombocytopenia. Blood. 2016; 128:1547–1554.
Article18. Durand JM, Lefèvre P, Hovette P, Mongin M, Soubeyrand J. Dapsone for idiopathic autoimmune thrombocytopenic purpura in elderly patients. Br J Haematol. 1991; 78:459–460.
Article19. Vancine-Califani SM, De Paula EV, Ozelo MC, Orsi FL, Fabri DR, Annichino-Bizzacchi JM. Efficacy and safety of dapsone as a second-line treatment in non-splenectomized adults with immune thrombocytopenic purpura. Platelets. 2008; 19:489–495.
Article20. Zaja F, Marin L, Chiozzotto M, Puglisi S, Volpetti S, Fanin R. Dapsone salvage therapy for adult patients with immune thrombocytopenia relapsed or refractory to steroid and rituximab. Am J Hematol. 2012; 87:321–323.
Article21. Hill QA. How does dapsone work in immune thrombocytopenia? Implications for dosing. Blood. 2015; 125:3666–3668.
Article22. Oo T, Hill QA. Disappointing response to dapsone as second line therapy for primary ITP: a case series. Ann Hematol. 2015; 94:1053–1054.
Article23. Rodrigo C, Gooneratne L. Dapsone for primary immune thrombocytopenia in adults and children: an evidence-based review. J Thromb Haemost. 2013; 11:1946–1953.
Article24. Sauvetre G, MahÈvas M, Limal N, et al. Cutaneous rash and dapsone-induced hypersensitivity syndrome a common manifestation in adult immune thrombocytopenia. Presentation and outcome in 16 cases. Am J Hematol. 2015; 90:E201–E202.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Advances in management of pediatric chronic immune thrombocytopenia: a narrative review
- Application of vincristine-loaded platelet therapy in three dogs with refractory immune-mediated thrombocytopenia
- Dapsone therapy for immune thrombocytopenic purpura: old but still unfamiliar
- Diagnostic Approach of Childhood Immune Thrombocytopenia
- Successful Treatment of Pyoderma Gangrenosum with Prednisolone, Cyclosporine, and Dapsone